Ninhydrin Test is a chemical test used to detect ammonia, primary amines, secondary amines and α-amino acids in a given sample. It is done by treating the sample with ninhydrin (2,2-dihydroxyindane-1,3-dione). The reagent acts as a strong oxidizing agent and the reaction is seen after heating.
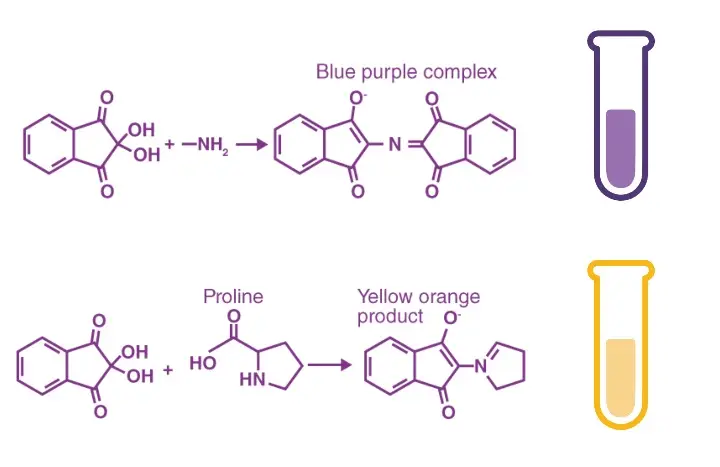
During this test, amino acids undergo oxidative deamination and decarboxylation. A deep purple or blue coloured product is formed which is called Ruhemann’s purple. The colour intensity is proportional to the concentration of amino acids present in the sample. In case of secondary amines (imino acids) like proline and hydroxyproline, yellow-orange complex is formed instead of purple.
Objectives of Ninhydrin Test
- To detect the presence of ammonia, primary amines, secondary amines and free α-amino acids in a given sample.
- To determine the concentration of amino acids and proteins by colour intensity (colorimetric or spectrophotometric estimation).
- To differentiate carbohydrates from amino acids in a test solution.
- To develop and visualize latent fingerprints on porous surfaces like paper, cardboard and untreated wood.
- To check the completeness of amino acid coupling and monitor deprotection in solid phase peptide synthesis (Kaiser test).
- To determine protein and total usable nitrogen content in food and beverage products like beer and wine.
- To detect and measure low level cyanide in industrial effluents and biological materials like blood.
- To aid in analysis, isolation and characterization of microbial products having ninhydrin reactive amino groups like antibiotics, bacterial toxins and other microbial products.
Principle of Ninhydrin Test
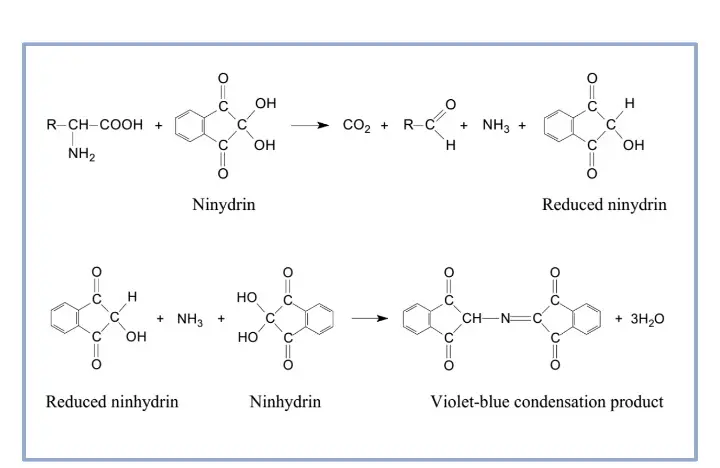
Principle of Ninhydrin Test is based on the reaction of ninhydrin with free α-amino acids and primary amines to produce a coloured complex. Ninhydrin acts as a strong oxidizing agent. On heating, oxidative deamination and decarboxylation of amino acid is caused.
During this process, aldehyde, CO2, ammonia and hydrindantin (reduced ninhydrin) are formed. The liberated ammonia then condenses with one molecule of hydrindantin and one molecule of unreacted ninhydrin. As a result, a deep purple or blue coloured complex (Ruhemann’s purple or diketohydrin complex) is formed. The intensity of the purple colour is proportional to the concentration of amino acids present in the sample.
Secondary amines (imino acids) like proline and hydroxyproline do not form Ruhemann’s purple. In this case a yellow-orange product (iminium salt) is produced.
Ninhydrin Test Reaction
Requirements for Ninhydrin Test
- Ninhydrin reagent (ninhydrin dissolved in ethanol. About 0.35 g in 100 ml). Isopropanol or acetone and butanol (1:1) or 2% w/v ninhydrin in ethanol/acetone can also be used.
- Diluent solvent (for quantitative test). It is prepared by mixing equal volume of deionized water and n-propanol.
- Standard solution (known concentration). It may be 1% protein solution or amino acid standard like leucine stock solution (example 1 mg/ml).
- Sample solution. It is the given test sample.
- Acetate buffer (to maintain pH). Example 0.2 M, pH 5.5 (as per protocol).
- Test tubes.
- Test tube stand.
- Pipettes.
- Water bath or heating block (for heating the reaction mixture around 90°C to 100°C or boiling for colour development).
- Spectrophotometer (for measuring absorbance of the coloured complex in quantitative estimation).
Procedure of Ninhydrin Test
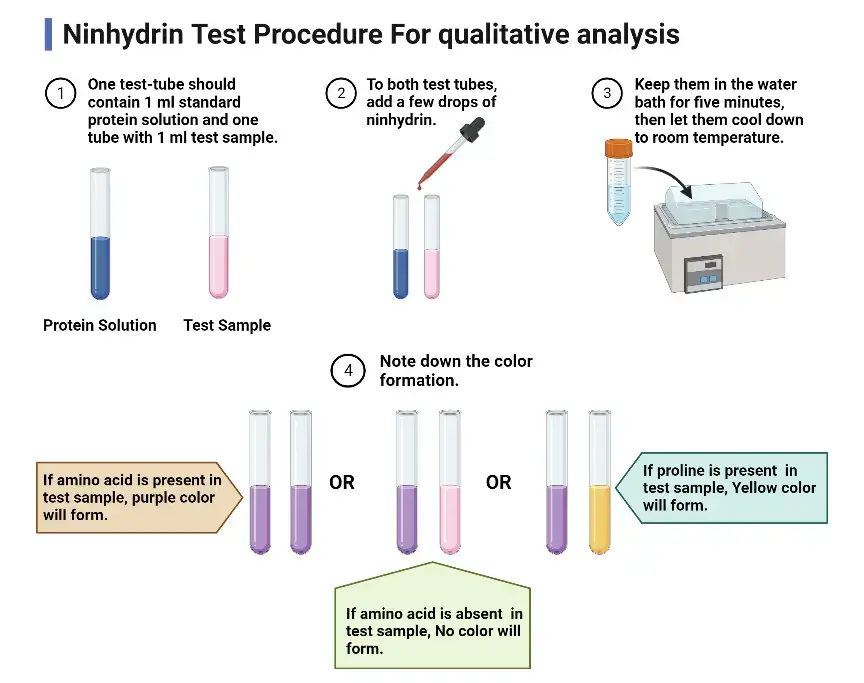
A. Qualitative analysis (simple detection)
- Take 1 mL of the test sample solution in a clean, dry test tube.
- Add a few drops of ninhydrin reagent (0.2–1.0% in ethanol or acetone). Mix the contents thoroughly.
- In this step place the test tube in a boiling water bath and heat for 5–10 minutes.
- Allow the test tube to cool to room temperature.
- Observe the colour development. Deep blue or purple is produced for free α-amino acids or primary amines. Yellow is produced for imino acids (e.g., proline).
B. Quantitative analysis (spectrophotometric measurement)
- Pipette 1.0 mL of the unknown sample solution into a labelled test tube.
- Prepare standards by placing varying volumes of a standard amino acid solution into a series of labelled test tubes. Add deionized water to each tube to make final volume 1.0 mL.
- Prepare a blank tube containing exactly 1.0 mL deionized water.
- Add 1.0 mL ninhydrin reagent to all tubes (standards, unknown, blank). Mix well (vortex).
- Cover tubes with caps or aluminium foil and heat. Use a boiling water bath for 15–20 minutes. In this step an alternative is heating in a block thermostat at 90°C for 45 minutes for improved colour stability.
- After incubation, cool tubes to room temperature under running cold water.
- Add diluent solvent (for example 5 mL of 1:1 n-propanol : deionized water) to each tube and mix well.
- Set spectrophotometer to zero absorbance at 570 nm using the blank.
- Measure absorbance of each standard and the unknown at 570 nm. For imino acids (proline, hydroxyproline) set to 440 nm.
- Plot standard curve of absorbance versus standard concentration. Use the linear regression equation from the standard curve to calculate the concentration of amino acids in the unknown sample.
The following are short notes
- The reaction is colour producing and used to detect primary amino groups.
- Heating is required for chromophore development.
- Use the same diluent and matched cuvettes for all measurements.
- A blank and standards must be run alongside the unknown for accurate quantitation.

Result of Ninhydrin Test
- Deep purple or blue (Ruhemann’s purple).
This is a standard positive result. It indicates presence of primary amines. It indicates free α-amino acids or ammonia in the sample. In forensic use this reaction makes latent fingerprints visible on paper. - Yellow or yellow-orange.
This result indicates presence of secondary amines or imino acids. Proline and hydroxyproline give this colour. An iminium salt is formed instead of the purple complex. - Brown or brown-purple.
This colour is produced specifically by the amino acid asparagine. It is a diagnostic colour for asparagine. - No colour change (colourless).
This is a negative result. No amino acids or amines are detected in the sample. - Kaiser test (peptide synthesis monitoring).
Dark blue beads and solution. This indicates failed or incomplete coupling. Unreacted free primary amines are present.
Colourless or yellow beads and solution. This indicates successful and complete coupling. No free primary amines remain.

Uses of Ninhydrin Test
- It is used to detect and quantify amino compounds (free α-amino acids, primary and secondary amines, ammonia).
- It is used to develop latent fingerprints on porous surfaces (paper, cardboard, unfinished wood) by reacting with sweat amino acids.
- It is used as the Kaiser test in solid-phase peptide synthesis to monitor coupling and deprotection.
- It is used as a spray reagent to locate amino acids and peptides on thin-layer chromatography or filter paper.
- It is used to estimate free amino acids, protein content, and total usable nitrogen in food and beverages (beer, wine, soy sauce, cheese, ripening fruits).
- It is used in spectrophotometric and densitometric assays to quantify certain drugs (for example gabapentin, baclofen, some α-aminocephalosporins).
- It is used to detect low levels of cyanide in alkaline media after appropriate adaptation.
- It is used in microbiological and enzymatic assays (for example estimating protease activity or detecting products of hippurate hydrolysis).
- It is used in paleontology and environmental isotope studies to release or trace carboxyl carbons from ancient collagen and to study microbial carbon assimilation.
- It is used as a quick go/no-go test for free primary amines in peptide chemistry.
Limitations of Ninhydrin Test
The following are some important limitations-
- Lack of specificity – Ninhydrin gives the same purple (Ruhemann’s) colour with almost all primary amino acids. Individual amino acid cannot be distinguished in a mixed sample.
- Free ammonia interference – Any trace ammonia present in water, air, or glassware is picked up by the reagent. The colour becomes deeper and false-positive value is obtained.
- Reaction with amine buffers – Buffers that contain primary amines (e.g. Tris, HEPES) also react. A strong coloured background is formed and reading is unusable.
- Poor response with large proteins – Intact high-molecular-weight and tightly folded proteins show little or no colour. Steric blockage hides the α-amino groups.
- Uneven colour yield for some amino acids – Secondary amines like proline, hydroxyproline give yellow-orange, asparagine may give brown. Different wavelengths are needed; mixed samples are over-estimated.
- Reagent instability – Prepared ninhydrin solution is light-sensitive and degrades quickly at room temperature. Fresh preparation or cold, dark storage is required.
- Strict reaction conditions – Colour intensity depends on exact pH, temperature, and time. Small deviation changes the absorbance value.
- Destructive in Kaiser (solid-phase peptide) test – The test consumes the resin sample, misses secondary amines, and shows false spots if Fmoc groups break down. Classic cocktail contains toxic KCN.
- Slow development in fingerprint work – At room temperature the reaction may need days. Heat and humidity are needed; some papers (e.g. newspaper) or dry prints fail.
- Health and safety hazard – Ninhydrin powder and vapour are flammable irritants. Repeated exposure may cause allergic rhinitis or occupational asthma in laboratory workers.
Advantages of Ninhydrin Test
- It is highly sensitive, so even minute free amino acid or faint latent print is visualised.
- The colour yield shows very small protein-to-protein difference; result is accurate and reproducible.
- The assay is cheap; chemicals are common and the reaction path is already well known.
- Development of colour is fast; many samples or many porous exhibits can be processed within short time.
- It is used in food check, clinical assay, peptide synthesis monitoring, and routine forensic work; same basic method suits all.
- Works uniformly on wide range of substrate; performance remains steady and dependable.
- Latent prints on porous sheet may still develop clearly even after long ageing, reported beyond fifty years.
Precautions of Ninhydrin Test
Some of the important precautions followed during the test are-
- Gloves and safety goggles are worn right from weighing reagent to final clean-up; the solution can blister skin and irritate eye.
- All spraying, dipping, and air-dry steps are done inside a working fume hood; its vapour is not breathed.
- The common ninhydrin stock is flammable, so no open flame, hot plate, or spark is kept near the bench.
- Only ammonia-free water, glassware, and filter paper are used; stray ammonium ion produces false purple colour.
- Fresh reagent is prepared each session or stored in a dark, cool bottle; light and room heat break it down fast.
- Kaiser test uses KCN and pyridine; the mix is handled under exhaust and waste is collected in cyanide jar.
- Spent solution, wipes, and rinse water are poured into a labelled hazardous-waste container, never into sink.
- Reagent kits are meant for laboratory study only; they are not used for household spot tests or drug checks.
- Hands are washed after work even when gloves were worn; tiny stains on skin may cause irritation later.
References
- AAPPTec. (n.d.). Monitoring of peptide coupling and capping; coupling tests.
- Aapptec Peptides. (n.d.). Technical support information bulletin 1188 – Kaiser test (ninhydrin test).
- Application of the ninhydrin reaction for quantification of total protein contents: Establishment of conversion formulas. (n.d.). PMC.
- BenchChem Technical Support Team. (2026). Application note & protocol: Quantitative analysis of amino acids using the ninhydrin method. BenchChem.
- BenchChem Technical Support Team. (2026). Ninhydrin reaction optimization: Technical support center. BenchChem.
- Bottom, C. B., Hanna, S. S., & Siehr, D. J. (n.d.). Mechanism of the ninhydrin reaction. Biochemical Education, 6(1), 4-5.
- Champod, C., Lennard, C., Margot, P., & Stoilovic, M. (2004). Fingerprints and other ridge skin impressions.
- Chemical development of latent fingerprints: 1,2-indanedione has come of age. (n.d.). PubMed.
- Comprehensive technical analysis of the ninhydrin reaction: Principles, mechanistic pathways, procedural methodologies, and multi-disciplinary applications. (n.d.).
- Dotterer, B. (2010). Processes to develop latent prints on porous surfaces. Caron Scientific.
- Drochioiu, G., Sandu, I., Olteanu, G. I., & Mangalagiu, I. (n.d.). Ninhydrin-based forensic investigations I. Fingerprints. International Journal of Criminal Investigation, 1(1), 37-58.
- Eason, J. (1995). Amino acids interfere with the ninhydrin assay for asparagine. Food Chemistry.
- Elgubbi, H. S. H. A. B. B. A. N. (n.d.). Colorimetric method for determination of amino acids on thin layer and filter paper chromatography using a modified ninhydrin reagent.
- Fish, J. T., Miller, L. S., Braswell, M. C., & Wallace, E. W. (2013). Crime scene investigation: Third edition.
- FUJIFILM Wako Chemicals. (2025). Amino acids analysis.
- Gardner, S., & Hewlett, D. F. (2003). Optimization and initial evaluation of 1,2-indandione as a reagent for fingerprint detection.
- Gerbig, D., & Fenk, C. (2007). A new colorimetric assay of tabletop sweeteners using a modified biuret reagent. An analytical chemistry experiment for the undergraduate curriculum. Journal of Chemical Education.
- Izreena, P. (n.d.). Lab report. Academia.edu.
- Jain, A. (2020). Identification tests for proteins (casein and albumin).
- Kennedy, I. (1965). Release of nitrogen from amino acids with ninhydrin for 15N analysis. Analytical Biochemistry.
- Kim, C., Cho, H.-W., Koh, H.-S., & Yu, J.-S. (2016). A study on application and comparison of effectiveness of ninhydrin reagents on special paper surfaces. The Journal of the Korea Contents Association, 16(9), 247-253. https://doi.org/10.5392/JKCA.2016.16.09.247
- Konno, H. (2020). Detection of N-terminal amino groups during solid-phase peptide synthesis. SYNFORM.
- Kundu, S. (2014). Modified ninhydrin spray reagent for the identification of amino acids on TLC plates.
- Lennard, C., Margot, P., Stoilovic, M., & Warrener, R. N. (1986). Synthesis of ninhydrin analogues and their application to fingerprint development: Preliminary results. Forensic Science Society Journal.
- Lennard, C., Margot, P., Stoilovic, M., & Warrener, R. N. (1988). Synthesis and evaluation of ninhydrin analogs as reagents for the development of latent fingerprints on paper surfaces. Forensic Science Society Journal.
- Lennard, C., Marriott, C., Lee, R., & Wilkes, Z. (2014). Evaluation of fingermark detection sequences on paper substrates. Forensic Science International.
- Macchi, F. (2003). Amino acid analysis, using postcolumn ninhydrin detection, in a biotechnology laboratory. Humana Press eBooks.
- Meesschaert, B. (1977). Separation of ninhydrin-positive compounds on a single-column amino acid analyzer using lithium buffers. Journal of Chromatography A.
- Merck KGaA. (2018). 60017 Kaiser test kit. Sigma-Aldrich.
- n, m. (1985). Measurement of protein using bicinchoninic acid. Analytical Biochemistry.
- Nikoui, K. (2024). Unveiling hidden clues: The science of ninhydrin fingerprint development. Nikoui and Associates.
- NJ Labs. (2023). Why is any viable amino acid technically ninhydrin positive substances?
- Odén, S., & Von Hofsten, B. (1954). Detection of fingerprints by the ninhydrin reaction.
- Optimisation and evaluation of 1,2-indanedione for use as a fingermark reagent and its application to real samples. (n.d.). PubMed.
- Parvin, R. (1965). On the colorimetric biuret method of protein determination. Analytical Biochemistry.
- Patel, R. (1949). Determination of serum proteins by means of the biuret reaction. J. biol. Chem.
- Pavon, R. (n.d.). Color reactions of intact protein and base hydrolyzate from casein.
- Pounds, C. A., Grigg, R., & Mongkolaussavaratana, T. (1990). The use of 1,8-diazafluoren-9-one (DFO) for the fluorescent detection of latent fingerprints on paper. A preliminary evaluation.
- Powers Scientific, Inc. (2015). Fingerprint detection with ninhydrin.
- Rajkhowa, S. (n.d.). Development of fingerprints. Haflong Government College.
- Robles, J., & Janairo, G. (n.d.). Determination of the sensitivity range of biuret test for undergraduate biochemistry experiments.
- Sapkota, A. (2022). Ninhydrin test- Definition, principle, procedure, result, uses. Microbe Notes.
- Spedding, G. (2009). Analysis of protein and total usable nitrogen in beer and wine using a microwell ninhydrin assay. Journal of the Institute of Brewing.
- Stauß, A. C., Fuchs, C., Jansen, P., Repert, S., Alcock, K., Ludewig, S., & Rozhon, W. (2024). The ninhydrin reaction revisited: Optimisation and application for quantification of free amino acids. Molecules, 29(14), 3262. https://doi.org/10.3390/molecules29143262
- Stoilovic, M. (1993). Improved method for DFO development of latent fingerprints. Forensic Science International.
- Verma, D., Pillai, V. N. R., & Tailor, G. (2020). Role of capping in peptide synthesis. International Journal of Research in Pharmaceutical Sciences, 11(4), 5225-5228. https://doi.org/10.26452/ijrps.v11i4.3134
- Wallace-Kunkel, C., Lennard, C., Stoilovic, M., & Roux, C. P. (2007). Optimisation and evaluation of 1,2-indanedione for use as a fingermark reagent and its application to real samples. Forensic Science International.
- Wikipedia contributors. (n.d.). Ninhydrin. In Wikipedia, The Free Encyclopedia.
- Wilkinson, D., Rumsby, D., Babin, B., Merritt, M., & Marsh, J. (2005). The results from a Canadian national field trial comparing 1,8-diazafluoren-9-one (DFO) with ninhydrin and the sequence DFO followed by ninhydrin (Technical Report TR-03-2005). Canadian Police Research Centre.