Nelson-Somogyi method is a classical colorimetric method used for quantitative estimation of reducing sugars like glucose in biological and industrial samples. It is a two step redox based assay where reducing sugars act as reducing agent and reaction is carried out in alkaline condition.
In first step, sample containing reducing sugar is heated with alkaline copper tartrate reagent. Here cupric ion (Cu²⁺) is reduced to cuprous ion (Cu⁺) and cuprous oxide precipitate is formed. In second step, arsenomolybdate reagent is added which is reduced by cuprous ion and stable blue coloured complex (molybdenum blue) is produced.
The intensity of blue colour formed is directly proportional to the amount of reducing sugar present in the sample. Absorbance is measured in spectrophotometer generally between 500 to 540 nm and concentration is calculated by comparing with standard glucose curve. It is also used in enzyme assays because it gives almost equal colour response for equimolar amount of different oligosaccharides and so glycosidic bond cleavage can be measured without overestimation like DNS method.
Principle of Nelson-Somogyi method
Principle of Nelson-Somogyi method is based on the reduction of cupric (Cu²⁺) ions by reducing sugars in alkaline copper-tartrate medium. The sugar donates electrons. Cupric ions are changed to cuprous (Cu⁺) oxide and a red precipitate appears.
The freshly formed cuprous oxide then reacts with arsenomolybdate reagent. Molybdenum is reduced step-wise. A stable blue molybdenum complex is produced and stays in solution.
The depth of this blue colour rises with more reducing sugar present. The absorbance is read in a colorimeter or spectrophotometer. The value is compared with standards, and the amount of reducing sugar is calculated from the colour intensity.
Objectives of Nelson-Somogyi method
- To quantify reducing sugars in biological, food, and industrial samples.
- To obtain true blood glucose value after deproteinization removes non-sugar reducers.
- To measure activity of carbohydrase enzymes (amylase, cellulase, xylanase) by counting glycosidic bonds cleaved.
- To monitor reducing sugar level during quality control of dairy, beverages, fruit processing, and brewing fermentation.
- To estimate sugars released in enzymatic hydrolysis of lignocellulosic biomass for biofuel studies.
Requirements for Nelson-Somogyi method
A. Equipment and materials
- Spectrophotometer or colorimeter (to take absorbance between 500 nm to 540 nm).
- Cuvettes (for keeping the final blue solution during reading).
- Boiling water bath (for heating at 100°C during copper reduction step).
- Cold water bath or ice bath (for rapid cooling of tubes after boiling).
- Glass test tubes (narrow type, Folin-Wu tube or similar size tubes like 16 × 120 mm).
- Test tube rack or holder (to keep tubes in order).
- Pipettes or micropipettes with tips (for accurate measurement of sample and reagents).
- Vortex mixer or stirrer (for proper mixing and for dissolving cuprous oxide after adding colour reagent).
- Test tube covers (glass bulbs, cotton wool, parafilm etc. for covering tubes during heating).
B. Reagents and chemicals
- Somogyi alkaline copper tartrate reagent (copper reagent).
It is prepared by using-
- Anhydrous sodium carbonate.
- Potassium sodium tartrate (Rochelle salt).
- Sodium bicarbonate.
- Anhydrous sodium sulfate.
- Copper sulfate pentahydrate.
- Nelson arsenomolybdate colour reagent (to produce molybdenum blue colour).
It is prepared by using-
- Ammonium molybdate.
- Concentrated sulfuric acid.
- Sodium arsenate (disodium hydrogen arsenate heptahydrate).
- Standard reducing sugar solution (usually glucose standard for standard curve).
- Distilled water (for blank, dilution and reagent preparation).
- Deproteinizing reagents (only when blood or protein rich sample is used)- Barium hydroxide. Zinc sulfate.
Procedure of Somogyi Method (Nelson-Somogyi)
- Label test tubes for standards, test sample and blank. Pipette fixed volume of glucose standard solutions into separate tubes. Pipette same volume of unknown sample into another tube. Blank is prepared by taking same volume of distilled water.
- Add equal volume of alkaline copper tartrate reagent (Somogyi copper reagent) to each tube. Mix properly by shaking or vortex.
- Keep all tubes in boiling water bath for fixed time (generally 10 to 20 minutes). In this step reducing sugars reduce copper and cuprous oxide is formed.
- Remove the tubes from water bath and cool immediately in cold water or ice bath. This step is done to stop reaction and prevent reoxidation of cuprous ion by oxygen.
- Add arsenomolybdate colour reagent (Nelson reagent) to each tube. Mix vigorously so that the cuprous oxide precipitate gets dissolved completely.
- Allow the tubes to stand for about 5 to 10 minutes at room temperature or at 37°C for full development of molybdenum blue colour.
- Make up the volume by adding distilled water (as per protocol like 5 ml, 10 ml or 25 ml). Mix well so final colour becomes uniform.
- Take absorbance of standards and unknown against blank in spectrophotometer at 520 nm or 540 nm.
- Prepare standard curve by plotting absorbance vs concentration of glucose standard. From this curve calculate the concentration of reducing sugar in unknown sample.
Calculation
Calculation of Somogyi Method
A. Iodometric titration calculation (Somogyi original)
In this method the final titration reading is taken as T (in cc). This T value is used for calculating glucose.
Basic equivalent.
Every 1 cc of T is equal to 0.135 mg of glucose.
If 5 cc of 1:10 blood filtrate is used (this represents 0.5 cc blood).
Blood sugar (mg per 100 cc) = 27 × T.
If 2 cc of 1:10 blood filtrate is used.
Blood sugar (mg per 100 cc) = 67.5 × T.
Micromethod using 0.2 cc of blood.
Blood sugar (mg per 100 cc) = 108 × T.
Micromethod using 0.1 cc of blood.
Blood sugar (mg per 100 cc) = 216 × T.
B. Colorimetric / spectrophotometric calculation (Nelson-Somogyi)
In this method titration is not done. The blue colour (molybdenum blue) intensity is measured by absorbance.
- Prepare glucose standards of known concentration. Take absorbance of each standard at suitable wavelength (generally 500 to 540 nm) against blank.
- Plot standard curve.
Concentration of glucose is taken on X-axis.
Absorbance is taken on Y-axis.
A straight line is obtained within working range.
- Take absorbance of unknown sample at same wavelength against blank.
- Find concentration of unknown from standard curve. If equation is used then-
y = mx + b
where y = absorbance of unknown.
x = concentration of glucose (unknown).
So, x = (y − b) / m.
- Apply dilution factor and sample factor.
Final reducing sugar in original sample = (value obtained from curve) × dilution factor (and any volume correction used in sample preparation).
Result is expressed as mg/dl or mg per 100 ml or mg per given sample volume as required.
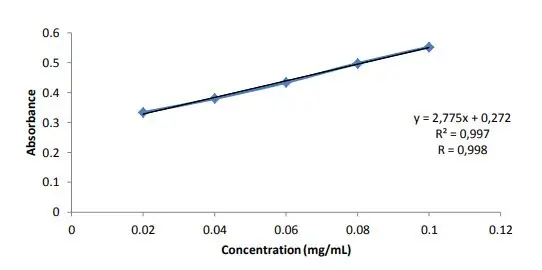
Result of Somogyi Method
In Somogyi (Nelson-Somogyi) method, molybdenum blue colour complex is finally formed after adding arsenomolybdate reagent. The blue colour is stable and intense.
The intensity of blue colour is measured in spectrophotometer generally between 500 to 540 nm. The absorbance value obtained is directly proportional to the initial concentration of reducing sugar present in the sample.
The absorbance of unknown is compared with standard glucose curve. From the standard calibration curve the exact concentration of reducing sugars in unknown sample is calculated and expressed as mg/dl or mg per 100 ml.
In clinical blood estimation, when barium hydroxide and zinc sulfate deproteinization is used, non-sugar reducing substances like glutathione and uric acid are removed. So the final result gives more true glucose value.
In enzyme assays, the result gives stoichiometric molar response even for different length oligosaccharides. So it reflects the number of glycosidic bonds cleaved by enzymes and does not show gross overestimation like DNS method.
Uses of Somogyi Method
- It is used to determine the blood sugar (glucose) level in clinical samples. It is known for giving more true glucose value because interfering non-sugar reducing substances are removed during deproteinization step.
- It is used in enzymology for estimating activity of carbohydrase enzymes (endo-xylanase, cellulase etc.). It helps in measuring number of glycosidic bonds cleaved because it gives consistent molar response for different oligosaccharides.
- It is used in food and beverage analysis for reducing sugar estimation. It is applied for monitoring lactose in milk, sugar content in fruit processing and reducing sugars in carbonated beverages for quality control.
- It is used in fermentation studies to measure reducing sugar in wort. It helps in controlling fermentation process and indirectly helps in deciding final alcohol production.
- It is used in biomass conversion studies to quantify sugars released during enzymatic hydrolysis of lignocellulosic materials (cellulose and hemicellulose). It is useful in biofuel related research works.
Advantages of Somogyi Method
- It gives consistent molar colour response for equimolar amount of different oligosaccharides. So measurement of cleaved glycosidic bonds can be done more accurately and it avoids gross overestimation as seen in DNS method.
- The molybdenum blue colour formed is highly stable. The colour can remain for several hours, so absorbance reading can be taken later also and it improves reproducibility between labs.
- Alkaline copper reagent contains high concentration of sodium sulfate. This decreases oxygen solubility and protects cuprous ions from reoxidation by atmospheric air, so result becomes more reliable.
- It gives more true glucose value in clinical samples due to deproteinization step. Barium hydroxide and zinc sulfate remove proteins and also remove some non-sugar reducing interferents like glutathione and uric acid, so physiological blood glucose reading becomes accurate.
- The method is sensitive and reproducible. It follows Beer-Lambert law in a wide concentration range and gives consistent results even at very low sugar concentration.
- It is versatile in application. It can be used for iodometric as well as colorimetric determination of cuprous oxide, and same reagent can be used for macro analysis and micro analysis.
- The procedure is comparatively simple to perform. Even with good accuracy and stoichiometric reliability, it can be carried out in routine way like other reducing sugar tests.
Limitations of Somogyi Method
- The Somogyi method is not strictly specific to reducing sugar only. It is based on copper reduction, so any strong reducing substance present in the sample can also reduce copper and give positive reading. Ascorbic acid (vitamin C), uric acid, glutathione and some drugs (streptomycin etc.) can interfere and cause false high sugar value.
- The color reagent used in Nelson-Somogyi step contains arsenomolybdate reagent. Sodium arsenate is used for preparation which is highly toxic. This creates health hazard to lab personnel and disposal becomes difficult, as it needs special hazardous waste handling.
- The cuprous ion (Cu⁺) formed during the reaction is unstable in air. Atmospheric oxygen can reoxidize Cu⁺ back to Cu²⁺ if cooling is not done quickly after boiling. In such condition the final reading becomes low and sugar concentration may be underestimated.
- Deproteinization step is critical especially for blood samples. Zinc sulfate and barium hydroxide must be added in exact equivalent amount to precipitate proteins properly. If excess zinc or barium remains in filtrate, it can reduce the final sugar reading, and barium hydroxide also deteriorates by absorbing CO₂ from air, so frequent standardization is needed.
- The method can be affected by optical and sample interferences. Hemolysed samples and icteric samples (high bilirubin) can disturb the colour development and spectrophotometric absorbance reading. This gives wrong result in clinical samples.
- The procedure is lengthy and cumbersome. It requires multiple steps with boiling water bath and rapid cooling in ice, and careful timing. It is less convenient and generally less sensitive compared to modern enzymatic methods (glucose oxidase) and chromatographic methods (HPLC).
Precautions of Somogyi Method
- The arsenomolybdate colour reagent contains arsenic, so it is highly toxic and poisonous. Gloves, lab coat and mask should be used and work should be done in fume hood or well ventilated place. Arsenic waste should not be thrown in normal waste and it must be disposed as hazardous waste.
- After heating in boiling water bath, the test tubes should be cooled immediately in cold water or ice bath. This is done to stop further reaction and to prevent reoxidation of cuprous ion (Cu⁺) into cupric ion (Cu²⁺) by atmospheric oxygen.
- Test tubes should be covered during boiling step by cotton plug, glass bulb or parafilm. It reduces evaporation loss and also reduces entry of oxygen. This is more important when low glucose concentration is tested.
- During deproteinization of blood or biological samples, barium hydroxide and zinc sulfate should be added in exact balanced volume. If excess acid or alkali remains in filtrate, the alkaline copper reduction step will get affected and final value becomes wrong.
- Samples should be free from other reducing substances as far as possible. Ascorbic acid, glutathione, uric acid and some antibiotics can also reduce copper and give false high result. Proper sample preparation and filtration should be done to remove these pseudochromogens.
- While preparing arsenomolybdate reagent (heating at around 55°C), proper stirring should be done and local overheating should be avoided. Overheating can decompose the chromogenic compound and yellow precipitate may form.
- Alkaline copper reagent should contain sufficient anhydrous sodium sulfate. High salt concentration reduces dissolved oxygen and protects cuprous ion from reoxidation during the reaction.
- Cuvettes should be handled carefully by the upper edge or frosted side only. Finger prints and scratches on optical surface will change absorbance reading.
- Accurate pipetting of standards, sample and reagents should be done. Colour intensity depends on sugar concentration, so small pipetting error can change the standard curve and final calculation.
References
- BenchChem Technical Support Team. (n.d.). Analytical evolution and technical implementation of the Nelson-Somogyi method for carbohydrate quantification. BenchChem.
- BenchChem Technical Support Team. (2025). An in-depth technical guide to the chemical composition of molybdenum blue (and related technical guides). BenchChem.
- BenchChem Technical Support Team. (2026). The molybdenum blue assay: A comprehensive technical guide. BenchChem.
- BMH learning. (n.d.). Estimation of reducing sugars by Nelson Somogyi method | Estimation of glucose by Nelson Somogyi | [Video]. YouTube.
- Cannadianti, N. (n.d.). Nelson-Somogyi method and result. Scribd.
- elleason. (n.d.). Nelson-Somogyi method for glucose analysis. Scribd.
- Expertsmind.com. (n.d.). Explain the principle of nelson-somogyi method, biology. Expertsmind.
- Irick, T., West, K., Brownell, H. H., & Schwald, W. (1988). Comparison of colorimetric and HPLC techniques for quantitating the carbohydrate components of steam-treated wood – Scientific note. Applied Biochemistry and Biotechnology, 17(1), 137-149. https://doi.org/10.1007/BF02779152.
- Kaczmarek, M. (n.d.). Estimation of reducing sugar by Nelson-Somogyi method.
- King Saud University. (n.d.). Determination of reducing sugars by Somogyi-Nelson method (Experiment 7, CLS 281).
- McCleary, B. V., & McGeough, P. (2015). A comparison of polysaccharide substrates and reducing sugar methods for the measurement of endo-1,4-β-xylanase. Applied Biochemistry and Biotechnology, 177(5). https://doi.org/10.1007/s12010-015-1803-z.
- Muthia, R., Afra, F. Y., Nurmansyah, D., Fauzana, E., & Istiqamah, Q. (2025). In vitro antidiabetes activity test of 96% ethanol extract from Melastoma malabathricum L. leaves using maceration method. Journal of Pharmaceutical and Sciences, 8(3), 1754-1764. https://doi.org/10.36490/journal-jps.com.v8i3.991.
- Nelson, N. (1944). A photometric adaptation of the Somogyi method for the determination of glucose. Journal of Biological Chemistry, 153, 375-380.
- Quilaneta, J. J. (n.d.). Nelson-Somogyi method for carbohydrate analysis. Scribd.
- Ramlan, A. (n.d.). Glucose estimation via Nelson-Somogyi method. Scribd.
- Schwald, W., Chan, M., Breuil, C., & Saddler, J. N. (1988). Comparison of HPLC and colorimetric methods for measuring cellulolytic activity. Applied Microbiology and Biotechnology, 28(4), 398-403. https://doi.org/10.1007/BF00268203.
- ScribdTranslations. (n.d.). Somogyi-Nelson method for sugar analysis. Scribd.
- Sharma, N. C., Sur, B. K., & Shukla, R. K. (1972). A simplified technique for estimation of blood glucose. Indian Journal of Physiology and Pharmacology, 16(4), 349-353.
- Somogyi, M. (1945). Determination of blood sugar. Journal of Biological Chemistry, 160(1), 69-73. https://doi.org/10.1016/S0021-9258(18)43098-0.
- Study.com. (n.d.). What is the theory behind Nelson-Somogyi’s method? Homework.Study.com.
- Uchida, M., Nakatani, K., Ono, M., & Nagami, K. (1991). Carbohydrates in brewing. I. Determination of fermentable sugars and oligosaccharides in wort and beer by partition high-performance liquid chromatography. Journal of the American Society of Brewing Chemists, 49(2), 65-73. https://doi.org/10.1094/ASBCJ-49-0065.
- ujwalp_1. (n.d.). Nelson-Somogyi method for sugar analysis. Scribd.
- Unknown. (n.d.). Preparation Nelson-Somogyi reagents.
- Wikipedia contributors. (n.d.). Blood sugar level. In Wikipedia, The Free Encyclopedia.
- Wikipedia contributors. (n.d.). Molybdenum blue. In Wikipedia, The Free Encyclopedia.
Nelson-Somogyi method can be used to quantify the reduction of sugar using arsenolmolibdat and copper reagents. The principle behind the Nelson Somogyi method is the quantity of deposro oxide deposits which react with arsenomolibdate, which reduces to molybdine blue. The blue hue is determined by as absorbance.
The keto and aldehyde free groups are regularly studied when reducing sugars. Lactose, galactose and maltose are all examples of sugars that are reduced. The cyclic form has to first break the ring to create reactive aldehyde prior to the process of oxidation can begin.