What is Lane-Eynon Method?
The Lane–Eynon method is a volumetric titration technique used for the quantitative estimation of reducing sugars in food and dairy products. It is commonly used for the determination of sugars such as dextrose, maltose, and lactose. This method is based on the reduction of Fehling’s solution by reducing sugars under boiling conditions. It is simple, economical and widely employed in sugar analysis laboratories.
The principle of this method depends on the reducing property of sugars. Reducing sugars reduce alkaline copper sulphate present in Fehling’s solution to form an insoluble brick–red precipitate of cuprous oxide (Cu₂O). Fehling’s solution consists of two components, Fehling’s solution A (copper sulphate) and Fehling’s solution B (alkaline sodium potassium tartrate). When the sample containing reducing sugars is added to the boiling Fehling’s solution, cupric ions (Cu²⁺) are reduced to cuprous ions (Cu⁺), resulting in the formation of cuprous oxide precipitate. The reaction is as follows–
Reducing sugar + 2Cu²⁺ + 5OH⁻ → Cu₂O + oxidized sugar + 3H₂O
To determine the exact end point of the titration, methylene blue is used as an internal oxidation–reduction indicator. As long as unreduced copper ions remain, the solution appears blue. When all the copper ions are reduced, methylene blue is decolourized and the brick–red precipitate becomes clearly visible, indicating the end point of the titration.
In this method, a known volume of Fehling’s solution is taken in a conical flask and heated to boiling. The standard sugar solution is titrated against the boiling Fehling’s solution until the blue colour almost disappears. In this step, methylene blue indicator is added and the titration is continued until the blue colour disappears completely. The appearance of a brick–red precipitate indicates the end point. The volume of sugar solution required is noted and used for the calculation of reducing sugar content.
The Lane–Eynon method is widely used for the determination of reducing sugars in food products, dairy samples, fruit juices and beverages. It is also used in quality control of sugar and fermentation industries. However, the method does not differentiate between individual reducing sugars and gives the total reducing sugar content. The accuracy of the method depends on strict control of boiling time, temperature and reagent concentration.
Objectives of Lane–Eynon Method
- To determine the concentration of reducing sugars such as glucose (dextrose) and maltose in various food samples.
- To estimate the lactose content in milk and other dairy products.
- To measure the concentration of sucrose in syrups, sugar preparations and liquid pharmaceutical formulations after hydrolysis into invert sugar.
- To determine the reducing power of starch hydrolysis products for calculating the Dextrose Equivalent (DE).
- To estimate the total reducing sugars and starch content in food materials after converting starch into reducing sugars by acid hydrolysis.
Principle of Lane-Eynon Method
The principle of the Lane–Eynon method is based on the reducing property of sugars. It is the process by which reducing sugars reduce a known volume of Fehling’s solution under standard boiling conditions. This method is employed for the quantitative estimation of reducing sugars in various food and dairy samples.
Fehling’s solution is a mixture of copper sulphate (Fehling’s solution A) and alkaline sodium potassium tartrate or Rochelle salt (Fehling’s solution B). Rochelle salt acts as a complexing agent which keeps the cupric ions in soluble form in alkaline medium. When the sugar solution is added from the burette into the boiling Fehling’s solution, the aldehyde groups of reducing sugars are oxidized into carboxyl groups, while the blue cupric ions (Cu²⁺) are reduced to brick–red cuprous oxide (Cu₂O) precipitate.
The reaction is as follows–
Reducing sugar + 2Cu²⁺ + 5OH⁻ → Cu₂O (brick–red precipitate) + oxidized sugar + 3H₂O
To detect the exact end point of the titration, methylene blue is used as an internal oxidation–reduction indicator. As long as unreduced cupric ions remain in the solution, the blue colour persists. When all the copper ions are completely reduced, the next drop of the sugar solution reduces the methylene blue indicator itself, causing the solution to lose its blue colour and become colourless, clearly revealing the bright brick–red precipitate. This is referred to as the end point of the Lane–Eynon titration.
Requirements for Lane-Eynon Method
Equipment and Apparatus
- Burette with off–set tip – A 50 mL burette is used to dispense the sugar solution. The off–set tip helps to keep the burette away from the steam generated during boiling.
- Volumetric flasks – Volumetric flasks of different capacities (100 mL, 200 mL, 500 mL, and 1000 mL) are used for preparing standard sugar solutions and dilutions.
- Conical (Erlenmeyer) flasks – Conical flasks of 250 mL or 300 mL capacity are used to carry out the boiling titration with Fehling’s solution.
- Heating source – A hot plate or gas burner with wire gauze or asbestos mat is used to maintain a uniform and continuous boiling of the solution.
- Anti–bumping agents – Glass beads, pumice powder or boiling chips are added to ensure smooth boiling and to prevent superheating and bumping of the solution.
- Analytical balance – A precision balance is used for accurate weighing of sugar standards and samples.
- Filtration apparatus – Filter paper (Whatman No. 1, 42 or 91), funnel and filtration setup are used for clarification of samples and removal of interfering substances.
Chemicals and Reagents
- Fehling’s Solution A – An aqueous solution of copper sulphate (CuSO₄·5H₂O) which provides cupric ions (Cu²⁺) acting as an oxidizing agent.
- Fehling’s Solution B – A strongly alkaline solution containing sodium potassium tartrate (Rochelle salt) and sodium hydroxide. The tartrate acts as a complexing agent to keep copper ions in solution.
- Methylene Blue Indicator – A 1% aqueous solution used as an internal oxidation–reduction indicator to detect the end point of titration.
- Standard Sugar Solution – Standard solutions of glucose (dextrose), invert sugar or lactose are used for standardization of Fehling’s solution.
- Clarification reagents – Reagents such as 10% acetic acid, Carrez reagents (potassium hexacyanoferrate(II) and zinc sulphate), or neutral lead acetate with potassium oxalate are used to remove proteins and fats from complex samples.
- Optional reagents – Liquid paraffin is used to prevent foaming, and EDTA (ethylene diamine tetra–acetic acid) is used to prevent calcium interference.
Procedural and Environmental Conditions
- Two–stage titration process – The method involves a preliminary titration to determine approximate titre value followed by a final accurate titration.
- Controlled boiling time – The solution is rapidly brought to boiling within 2–4 minutes and maintained at a steady boil for a fixed duration to ensure accuracy.
- Exclusion of atmospheric oxygen – Continuous boiling is maintained during titration so that steam expels air and prevents re–oxidation of cuprous oxide to cupric ions.
- Accurate visual end–point detection – The end point is determined by the complete disappearance of blue colour of methylene blue leaving a brick–red precipitate of cuprous oxide. Artificial or indirect lighting is preferred for clear observation.
Procedure of Lane–Eynon Method
- Preparation of test solution – The sample is clarified by precipitating and filtering the interfering substances such as proteins and fats. The clear filtrate is used as the test solution for titration.
- Preparation of Fehling’s solution – Exactly 5 mL of Fehling’s solution A and 5 mL of Fehling’s solution B are pipetted into a 200 mL or 250 mL conical (Erlenmeyer) flask.
- Addition of distilled water and anti–bumping agents – A small quantity of distilled water and a few glass beads or pumice stones are added to ensure smooth and uniform boiling of the solution.
- Filling of burette – The burette is filled with the prepared sugar solution and an initial volume (about 10–15 mL) is added into the flask.
- Boiling of the mixture – The flask is heated on a hot plate or burner and the mixture is brought to a moderate boil.
- Addition of indicator – In this step, 3–4 drops of methylene blue indicator is added to the boiling solution.
- Preliminary titration – The sugar solution is added dropwise from the burette without interrupting the boiling process until the blue colour disappears and a brick–red precipitate of cuprous oxide is formed. The approximate titre value is noted.
- Preparation for final titration – A fresh flask containing 5 mL of Fehling’s solution A and 5 mL of Fehling’s solution B is prepared for the final titration.
- Addition of sample before boiling – The test solution is added from the burette into the cold flask up to about 0.5–1.0 mL less than the titre obtained in the preliminary titration.
- Controlled boiling – The mixture is brought to boiling and maintained at a steady boil for exactly 2 minutes.
- Addition of methylene blue indicator – Without removing the flask from heat, 3–4 drops of methylene blue indicator are added.
- Completion of final titration – The titration is completed by adding the sugar solution dropwise (approximately one drop every two seconds) until the indicator is decolourized and a bright brick–red precipitate appears.
- Maintenance of boiling time – The final titration is completed within a maximum boiling time of 3 minutes to ensure accuracy of the reaction.
- Recording of titre value – The final volume of the sugar solution required to reach the end point is recorded and used for the calculation of reducing sugar content in the sample.
Results of Lane-Eynon Method
The results of the Lane–Eynon method are expressed in terms of reducing sugar content based on the volume of the sample required to completely reduce a standardized Fehling’s solution. It is the process by which the concentration or percentage of sugars present in the sample is determined using standard Lane–Eynon factor tables.
Direct Reducing Sugars
The method is used for the quantitative estimation of direct reducing sugars such as dextrose, maltose and lactose. The concentration of these sugars is calculated from the volume of the test solution required to reduce a known volume of Fehling’s solution.
Total Reducing Sugars
The total reducing sugar content of a sample is determined after hydrolysis of non–reducing sugars into reducing sugars by acid inversion. The result represents the overall reducing sugar content present in the sample.
Sucrose Content
Sucrose is a non–reducing sugar and is estimated indirectly. It is calculated by determining the reducing sugar content before and after inversion and multiplying the difference by a specific conversion factor.
Lactose Content
In milk and dairy products, the lactose content is calculated from the titration results using the standardization factor of Fehling’s solution and the volume of clarified milk filtrate required to reach the end point.
Dextrose Equivalent (DE)
For starch hydrolysis products, the dextrose equivalent is determined using the titration value, weight of the sample, percentage of dry solids and a suitable conversion factor obtained from standard tables.
Starch Content
Starch content is estimated by first hydrolyzing starch into glucose using strong acid, followed by titration of the resulting reducing sugars. The starch content is then calculated using a theoretical conversion factor.
Total Reducing Power (Limitation)
The final results represent the total reducing sugars or overall reducing power of the sample. This method does not differentiate between individual reducing sugars when they are present in a mixture, and therefore provides the cumulative reducing sugar content.
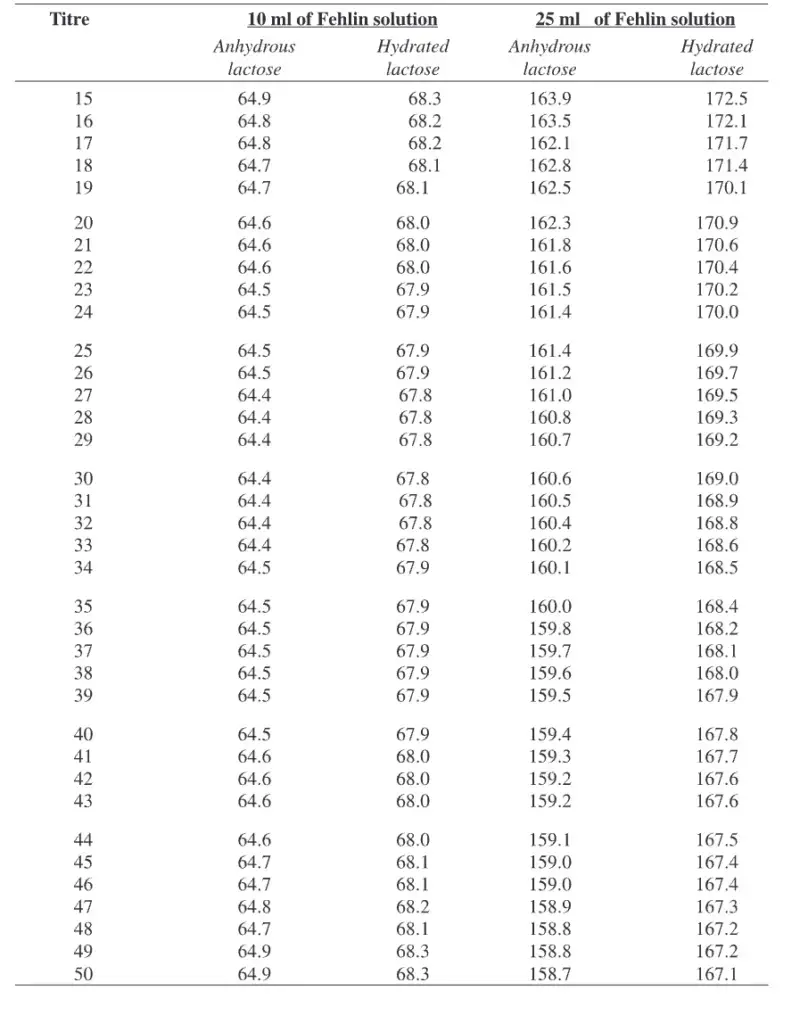
Factor for Fehlin solution to be used in correction with the Lane-Eynon general volumetric method.
Uses of Lane-Eynon Method
- To determine the concentration of direct reducing sugars or total reducing sugars in various samples.
- To estimate the lactose content in milk, milk powder, infant formula and other dairy products.
- To quantify the sucrose content in milk products, infant foods and sugar preparations after hydrolysis into invert sugar.
- To estimate the starch content in food materials after complete hydrolysis into glucose by strong acids.
- To perform routine analysis of fruit juices, beverages, jams and preserves for determining their reducing and total sugar content.
- To determine the reducing sugar content in dextrin and starch degradation products for calculating the Dextrose Equivalent (DE).
Advantages of Lane-Eynon Method
- It requires minimal equipment and does not need specialized or expensive instrumentation.
- It is a cost–effective analytical method and is suitable for routine laboratory analysis.
- The method utilizes simple and easily prepared chemical reagents such as Fehling’s solution and methylene blue indicator.
- It is a well–established and historically validated technique used as a standard reference method in food and sugar analysis.
- It is simple to perform and can be applied for the estimation of reducing sugars in a wide variety of food and dairy samples.
Limitations of Lane–Eynon Method
- It lacks specificity and measures only total reducing sugars without differentiating between individual monosaccharides and disaccharides such as glucose, fructose or lactose.
- It cannot directly estimate non–reducing sugars such as sucrose and therefore requires prior hydrolysis into reducing sugars.
- The method is susceptible to interference from other reducing substances present in the sample matrix which may affect the accuracy of the results.
- High concentrations of non–reducing sugars may undergo inversion during boiling, resulting in overestimation of reducing sugar content.
- The accuracy of the method is strictly dependent on controlled reaction time, boiling rate, temperature and precise reagent concentration.
- Interruption of continuous boiling allows atmospheric oxygen to enter the flask which may re–oxidize cuprous oxide (Cu₂O) into cupric ions (Cu²⁺), leading to underestimation of sugar content.
- The end point detection is subjective and depends on visual observation of colour change from blue to brick–red precipitate, which may vary between operators and lighting conditions.
- The method has a relatively high detection limit and is not suitable for estimation of trace amounts of sugars or for certification of lactose–free or sugar–free products.
- It involves handling of boiling alkaline solutions and hazardous chemicals such as sodium hydroxide and copper sulphate, which pose safety hazards.
- The procedure is labor–intensive and time consuming, allowing only one analysis at a time.
- Being a manual volumetric titration technique, it requires skilled operation and is prone to human errors such as parallax error during burette reading.
Precautions of Lane–Eynon Method
- Fehling’s solutions must be accurately standardized against a pure standard sugar solution, since the copper reduction reaction is non–stoichiometric and depends on reaction conditions. Duplicate standard titrations should agree within 0.1 mL.
- A moderate and continuous boiling must be maintained throughout the titration without removing the flask from the heat source. The continuous evolution of steam prevents atmospheric oxygen from re–oxidizing cuprous oxide into cupric ions.
- The total boiling time must be strictly controlled and should not exceed 3 minutes to ensure accuracy of the results.
- Anti–bumping agents such as glass beads, pumice stones or boiling chips must be added to ensure smooth and uniform boiling and to prevent superheating.
- A two–stage titration should always be performed. A preliminary titration is carried out to determine the approximate titre value, followed by a final accurate titration with nearly the entire required volume of sample added before heating.
- Methylene blue indicator should be added without removing the flask from the flame. The end point must be carefully observed at the first complete disappearance of the blue colour.
- Artificial or indirect lighting should be used to clearly distinguish the colour change from blue to brick–red precipitate.
- Proper laboratory safety measures must be followed. Protective equipment such as gloves, lab coat and safety goggles should be worn, as sodium hydroxide is corrosive and copper sulphate is an irritant.
- When using Carrez clarification reagents, Carrez I (potassium hexacyanoferrate) must not be mixed with strong acids, as it may release toxic hydrogen cyanide gas.
References
- Comprehensive analytical evaluation of lactose determination in milk and dairy matrices via the Lane-Eynon volumetric method. (n.d.).
- Determination of glucose by titration with Fehling’s reagent. (n.d.).
- eGyanKosh. (n.d.). Experiment 4 determination of reducing sugars, total reducing sugars, sucrose and starch.
- Estimating lactose in milk: Lane-Eynon method. (n.d.).
- Guo, X., Guo, X., Zhang, M., Ma, J., & Kang, W. (2016). Comparative detection of lactose and sucrose in milk powder by Lane-Eynon’s method and high performance liquid chromatography with refractive index detector. Food Science, 37(12). https://doi.org/10.7506/spkx1002-6630-201612024
- International Starch Institute. (1999). ISI 27-1e: Determination of reducing power and DE by Lane and Eynon’s method.
- Japan Customs. (2001). 114 Quantitative analysis of reducing sugars in sugar preparations consisting of sugar and dextrin.
- Ministry of Health of the People’s Republic of China. (2010). National food safety standard GB 5413.5-2010: Determination of lactose and sucrose in foods for infants and young children, milk and milk products.
- South African Sugar Technologists’ Association (SASTA). (2005). Method 5.4 – Syrup: reducing sugars and sucrose by the Lane and Eynon method.
- Tiwari, M., Mhatre, S., Vyas, T., Bapna, A., & Raghavan, G. (2023). A validated HPLC-RID method for quantification and optimization of total sugars: Fructose, glucose, sucrose, and lactose in eggless mayonnaise. Separations, 10(3), 199. https://doi.org/10.3390/separations10030199