What is the ornithine decarboxylase test?
Ornithine decarboxylase test is a biochemical test used for identification and differentiation of Gram-negative bacteria mainly Enterobacteriaceae family. It is used for differentiating closely related organisms like Klebsiella and Enterobacter species and also for identifying Proteus.
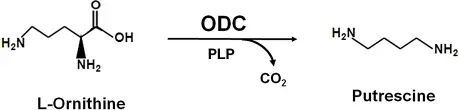
It is the test in which microorganism ability is measured to produce enzyme ornithine decarboxylase. This enzyme is involved in breakdown of amino acid L-ornithine into carbon dioxide (CO2) and an alkaline diamine called putrescine.
In this test bacteria is inoculated into a specific broth medium containing ornithine small amount of glucose and pH indicator like bromcresol purple. Initially glucose is fermented by bacteria and acids is produced which lowers the pH and the medium is turned yellow. This process also induces the expression of decarboxylase enzyme.
If organism possess ornithine decarboxylase then ornithine is broken down and putrescine is formed. This basic by-product neutralizes the acid and pH is raised and the medium is shifted back to purple or violet colour which indicates positive result.
If organism do not produce the enzyme then ornithine is not utilized and medium remains acidic and bright yellow which indicates negative result.
Objectives of Decarboxylase Test
- To test the ability of organism to produce a specific decarboxylase enzyme.
- To differentiate members of Enterobacteriaceae family from other gram-negative rods having closely related physiological characteristics.
- To help in identification and differentiation of specific bacterial species like distinguishing Salmonella from Shigella or identifying Enterococcus to species level.
- To analyze and determine the decarboxylation potential of a given microbe.
- To serve as educational and laboratory tool for understanding biochemical basis of decarboxylation reaction.
Principle of Ornithine decarboxylase test
Principle of ornithine decarboxylase test is based on ability of bacteria to produce specific decarboxylase enzyme which removes carboxyl group from amino acid and forms alkaline amine. In this test L-ornithine is used as substrate and the alkaline amine formed is putrescine.
The test medium is a liquid broth which contains L-ornithine glucose pyridoxal (enzyme co-factor) and pH indicator like bromcresol purple and cresol red. After inoculation the medium is sealed with mineral oil layer to create anaerobic condition which promotes fermentation.
Initially glucose is fermented by bacteria and acids is produced. This lowers the pH of medium and indicator colour is changed from purple to yellow. This acidic condition acts as trigger and decarboxylase enzyme production is stimulated in the organism.
If organism possess ornithine decarboxylase then L-ornithine is decarboxylated and carbon dioxide (CO2) and putrescine is formed. The putrescine is alkaline and it neutralizes the acid gradually and pH is raised. Due to this indicator colour is shifted from yellow to purple or violet which indicates positive result.
If organism do not possess the decarboxylase enzyme then putrescine is not formed and medium remains acidic and permanently yellow which indicates negative result.
Requirement for Decarboxylase Test
- Bacterial culture– A pure bacterial culture of 18 to 24 hours old is required for inoculum.
- Decarboxylase broth base– A liquid basal medium (Moeller’s formula) is required which contains nitrogenous nutrients (beef extract and peptones) fermentable carbohydrate (glucose/dextrose) pyridoxal (enzyme co-factor) and pH indicators (bromcresol purple and cresol red).
- Target amino acid– Specific amino acid is added separately like L-arginine L-lysine or L-ornithine for testing specific enzyme activity.
- Control tube– A separate tube containing only basal medium without amino acid is required as control for comparing colour reaction.
- Sterile anaerobic seal– Sterile mineral oil or liquid paraffin or Vaspar or petroleum jelly is required for overlaying the inoculated medium. This creates anaerobic condition and fermentation is promoted and exposure to air is prevented which can give false alkaline results.
- Inoculation supplies– Sterile inoculating loop needle or stick is required for aseptically transferring the bacteria into test tubes.
- Incubator– Incubator is required at 33°C to 37°C for incubation of test and control tubes. Incubation time is usually 18 to 96 hours and sometimes up to 10 days for slow reacting microorganism.
Preparation of the media for Decarboxylase Test
- Suspend the base– Recommended amount of dehydrated decarboxylase broth base (about 9.02 g to 10.52 g as per manufacturer) is added into 1000 mL of pure distilled water or deionized water.
- Add the amino acid– Specific target amino acid is added like L-ornithine or L-lysine or L-arginine usually at concentration of 0.5% to 1% (10 g/L). A portion of basal medium is kept without amino acid for negative control tube.
- Dissolve the medium– The mixture is heated with frequent agitation and it is brought to gentle boil so that ingredients is completely dissolved.
- Adjust the pH– If L-ornithine is added then pH is readjusted and final pH should be 6.0 ± 0.2 at 25°C.
- Dispense into tubes– The liquid medium is distributed in 3 to 5 mL quantity into screw capped test tubes.
- Sterilize– The capped tubes is autoclaved at 121°C (15 lbs pressure) for 10 to 15 minutes.
- Add an anaerobic seal– A 5 mm layer of liquid paraffin is added in tubes (in some procedures) before sterilization to prevent oxygen exposure and false alkalinization. Alternatively sterile mineral oil layer is added later immediately after inoculation of bacteria.
Procedure of Ornithine decarboxylase test
- Prepare the inoculum– A pure bacterial culture of 18 to 24 hours old is taken from agar plate or a broth suspension is prepared.
- Inoculate the test broth– Bacteria is transferred aseptically into ornithine decarboxylase broth by sterile loop (1 to 2 colonies) or by adding 1 to 4 drops of prepared bacterial suspension.
- Inoculate the control tube– Control tube containing only basal medium (without ornithine amino acid) is inoculated parallel for baseline colour comparison.
- Add an anaerobic seal– Sterile mineral oil is added over the medium about 1 mL or 4 mm thick layer so that broth is protected from air and fermentation is promoted.
- Seal the tubes– Caps is tightened properly on all test tubes.
- Incubate– Tubes is incubated at 33°C to 37°C for 18 to 96 hours and slow reacting microorganism may require up to 10 days incubation.
- Examine and read results– Tubes is checked daily for colour changes and final result is interpreted by comparing ornithine test tube colour with control tube.
Result of Ornithine decarboxylase test
Positive result– Medium is changed to turbid purple or violet or pale yellow-purple colour. It indicates organism produced ornithine decarboxylase enzyme and amino acid is broken down and alkaline by-products is formed and pH is raised.
Negative result (glucose fermenters)– Medium becomes bright clear yellow colour and it remains yellow. It indicates glucose is fermented and acids is produced but enzyme is absent so ornithine is not decarboxylated.
Negative result (non-glucose fermenters)– Medium shows little or no colour change and original dull purple colour is retained. In this case acid is not produced from glucose fermentation so enzyme is not induced.
Delayed positive result– Medium is turned yellow first then it is shifted to faded purple or grey colour after some time. It indicates weak or slow enzyme activity and incubation may be required up to 10 days for confirmation.
Invalid test (control tube)– Control tube without amino acid must be kept along with test tube. For glucose fermenting organism control tube must turn yellow. If control tube becomes purple then false alkalinization is present and test result is invalid.
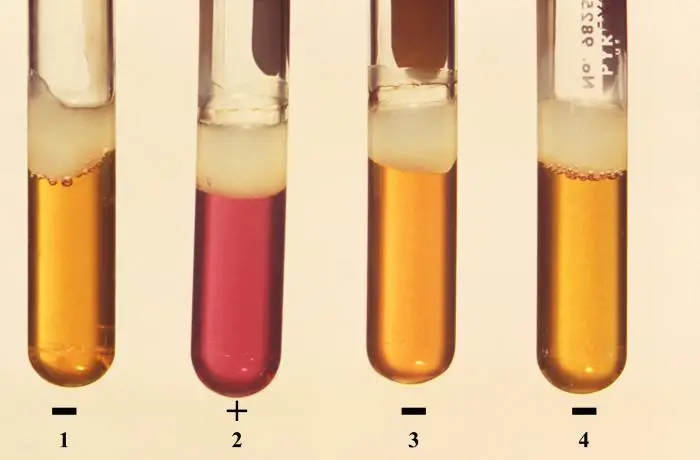




Ornithine decarboxylase test results of some organisms
Organisms with a Positive Ornithine Decarboxylase Result:
- Enterobacter aerogenes
- Enterobacter cloacae
- Escherichia coli (most strains)
- Proteus mirabilis
- Serratia marcescens
- Shigella sonnei
- Salmonella Paratyphi A
- Vibrio cholerae
- Edwardsiella tarda
- Edwardsiella hoshinae
- Morganella morganii (formerly Proteus morganii)
Organisms with a Negative Ornithine Decarboxylase Result:
- Klebsiella pneumoniae
- Klebsiella oxytoca
- Proteus vulgaris
- Proteus penneri
- Pseudomonas aeruginosa
- Salmonella Typhi
- Shigella flexneri
- Shigella dysenteriae
- Providencia species (such as P. rettgeri, P. stuartii, P. alcalifaciens, and P. rustigianii)
Organisms with Variable Ornithine Decarboxylase Results:
- Citrobacter freundii
Quality Control organisms of Ornithine decarboxylase test
- Positive quality control organisms (ornithine decarboxylation positive– purple colour)
- Enterobacter aerogenes (Klebsiella aerogenes)
- Enterobacter cloacae
- Proteus mirabilis
- Serratia marcescens
- Shigella sonnei
- Salmonella Paratyphi A
- Negative quality control organisms (ornithine decarboxylation negative– yellow colour)
- Proteus vulgaris
- Klebsiella pneumoniae
- Proteus hauseri
- Salmonella Typhi
- Shigella dysenteriae
- Shigella flexneri
Uses of Decarboxylase Test
- To determine the microorganism has the enzymatic ability to produce specific decarboxylase enzyme like arginine decarboxylase lysine decarboxylase or ornithine decarboxylase.
- To differentiate decarboxylase producing members of Enterobacteriaceae family from other Gram-negative rods.
- To distinguish between closely related enteric bacteria like separating members of Klebsiella-Enterobacter-Serratia (KES) group.
- To aid in identification of Enterococcus to species level by arginine decarboxylase test (example E. faecalis is positive and E. avium is negative).
- To help in differentiating Salmonella species (usually positive) from Shigella species (usually negative) by lysine decarboxylase test.
- To identify and differentiate species within genus Proteus.
- To help in speciation and identification of non-fermentative Gram-negative bacilli like Aeromonas Plesiomonas and Vibrio species.
- To evaluate and characterize organisms within Edwardsielleae family.
- To monitor microbial flora in food and beverage industry for evaluating food safety and screening safe probiotic strains which do not accumulate toxic biogenic amines.
Limitations of Decarboxylase Test
- Cannot measure enzyme quantity– This test only detects presence of enzyme when polyamine produced is sufficient to raise pH of medium. Actual intracellular amount of enzyme is not measured.
- Sensitivity to growth conditions– Change in concentration of glucose target amino acid or other amino acids can influence enzyme activity. This is seen mainly for lysine decarboxylase in coliforms.
- Reliance on glucose fermentation– Organism must be able to ferment glucose and acid is produced because acidic condition is required for inducing decarboxylase activity. Glucose nonfermenting organisms may give weak insufficient or unreliable results.
- Strict incubation timing– Result should not be read before 18 to 24 hours. Initial glucose fermentation makes medium yellow within 10 to 12 hours and if it is read early then false negative result can occur. Some weak organism requires prolonged incubation up to 10 days to show positive result.
- Requirement for anaerobic seal– Inoculated medium must be overlaid with mineral oil. If medium is exposed to air then oxidation and deamination of peptones at surface can cause alkalinization and a negative organism can show false positive result.
- Color separation and fading– During incubation two colour layers may be formed (yellow and purple) so tube is shaken gently before interpreting result. Medium may turn grey if indicator is reduced and then extra bromcresol purple is added for reading true reaction.
- Supplemental testing required– Positive decarboxylation result is only indicative and it must be combined with other biochemical tests for confirming bacterial genus or species.
FAQ
how is ornithine decarboxylase test used for identification of enterobacteriaceae?
An inoculum from a pure culture is transferred aseptically to a sterile tube of ornithine decarboxylase broth. The inoculated tube is incubated at 35-37 C for 24 hours and the preliminary results are determined. The microbe must first use the glucose present to cause the pH to drop. Increases the acidity of the solution, thereby changing colour of indicator from yellow to purple.
If the organism fails to produce ornithine-decarboxylase, the medium is acid (yellow).
What is the basis for the key test for ornithine or lysine decarboxylase?
production of decarboxylase enzyme by the sleeted organisms.
References
- Amino acid decarboxylase test – Procedure, uses and interpretation. (n.d.). MicrobiologyInfo.com.
- Central Drug House (P) Ltd. (n.d.). Technical information: Moeller decarboxylase broth base.
- Cooper, C. R., Jr. (2018). BIOL 3702 lab exercise – Decarboxylation test.
- HiMedia Laboratories. (2015). Decarboxylase test medium base (Falkow).
- HiMedia Laboratories. (2026). Decarboxylase broth base, Moeller (Moeller decarboxylase broth base).
- Lal, A., & Cheeptham, N. (2015). Decarboxylase broth protocol. American Society for Microbiology.
- O’Hara, C. M., Brenner, F. W., & Miller, J. M. (2000). Classification, identification, and clinical significance of Proteus, Providencia, and Morganella. Clinical Microbiology Reviews, 13(4), 534–546.
- Remel. (2014). Decarboxylase broth (Moeller) base control, arginine, lysine, and ornithine.
- Sapkota, A. (2022). Decarboxylase test: Principle, procedure, results, uses. Microbe Notes.
- Sigma-Aldrich. (2018). D2935 decarboxylase broth base, Moeller. Merck KGaA.
- TM Media. (2019). TM 2265 – L-ornithine decarboxylase broth (ISO / TS 22964: 2017).