What is Nitrate Reduction Test?
Nitrate reduction test is a biochemical test used to determine the ability of bacteria to reduce nitrate (NO₃⁻) into nitrite (NO₂⁻) or into other nitrogenous compounds such as ammonia or nitrogen gas. It is the process in which nitrate acts as a terminal electron acceptor during anaerobic respiration. This test is used in microbiology laboratory for identification and differentiation of different bacteria especially intestinal bacteria and some pathogenic species.
The principle of nitrate reduction test is based on the production of enzyme nitrate reductase. This enzyme is produced by certain bacteria and it catalyzes the reduction of nitrate into nitrite. The reaction is as follows–
NO₃⁻ → NO₂⁻
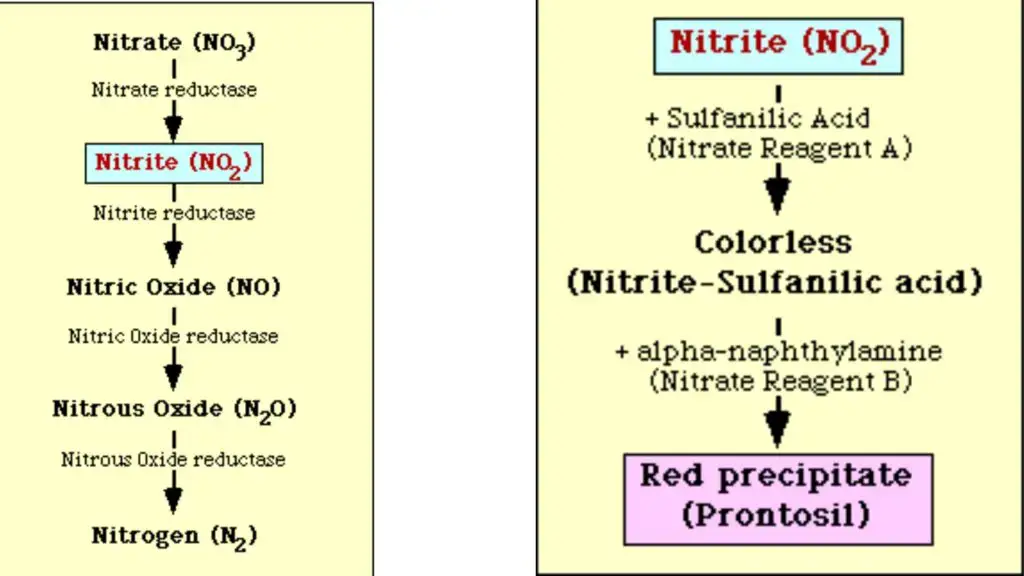
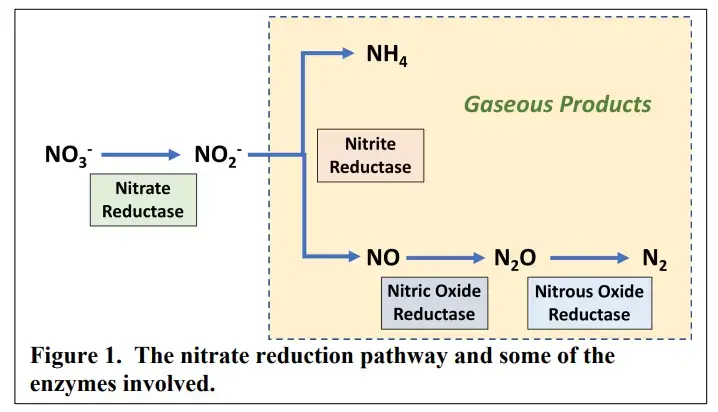
In some bacteria the reduction does not stop at nitrite stage. Nitrite is further reduced into ammonia (NH₃) or nitrogen gas (N₂). If nitrite is present in the medium it reacts with sulfanilic acid and α–naphthylamine forming a red or pink colour. If no colour is produced it may indicate that nitrate is not reduced or it is reduced beyond nitrite stage.
In this test bacteria is inoculated into nitrate broth containing potassium nitrate and incubated for 24–48 hours. A Durham tube may be placed inside the broth to detect gas formation. After incubation reagent A (sulfanilic acid) and reagent B (α–naphthylamine or N,N–dimethyl–1–naphthylamine) is added. If red colour appears the test is positive and nitrate is reduced to nitrite. If no colour develops zinc dust is added. If red colour appears after addition of zinc the test is negative because nitrate was not reduced by bacteria. If no colour appears even after zinc addition it indicates complete reduction of nitrate beyond nitrite stage.
Thus nitrate reduction test is important in identification of bacteria based on their ability to reduce nitrate and it is routinely performed in microbiology laboratory.
Purpose of Nitrate Reduction Test
The objectives of nitrate reduction test are–
- To detect the production of enzyme nitrate reductase by bacteria. It is done to determine whether the organism can reduce nitrate (NO₃⁻) to nitrite (NO₂⁻).
- To determine further reduction of nitrate beyond nitrite stage into ammonia (NH₃) or nitrogen gas (N₂). This shows the complete reduction pathway of nitrate.
- To identify and classify bacteria based on their biochemical property of nitrate reduction. It helps in differentiating facultative anaerobes and obligate anaerobes.
- To differentiate members of Enterobacteriaceae from other Gram–negative bacilli and to distinguish species of Neisseria, Moraxella, Kingella, Mycobacterium and Corynebacterium.
- To study the metabolic function of bacteria in nitrogen metabolism during anaerobic respiration.
Principle of Nitrate Reduction Test
The principle of nitrate reduction test is based on the ability of bacteria to produce the enzyme nitrate reductase. It is the process in which nitrate (NO₃⁻) is used as a terminal electron acceptor during anaerobic respiration. This enzyme catalyzes the reduction of nitrate into nitrite or further into other nitrogenous compounds.
The reaction is as follows–
NO₃⁻ → NO₂⁻
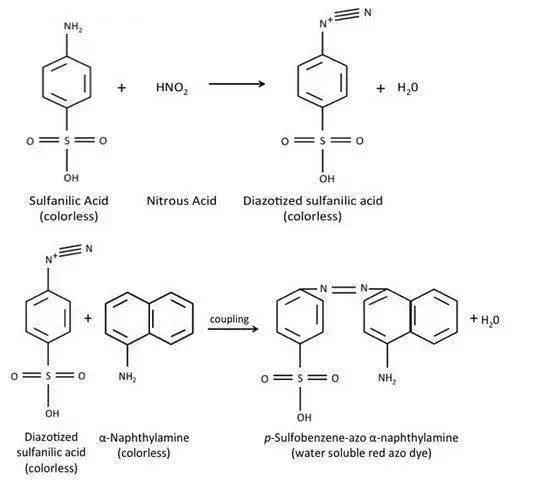
If the bacteria reduce nitrate into nitrite, the nitrite formed reacts with sulfanilic acid and α–naphthylamine (or N,N–dimethyl–α–naphthylamine) forming a red coloured azo dye. The appearance of red colour indicates positive result.
If no colour is produced after addition of reagents, it may indicate that nitrate is not reduced or it is reduced beyond nitrite stage into nitrogen gas (N₂) or ammonia (NH₃). In this condition zinc powder is added to the medium. Zinc reduces the remaining nitrate chemically into nitrite.
If red colour appears after addition of zinc, the test is negative because nitrate was not reduced by the bacteria. If no colour appears even after zinc addition, it indicates complete reduction of nitrate into other nitrogenous compounds. Thus the principle of this test depends on the enzymatic reduction of nitrate and detection of nitrite by colour reaction.
Reactions of Nitrate Reduction Test

Requirements
The requirements for nitrate reduction test are divided into the following–
Culture Media
- Nitrate Broth – It is a nutrient medium containing peptone, beef extract and 0.1% potassium nitrate. Potassium nitrate acts as the main substrate for the reaction.
- Durham Tube – It is a small inverted glass tube placed inside the nitrate broth to detect nitrogen gas produced during incubation.
Chemical Reagents
- Reagent A (Sulfanilic acid) – It is prepared in 5N acetic acid. It reacts with nitrite present in the medium.
- Reagent B (α–Naphthylamine or N,N–dimethyl–1–naphthylamine) – It is an aromatic amine solution prepared in 5N acetic acid. It reacts with sulfanilic acid and nitrite forming red colour.
- Reagent C (Zinc dust) – It is a powdered metal used to confirm the presence of unreduced nitrate. It chemically reduces nitrate into nitrite.
Equipment
- Incubator – It is used to maintain temperature usually 35–37°C for growth of bacteria.
- Sterile inoculating loop or swab – It is used for inoculation of organism into the broth.
- Test tubes and test tube rack – It is used for holding the culture medium.
- Pipette or dropper – It is used for adding reagents into the broth.
Test Organism and Control
- Pure bacterial culture – A fresh culture (18–24 hours old) is used for inoculation of nitrate broth.
- Quality control strains – Known positive and negative organisms are used to check proper functioning of media and reagents.
Composition and Preparation of Culture Media and Chemical Reagents for Nitrate Reduction Test
The composition and preparation of culture media and reagents are as follows–
Culture Media (Nitrate Broth)
Composition –
- Peptone – 5.0 g/L
- Beef extract – 3.0 g/L
- Potassium nitrate – 1.0 g/L
- Final pH – 7.0 ± 0.2 at 25°C
Preparation procedure –
- All dry ingredients are dissolved in 1 litre of distilled water. Heat is applied if necessary to dissolve completely.
- The broth is dispensed in 4–10 mL quantity into test tubes fitted with inverted Durham tube.
- The tubes are sterilized by autoclaving at 121°C (15 lbs pressure) for 15 minutes.
- During autoclaving the broth enters into the Durham tube and expels air.
- The tubes are allowed to cool to room temperature before inoculation.
Nitrate Reagent A (Sulfanilic acid)
Composition –
- Sulfanilic acid – 8.0 g
- 5N Acetic acid – 1 litre (0.8% solution)
Preparation procedure –
- Sulfanilic acid is dissolved in distilled water by applying heat.
- After cooling glacial acetic acid is added slowly to obtain 5N concentration.
- The reagent is stored in dark brown bottle at room temperature protected from light.
Nitrate Reagent B (α–Naphthylamine or N,N–dimethyl–1–naphthylamine)
Composition –
- N,N–dimethyl–1–naphthylamine – 6.0 g (0.6% solution) in 1 litre of 5N acetic acid
- (If α–naphthylamine is used – 5.0 g per litre of 5N acetic acid)
Preparation procedure –
- 5N acetic acid is prepared by mixing 287 mL glacial acetic acid with 713 mL distilled water.
- The naphthylamine compound is added slowly into the acid and mixed properly.
- The reagent is stored in dark brown bottle in refrigerator (2–8°C). Discoloured reagent should be discarded.
Nitrate Reagent C (Zinc dust)
Composition –
- Pure powdered zinc metal free from nitrate or nitrite contamination.
Preparation procedure –
- No chemical preparation is required. Zinc dust is used directly in dry powdered form.
- The container should be tightly closed and stored in dry place at room temperature. Contact with moisture should be avoided.
Procedure of Nitrate Reduction Test
The procedure of nitrate reduction test are carried out by the following methods–
Tube Method
- Autoclaved test tubes containing nitrate broth and inverted Durham tube are allowed to cool to room temperature.
- The broth is inoculated with isolated colony from fresh (18–24 hours old) culture or 2–3 drops of overnight broth culture is added.
- The tubes are incubated at 35 ±2°C for most bacteria. Some non–fermenting Gram–negative bacilli are incubated at 25–30°C for 24 hours up to 5 days.
- After 24 hours tubes are examined for visible growth and gas bubbles in Durham tube. If no growth is observed incubation is continued for further 24 hours.
- If gas is present in Durham tube and organism is non–glucose fermenter the test is considered positive for nitrate reduction and gas production.
- If no gas is present or organism is glucose fermenter, 0.5 mL of well mixed culture is transferred into clean test tube.
- Three drops of Nitrate Reagent A is added and mixed gently. Then three drops of Nitrate Reagent B is added and mixed.
- The tube is observed for development of red colour within 2 minutes.
- If no red colour appears a small pinch of zinc dust is added and observed for red colour within 10 minutes.
- If no gas and no red colour after reagents A and B, the original tube is re–incubated and test is repeated after 48 hours and on 5th day.
Disk Method (For anaerobic organisms)
- A nitrate disk is placed on the area of heavy growth from fresh culture.
- The culture is incubated anaerobically for 24–48 hours.
- The nitrate disk is removed and placed on clean glass slide or petri dish.
- One drop of Nitrate Reagent A and one drop of Nitrate Reagent B is added directly on the disk.
- The disk is observed for red colour within 2 minutes.
- If no red colour develops a small amount of zinc dust is added and observed for red colour within 5 minutes.
Rapid Method
- 0.5 mL of nitrate broth is taken in clean test tube and sterilized by autoclaving at 121°C (15 lbs pressure) for 15 minutes. It is allowed to cool.
- The broth is heavily inoculated with fresh culture.
- The tube is incubated at 35°C for 2 hours.
- Two drops of Nitrate Reagent A and two drops of Nitrate Reagent B is added and mixed well.
- The tube is observed for development of red colour within 2 minutes.
- If no red colour appears a small pinch of zinc dust is added and observed for red colour within 5 minutes.
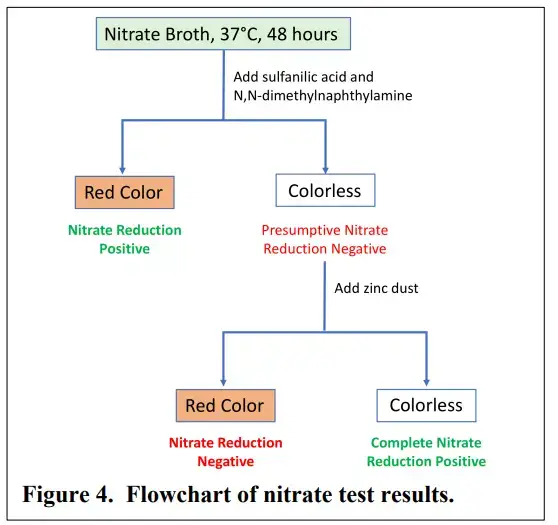
Result and Interpretation of Nitrate Reduction Test
The results of nitrate reduction test are interpreted as follows–
Positive result (Reduction of nitrate to nitrite) – Red or pink colour develops within 1–2 minutes after addition of Reagent A (Sulfanilic acid) and Reagent B (α–naphthylamine). It indicates that nitrate is reduced to nitrite by the enzyme nitrate reductase produced by the bacteria.
Positive result (Complete reduction of nitrate) – No colour appears after addition of Reagent A and Reagent B. After addition of zinc dust also no colour is produced. It indicates that nitrate is reduced beyond nitrite stage into ammonia (NH₃) or nitrogen gas (N₂).
Positive result (Gas formation) – Gas bubbles are seen in the Durham tube. It indicates denitrification and nitrate is reduced to nitrogen gas. In fermentative organisms gas may be produced due to fermentation therefore chemical reagents are required for confirmation.
Negative result (No nitrate reduction) – No colour appears after addition of Reagent A and Reagent B but red colour develops after addition of zinc dust. It proves that nitrate was not reduced by the bacteria and it was reduced chemically by zinc.


List of Bacteria Showing Positive and Negative Result in Nitrate Reduction Test
The bacteria showing positive and negative result in nitrate reduction test are–
| Bacteria Showing Positive Result (Nitrate Reducers) | Result |
|---|---|
| Escherichia coli | Nitrate is reduced to nitrite |
| Enterobacter aerogenes | Positive nitrate reduction is shown |
| Salmonella typhimurium | Nitrate is reduced |
| Pseudomonas aeruginosa | Nitrate is reduced and gas may be formed |
| Staphylococcus aureus | Positive reaction is observed |
| Mycobacterium tuberculosis | Nitrate is reduced to nitrite |
| Bacillus cereus | Nitrate is reduced |
| Citrobacter species | Positive nitrate reduction |
| Klebsiella species | Nitrate is reduced |
| Proteus species | Positive reaction is produced |
| Kingella denitrificans | Strong nitrate reducing organism |
| Moraxella catarrhalis | Nitrate is reduced |
| Neisseria mucosa | Positive nitrate reduction |
| Rothia species (R. mucilaginosa, R. dentocariosa, R. aeria) | Nitrate is reduced |
| Actinomyces species (A. viscosus, A. oris) | Positive reaction is shown |
| Bacteria Showing Negative Result (Non–Nitrate Reducers) | Result |
|---|---|
| Acinetobacter baumannii | Nitrate is not reduced |
| Acinetobacter calcoaceticus | Negative reaction is shown |
| Neisseria gonorrhoeae | Does not reduce nitrate |
| Neisseria lactamica | Negative nitrate reduction |
| Streptococcus species | Nitrate is not reduced |
| Mycobacterium bovis | Negative test is observed |
| Mycobacterium africanum | Nitrate is not reduced |
| Alcaligenes faecalis | Does not reduce nitrate |
Precautions of Nitrate Reduction Test
The precautions to be followed in nitrate reduction test are–
- Handle Reagents A and Reagent B carefully. These reagents are prepared in acetic acid and may cause irritation to skin and eyes. Protective measures should be taken while handling.
- α–naphthylamine should be handled with care as it is carcinogenic. N,N–dimethyl–1–naphthylamine is preferred as it is safer and gives stable colour.
- Zinc dust should be kept dry and tightly closed. Only a small pinch of zinc should be added. Excess zinc may reduce nitrate beyond nitrite stage and may give false negative result.
- Fresh pure culture (18–24 hours old) should be used for inoculation. The inoculum should be heavy. Light inoculum may not create proper anaerobic condition and result may be affected.
- Reagents A and B should be stored in dark coloured bottle and kept in refrigerator (2–8°C). Discoloured reagents should not be used.
- Durham tube should be checked before inoculation. It must be completely filled with broth and free from air bubbles. Pre–existing bubbles may give false positive gas result.
- Result should be read immediately within 1–2 minutes after addition of reagents. Delay in observation may cause fading of red colour.
- An uninoculated control tube should be kept along with test. If control shows red colour it indicates contamination of medium or reagents.
Uses of Nitrate Reduction Test
The uses of nitrate reduction test are–
- For general phenotypic identification of bacteria. It is used to study the biochemical characteristic of organism based on nitrate reduction property.
- For identification of Enterobacteriaceae. Members of Enterobacteriaceae characteristically reduce nitrate to nitrite and this test is used as a primary screening test to differentiate them from other Gram–negative bacilli.
- For differentiation of Neisseria and related genera. It helps in distinguishing nitrate negative Neisseria gonorrhoeae from nitrate positive organisms such as Kingella denitrificans and Moraxella catarrhalis.
- For differentiation of Mycobacterium species. Mycobacterium tuberculosis shows strong nitrate reductase activity whereas Mycobacterium bovis and Mycobacterium africanum are nitrate negative.
- For identification of Corynebacterium species. It is used at species level identification within the genus based on nitrate reduction reaction.
Limitations of Nitrate Reduction Test
The limitations of nitrate reduction test are–
- It is only an aid for identification of bacteria. Additional biochemical tests are required for complete identification of species.
- Red colour formed after addition of reagents may fade rapidly. Result must be observed immediately otherwise misinterpretation may occur.
- Excess zinc dust may lead to false result. It may reduce nitrate completely beyond nitrite stage or produce excess hydrogen gas which may interfere with interpretation.
- If no colour appears after addition of Reagent A and Reagent B, zinc dust must be added for confirmation. Without this step false negative result may be reported.
- The test depends on proper growth of bacteria and enzyme activity. Poor growth or light inoculum may not create anaerobic condition in the tube and false negative result may be obtained.
- Contamination of media or reagents with trace nitrites may give false positive reaction. Uninoculated control tube should be used to avoid this error.
- If no colour appears even after zinc addition it only indicates reduction beyond nitrite stage. Exact end product such as ammonia or nitrogen gas cannot be confirmed without further test.
- It is a culture based method and incubation period may be required from 24 hours up to 5 days before reporting negative result.
- Gas formation in Durham tube may be due to glucose fermentation in some organisms. It may be confused with denitrification if proper knowledge of organism is not known.
- Standard nitrate broth and reagents are not recommended for determination of nitrate utilization in some Mycobacterium species.
Quality Control Organisms of Nitrate Reduction Test
The quality control organisms used in nitrate reduction test are–
Positive control (Reduction of nitrate to nitrite) –
Escherichia coli (e.g., ATCC 25922) – Nitrate positive, gas negative.
Enterobacter aerogenes (e.g., ATCC 13048) – Nitrate is reduced to nitrite.
Salmonella typhimurium (e.g., ATCC 14028) – Nitrate positive reaction is shown.
Kingella denitrificans (e.g., CDC 10,236) – It is used as positive control especially in differentiation of Neisseria and related species.
Positive control (Complete reduction to nitrogen gas) –
Pseudomonas aeruginosa (e.g., ATCC 27853) – Nitrate positive, gas positive. It reduces nitrate completely to nitrogen gas (N₂).
Negative control (Do not reduce nitrate) –
Acinetobacter baumannii (e.g., ATCC 19606) – Nitrate is not reduced.
Acinetobacter calcoaceticus (e.g., ATCC 19606) – Negative reaction is shown.
Neisseria gonorrhoeae (e.g., ATCC 43069) – It is used as negative control in differentiation of Neisseria species.
Other quality control organisms –
Alcaligenes faecalis – Nitrate is not reduced but nitrite may be reduced.
Neisseria lactamica – It does not reduce nitrate but reduces nitrite.
References
- Buxton, R. (2011, November 1). Nitrate and nitrite reduction test protocols. American Society for Microbiology.
- Cappuccino, J. G., & Sherman, N. (2014). Experiment 29: Nitrate reduction test. In Microbiology: A laboratory manual (10th ed., pp. 133–136). Pearson Education, Inc.
- Centers for Disease Control and Prevention. (1971). Details – Public health image library (PHIL) (ID: 15520) [Photograph].
- Centers for Disease Control and Prevention. (2008, October 17). Nitrate reduction test – gonorrhea. Division of STD Prevention.
- Cooper, C. R., Jr. (2019). The nitrate reduction test. Youngstown State University.
- Dahal, P. (2024, February 18). Nitrate reduction test- principle, procedure, types, results, uses. Microbe Notes.
- Durand, S., & Guillier, M. (2021). Transcriptional and post-transcriptional control of the nitrate respiration in bacteria. Frontiers in Molecular Biosciences, 8, 667758. https://doi.org/10.3389/fmolb.2021.667758
- GMP Plastics. (2025, March 24). Nitrate reduction test: Principle, procedure, and interpretation.
- Hardy Diagnostics. (2020). Nitrate broth with durham tube [Instructions for use].
- Hardy Diagnostics. (2020). Nitrate reagent [Instructions for use].
- Hartline, R. (2023, February 18). 1.26: Nitrate reduction. In Microbiology laboratory manual. Biology LibreTexts.
- HiMedia Laboratories Pvt. Ltd. (2015). Nitrate broth [Technical data].
- Liu, H., Huang, Y., Huang, M., Wang, M., Ming, Y., Chen, W., Chen, Y., Tang, Z., & Jia, B. (2023). From nitrate to NO: Potential effects of nitrate-reducing bacteria on systemic health and disease. European Journal of Medical Research, 28, 425. https://doi.org/10.1186/s40001-023-01413-y
- Moreno-Vivián, C., Cabello, P., Martínez-Luque, M., Blasco, R., & Castillo, F. (1999). Prokaryotic nitrate reduction: Molecular properties and functional distinction among bacterial nitrate reductases. Journal of Bacteriology, 181(21), 6573–6584. https://doi.org/10.1128/jb.181.21.6573-6584.1999
- Remel. (2009, June 24). Nitrate agar [Instructions for use]. Thermo Fisher Scientific.
- Remel. (2009, June 24). Nitrate broth w/ and w/o durham tube [Instructions for use]. Thermo Fisher Scientific.
- Remel. (2009, June 24). Nitrate reagent B [Instructions for use]. Thermo Fisher Scientific.
- Rosier, B. T., Moya-Gonzalvez, E. M., Corell-Escuin, P., & Mira, A. (2020). Isolation and characterization of nitrate-reducing bacteria as potential probiotics for oral and systemic health. Frontiers in Microbiology, 11, 555465. https://doi.org/10.3389/fmicb.2020.555465
- Sigma-Aldrich. (2018). 38497 Nitrate reagent A (1-naphthylamine solution) [Product information]. Merck KGaA.
- Sigma-Aldrich. (2018). 73426 Nitrate reduction test [Product information]. Merck KGaA.
- Sparacino-Watkins, C., Stolz, J. F., & Basu, P. (2014). Nitrate and periplasmic nitrate reductases. Chemical Society Reviews, 43(2), 676–706. https://doi.org/10.1039/c3cs60249d
- The biochemical and diagnostic framework of the nitrate reduction test in microbiology. (n.d.).
- VUMIE. (2022, June 16). Nitrate broth with durham tube. Virtual Microbiology Lab Simulator Software.
- Werkman, C. H. (1930). Dimethyl-alpha-naphthylamine for the determination of bacterial reduction of nitrates. Proceedings of the Iowa Academy of Science, 37(1), 53–55.