Biuret test is a chemical test which is used to detect the presence of peptide bond in a given sample. It is mainly used for identification and estimation of proteins in the solution. It is based on the reaction of copper(II) ions with peptide bonds in alkaline condition. The reagent contains copper sulphate(CuSO4) and sodium or potassium hydroxide(NaOH/KOH) which provides alkaline medium and sodium potassium tartrate(Rochelle salt) is present to stabilize the copper ions. When the reagent is added to protein sample the Cu2+ ions binds with nitrogen of peptide bond and a chelate complex is formed and due to this reaction the colour is changed from light blue to violet or purple colour. The intensity of purple colour depends on number of peptide bonds present in sample and it is measured for total protein concentration by spectrophotometer at around 540 nm and the calculation is done by Beer-Lambert law. The test is named after the compound biuret because it also gives purple colour with this reagent though actual biuret is not present in reagent. Violet or purple colour indicates proteins is present in the sample and no colour change shows proteins is absent.
Objectives of Biuret Test
- It is used to detect the presence of peptide bonds in a given sample.
- It is used to confirm proteins or peptides is present in an unknown solution.
- It is used to estimate total protein concentration by measuring intensity of purple colour formed.
- It is used in clinical diagnosis to determine total protein level in body fluids like serum plasma and urine for conditions like kidney disease liver disorders and malnutrition.
- It is used to verify protein content in food and dairy products like casein in milk and protein in meat products.
- It is used to detect food adulteration by differentiating true proteins from nitrogen rich additives like melamine which do not give Biuret reaction due to absence of peptide bonds.
- It is used in biochemical and biotechnology laboratory as a simple and cost effective tool to track protein concentration during experiments.
Principle of Biuret Test
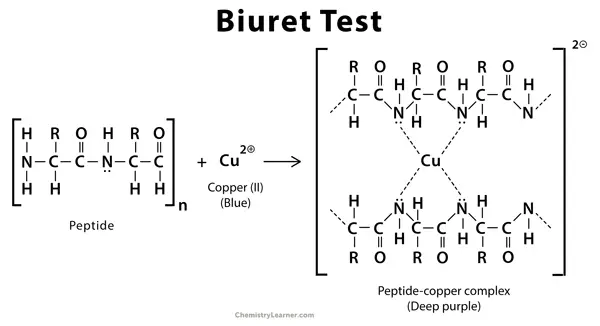
Principle of Biuret test is based on a colour reaction which is used to detect the presence of peptide bonds and to determine the amount of protein in a given sample. When the protein sample is kept in strongly alkaline medium the nitrogen present in peptide bond becomes deprotonated and due to this cupric ions(Cu2+) of Biuret reagent is allowed to bind with these nitrogen atoms. A copper-peptide coordination complex is formed and the colour of solution is changed from pale blue to violet or purple colour. The intensity of purple colour is directly proportional to number of peptide bonds present in the sample and it is measured by spectrophotometer by taking absorbance of colour and total protein concentration is calculated.
Requirements for Biuret Test
- Biuret reagent is required(Copper(II) sulphate + sodium hydroxide or potassium hydroxide + sodium potassium tartrate).
- Test sample is required which is to be analysed for protein.
- Positive control is required(known protein solution like albumin or egg white solution).
- Negative control is required(distilled water/deionized water or sugar solution).
- Dry test tubes and test tube rack is required.
- Pipette or micropipette with tips is required for accurate measurement.
- Dropper is required for adding reagent.
- Water bath or incubation chamber is required for maintaining reaction temperature.
- Timer is required for incubation time and tissue is required for cleaning.
- PPE is required(gloves lab coat goggles) with general lab supplies.
- Spectrophotometer or photometer is required for quantitative estimation(optional).
- Cuvettes(glass or quartz) is required for spectrophotometer.
- Analytical balance is required for weighing reagents or sample material(optional).
Reaction of Biuret Test
- Alkaline medium is created by adding NaOH or KOH to the sample.
- In alkaline condition the peptide bond nitrogen is deprotonated and lone pair electrons are available.
- Copper(II) sulphate is added and Cu2+ ions is released which is pale blue in colour.
- Cu2+ ions coordinate with nitrogen atoms of peptide bonds and a chelate complex is formed(tetradentate complex).
- Due to formation of copper-peptide complex the colour is changed from pale blue to violet or purple.
- The intensity of purple colour is proportional to number of peptide bonds involved and maximum absorbance is seen around 540–550 nm.
- At least two peptide bonds is required for this reaction(tripeptide or more).
Biuret Reagent Preparation
- Standard 1 litre preparation
- Copper(II) sulphate pentahydrate(1.5 g) is taken. Sodium potassium tartrate(Rochelle salt)(6.0 g) is taken. It is dissolved in 500 mL distilled water.
- The solution is stirred continuously and 10% NaOH solution(300 mL) is added slowly and alkaline medium is maintained.
- Distilled water is added and final volume is made up to 1000 mL(1 litre).
- The reagent is stored in plastic bottle(HDPE) or paraffin lined glass container because strong alkali can react with normal glass and black precipitate is formed.
- Small scale preparation
- Copper sulphate crystals(1 g) is dissolved in 100 mL distilled water.
- Sodium potassium tartrate(1.2 g) is added and it is mixed properly for stabilizing copper ions.
- 10% NaOH solution is prepared separately by dissolving NaOH pellets(10 g) in 90 mL distilled water.
- 10 mL of 10% NaOH solution is added to copper sulphate and tartrate mixture and Biuret reagent is prepared.
Procedure of Biuret Test
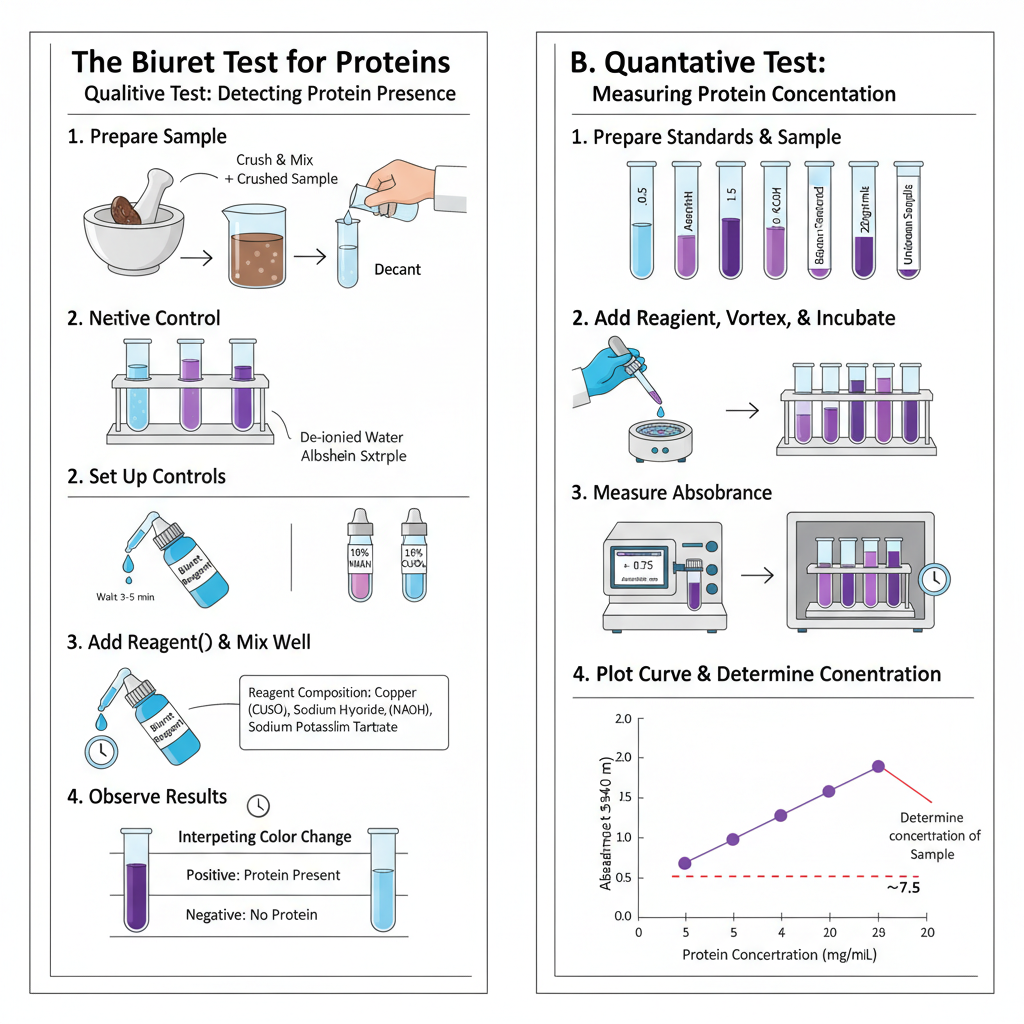
- Three clean test tubes is taken and it is labelled as test positive and negative.
- Test sample(1–2 mL) is added in the test tube.
- Known protein solution(albumin)(1–2 mL) is added in positive control tube.
- Distilled water(1–2 mL) is added in negative control tube.
- Equal volume of Biuret reagent(1–2 mL) is added to all the three tubes.
- If pre mixed reagent is not available then 1% NaOH/KOH is added in equal volume and then few drops of copper(II) sulphate solution is added.
- The contents are mixed by gentle shaking or vortexing and proper mixing is done.
- The test tubes are kept at room temperature for about 3–5 minutes.
- Colour change is observed and violet or purple colour indicates proteins or peptides is present.
- No colour change and solution remains blue indicates protein is absent.
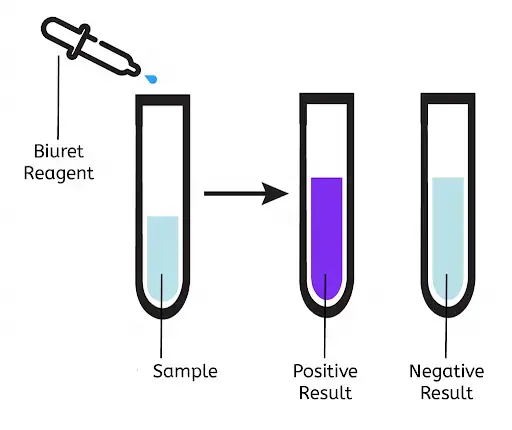
Result and Interpretation of Biuret Test

- Positive result
- Observation – The colour is changed from pale blue to violet or purple after adding Biuret reagent. Sometimes pink colour is also seen.
- Interpretation – It indicates proteins or peptides(peptide bonds) is present in the sample.
- Colour variation – Deep violet or purple colour shows higher concentration of proteins or longer polypeptide chain. Pinkish colour indicates shorter chain peptides.
- Negative result
- Observation – No colour change is observed and solution remains pale blue after incubation about 5 minutes.
- Interpretation – It indicates proteins or peptides is absent in the sample. It may also show only free amino acids is present because multiple peptide bonds is not present to give Biuret reaction.


Uses of Biuret Test
- It is used to detect the presence of proteins or peptides in unknown solution and laboratory extracts.
- It is used to measure protein concentration in a given sample by colour intensity.
- It is used in clinical diagnosis to determine total protein level in body fluids like serum plasma urine and cerebrospinal fluid(CSF).
- It is used to help in diagnosis and monitoring of kidney function liver disorders inflammation and drug induced nephrotoxicity.
- It is used in nutritional analysis to estimate protein content in food and dairy products like casein in milk and protein in meat tofu etc.
- It is used in food safety to detect adulteration by confirming genuine polypeptides and it helps to differentiate nitrogen additives like melamine which do not give Biuret reaction.
- It is used in biotechnology and biochemistry laboratory as a quick and cost effective assay for estimation of protein concentration.
Advantages of Biuret Test
- It is based on peptide bond and amino acid sequence does not affect the reaction and colour development is consistent.
- Free amino acids do not interfere because at least two peptide bonds is required and amino acids alone do not give Biuret reaction.
- It is specific for peptide bonds and other biomolecules like nucleic acids carbohydrates lipids and starch do not affect the test.
- It is compatible with many detergents used in sample preparation and it can be used in industrial and laboratory samples.
- It is suitable for samples having high protein concentration like blood serum and tissue extract and result is obtained clearly.
- It is simple rapid and cost effective test and it is easy to perform for detection and estimation of proteins.
Limitation of Biuret Test
- It is a low sensitive test and high concentration of protein is required for visible colour change and it is not suitable for highly dilute samples like normal urine or cerebrospinal fluid.
- At least two peptide bonds is required for Biuret reaction and free amino acids and dipeptides cannot be detected and tripeptide or more is required.
- Some chemicals can interfere and accuracy is affected. Ammonium and magnesium salts can give false high result. Tris buffer and chelating agents like EDTA can suppress colour development by removing copper.
- Free histidine can bind with copper and false positive result is obtained. High concentration of serine or threonine can alter absorbance reading.
- Turbid sample gives improper result and fats carbohydrates and bile pigments can affect reaction and colour and it is not suitable for highly turbid samples.
- It detects only soluble proteins and proteins should be completely soluble for proper reaction.
- It measures total peptide bond concentration only and it cannot identify the specific type of proteins present in a mixed sample.
Precautions of Biuret Test
- Sample and reagent ratio should be maintained properly(usually 1:1). Excess Biuret reagent should be avoided because it can keep solution blue and false negative result is obtained.
- Result should not be read immediately and the tubes should be kept for 3–5 minutes for colour development. For accurate quantitative test more time is required(20–30 minutes).
- Timing should be kept same for all tubes when multiple samples are tested and observation time should be consistent.
- Biuret reagent should be stored in plastic bottle(HDPE) or paraffin lined glass container because strong alkali can leach minerals from normal glass and black precipitate is formed.
- After colour is developed the tubes should be protected from direct sunlight and extreme temperature because colour stability can be affected.
- Turbid or cloudy samples should be clarified before test. Ethanol or petroleum ether can be used and centrifugation is done and clear supernatant is taken for test.
- Interfering substances should be avoided like ammonium salts Tris buffer and chelators like EDTA because it affects copper ions and test result is disturbed. If present proteins can be precipitated by trichloroacetic acid(TCA) and separated before testing.
References
- Aaron. (2023, June 5). Which protein assay is best for you? Azure Biosystems.
- Advanced analytical biochemistry: A comprehensive technical treatise on the biuret method for protein quantification. (n.d.).
- Al Bakain, R., Al-Degs, Y., El-Sheikh, A., & Al-Reyahi, A. Y. (2015). A quick detection of melamine adulteration in milk and dairy products using first‐order multivariate calibration. Journal of Food Processing and Preservation, 39(6), 2718-2727.
- Beliber, A. (n.d.). Biuret test for protein presence. Scribd.
- Biology Online. (2023, March 3). Biuret test – Definition and examples. Biology Online Dictionary.
- Blaber, M. (2021, March 5). 5.3: Quantification of protein concentration. Biology LibreTexts.
- Brilliant Biology Student. (n.d.). Biuret test for protein.
- Busher, J. T. (1990). Serum albumin and globulin. In H. K. Walker, W. D. Hall, & J. W. Hurst (Eds.), Clinical methods: The history, physical, and laboratory examinations (3rd ed.). Butterworths.
- BYJU’S. (n.d.). Biuret test principle.
- Caprette, D. R. (2015, June 12). Protein determination by the biuret method. Rice University.
- Comunale, J., & Foist, L. (n.d.). Biuret test | Overview, process & results. Study.com.
- Comparative study of the most commonly used methods for total protein determination in milk of different species and their ultrafiltration products. (n.d.). PMC.
- Creative BioMart. (n.d.). Principle and protocol of biuret method.
- Creative Proteomics. (n.d.). Protocol for biuret protein assay.
- Dahal, P. (2024, May 26). Biuret test for protein: Principle, procedure, results, uses. Microbe Notes.
- Dawood, H. (n.d.). Color reactions of proteins.
- eClinpath. (n.d.). Total protein. Cornell University College of Veterinary Medicine.
- Gregory, R. (2025, February 12). Differences in protein assays and their results. ABS Bio.
- Mettler-Toledo. (n.d.). Biuret protein assay (M9107 V1.0).
- Mihalcea, E., & Drochioiu, G. (2025). Improved protein assay based on the biuret reaction: Interference of proteinogenic amino acids. Revue Roumaine de Chimie, 70(7-8), 513-522.
- MilliporeSigma. (n.d.). Tools for protein quantitation.
- Novonesis. (n.d.). Melamine detection tests.
- PerkinElmer. (2013). Milk adulteration with melamine – Screening, testing and real-time detection.
- Sari, N. P. Y. P., Amril, A., & Muttaminnah. (2024). Total protein examination using the biuret method. International Journal of Scientific Advances, 5(6).
- Singla, R. (n.d.). Biuret test: Principle, procedure, and results explained. Vedantu.
- Slightam, C. (2023, April 19). Total protein test: Purpose, procedure & results (N. Butler, Rev.). Healthline.
- Sri, K. B., Hema, B., Anila, B., & Sumakanth, M. (2023). A simple spectrophotometric method for quantification of casein in milk and milk products. International Journal of Advanced Research, 11(1), 1741-1745.
- Taylor & Francis. (2019). Biuret. In Dictionary of Biomedical Science.
- Thermo Fisher Scientific. (2018). A theoretical and practical guide for spectrophotometric determination of protein concentrations at 280 nm [Technical note].
- Thermo Fisher Scientific. (n.d.). Chemistry of protein assays.
- Wikipedia. (2025, December 5). Biuret test.