Iodine test is a common chemical and biochemical test which is used to detect presence of starch and to differentiate it from mono and disaccharides. It is a qualitative test and it confirms starch but it do not measure the exact concentration.
In this test aqueous iodine solution with potassium iodide is used and it is commonly referred to as Lugol’s iodine. When this reagent is added to a sample containing starch it reacts mainly with amylose which is a linear glucose polymer and it forms a helical structure.
Potassium iodide helps in formation of polyiodide ions (triiodide, pentaiodide) and these ions enter inside the cavity of amylose helix. A starch-iodine complex is formed and it absorbs visible light producing a characteristic deep blue-black colour which indicates positive result for starch.
Different polysaccharides gives different colours based on length and branching of glucose chains. Amylopectin gives orange-yellow or reddish-brown colour. Glycogen gives reddish-brown colour and dextrins forms red colour. Cellulose and simple sugars like glucose do not form this complex and iodine solution remains yellowish-brown showing negative result.
This starch-iodine complex is heat sensitive so on heating the blue-black colour disappears due to uncoiling of helix but it reappears on cooling. The test is also not valid in highly acidic condition because starch is hydrolyzed in acid.
In plant biology iodine test is used to detect starch reserve in leaves and it is used to confirm occurrence of photosynthesis. It is also used in food safety to detect starch adulteration in products such as milk, butter, spices and honey. In industries it is used for monitoring starch removal in textile manufacturing and to track starch distribution in paper production.
A medical variation is also present which is referred to as Schiller’s test (Visual inspection with Lugol’s iodine) and it is used for screening of cervical cancer. Normal epithelial cells containing glycogen stains mahogany brown with iodine while abnormal glycogen depleted cells do not take stain properly.
Objectives of Iodine Test
- To detect starch in a given sample and confirm presence of polysaccharides mainly starch.
- To differentiate carbohydrates such as starch, glycogen and dextrins from simple sugars (monosaccharides and disaccharides).
- To determine fruit maturity and ideal harvest time in fruits such as apples by checking starch to sugar conversion using Starch Pattern Index (SPI).
- To detect food adulteration by identifying starch fillers added in milk, ghee, spices and honey.
- To differentiate healthy glycogen rich cervical cells from glycogen depleted precancerous/cancerous cells by Schiller’s test (Visual inspection with Lugol’s iodine).
- To map sweat gland function and help in diagnosis of autonomic nervous system disorders such as Horner’s syndrome by Minor test.
- To determine starch hydrolysis by bacteria and to check amylase enzyme production in organisms such as Bacillus subtilis.
- To confirm photosynthesis in plants by detecting starch reserves in leaves.
- To use as an indicator in iodometric redox titrations because it is highly sensitive indicator.
Principle of Iodine Test
Iodine test works on the principle of interaction between polyiodide ions and the helical structure of some polysaccharides to form a coloured complex. When Lugol’s iodine (iodine with potassium iodide) is added to a sample linear polyiodide ions are formed and these ions enter inside the coiled helix of sugar chain.
In this step a charge-transfer complex is formed in which polysaccharide acts as charge donor and polyiodide acts as acceptor. This changes the electron energy level and the complex absorbs visible light and shows a distinct colour.
The colour depends on the length and branching of glucose chain. Amylose which is linear and coiled gives intense blue-black colour. Amylopectin and glycogen which are highly branched gives orange-yellow and reddish-brown colour respectively.
Simple sugars (mono and disaccharides) and rigid linear polysaccharides like cellulose do not change colour because they lack the coiled structure to trap iodine molecules so iodine remains yellowish-brown.
The reaction is temperature dependent. On heating the helix coils unwind and colour disappears but on cooling helix reforms and iodine is trapped again so colour reappears.
Requirements for Iodine test
- Reagents– Lugol’s iodine solution (aqueous iodine with potassium iodide) and distilled water is used as negative control.
- Test sample– Sample to be tested such as liquid solution or solid food items (potato, bread, crackers) or plant leaf.
- Materials– Clean and dry test tubes, test tube stand and dropper/pipette for transferring liquids.
- Equipment– Vortex mixer for mixing and water bath for heating the sample to observe temperature effect on colour complex.
Reagent preparation for Iodine test (Lugol’s iodine)
- 5 g elemental iodine, 10 g potassium iodide (KI) and 100 mL distilled water is measured.
- Potassium iodide is dissolved first in about 20 to 30 mL distilled water. This step is important because iodine do not dissolve in water without iodide ions (triiodide complex is formed).
- Elemental iodine crystals are added to the potassium iodide solution.
- The mixture is stirred/shaked or gently heated with constant mixing until all iodine crystals are dissolved.
- Remaining distilled water is added to make the final volume upto 100 mL.
- The reagent is stored in tightly sealed dark amber glass bottle and kept in dark place because iodine is light sensitive and volatile and exposure to light or air degrades the solution.
Procedure of Iodine Test
A. For liquid samples
- 1 mL of liquid test sample is taken in a clean dry test tube.
- In another test tube 1 mL distilled water is taken as negative control.
- 2 to 3 drops of Lugol’s iodine solution is added to both tubes.
- The contents are mixed properly using vortex mixer.
- Colour change is observed. Blue-black or purple colour indicates positive result for starch while brownish-yellow colour indicates negative result.
- (Optional) The tubes can be heated in water bath until colour disappears and then cooled to see if blue-black colour reappears.
B. For solid food samples (potato, bread, crackers)
- Few drops of iodine reagent is added directly on the surface of the solid sample.
- Surface is observed for rapid colour change. Blue-black colour indicates starch is present.
C. For plant leaves (photosynthesis test)
- Green leaf is boiled in water for about 2 minutes to kill the tissue.
- The leaf is transferred into test tube containing 90% ethanol and heated in water bath for about 10 minutes to remove chlorophyll.
- The bleached leaf is taken out and rinsed with cold water to soften it.
- The leaf is placed on a clean flat surface and few drops iodine solution is added.
- Blue-black colour development indicates starch accumulation in leaf.
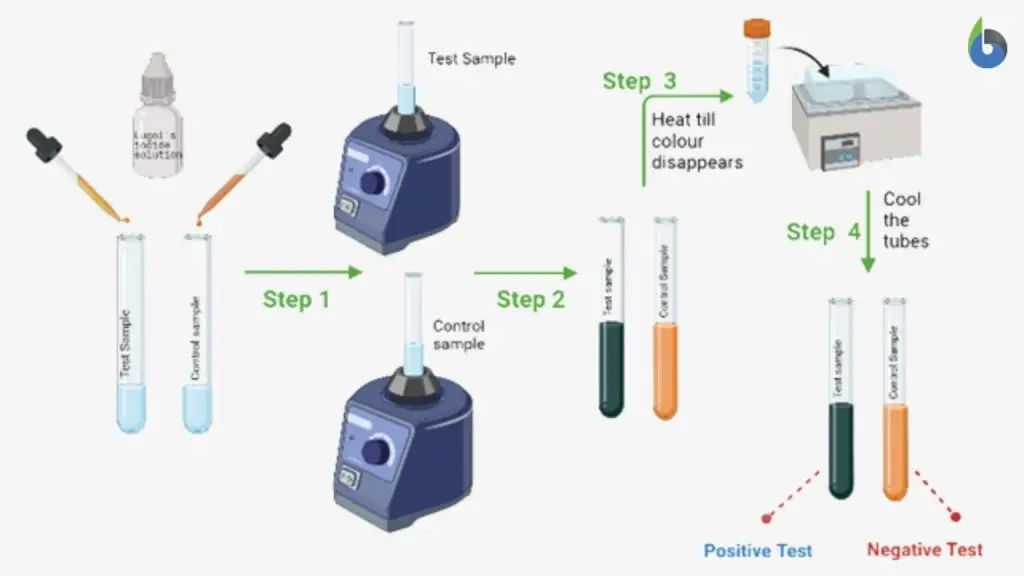
D. Confirmatory Test for Iodine Test
- The test tube showing blue-black or purple colour after adding iodine reagent is taken.
- The test tube is heated in boiling water bath or gently heated over burner.
- During heating the blue-black colour gradually disappears and solution becomes clear or pale yellow. This occurs because starch helix uncoils and trapped iodine is released.
- The test tube is removed from heat and allowed to cool to room temperature or it is kept in cold water.
- On cooling the blue-black colour reappears again because starch chains regain the helical structure and iodine is trapped again.
- This reversible disappearance and reappearance of colour confirms that the initial colour change was due to starch-iodine complex and it is taken as confirmatory result.
Result and Interpretation of Iodine Test
Positive result for starch
Blue-black or purple colour is produced in the sample after adding iodine reagent. This indicates starch is present. Amylose gives intense blue-black colour while amylopectin can give orange-yellow to red-violet shade.
Negative result
No colour change is seen and the sample remains brownish-yellow colour of iodine reagent. This indicates starch is absent and it is seen with simple sugars such as glucose and sucrose and also rigid polysaccharides like cellulose.
Positive result for glycogen
Reddish-brown colour is produced. This indicates glycogen is present because glycogen is highly branched polysaccharide.
Positive result for dextrins
Red, purple or sometimes black colour is produced depending on size of glucose polymer. This indicates dextrins are present.
Temperature variation (confirmatory interpretation)
When blue-black starch positive sample is heated colour disappears and solution becomes clear or pale yellow because starch helix is disrupted. On cooling blue-black colour reappears again which confirms starch-iodine complex.
Medical screening interpretation (Schiller’s test / VILI)
Healthy squamous epithelial cells containing glycogen stains dark mahogany brown or black with iodine. Abnormal precancerous or cancerous cells are glycogen depleted and they fail to take stain and remains pale yellowish or saffron colour.

Uses of Iodine test
- It is used to detect starch in a sample and to differentiate starch from simple sugars and other polysaccharides such as cellulose and glycogen.
- It is used to detect food adulteration by identifying starch fillers added in milk, butter, ghee, honey and spices.
- It is used in agriculture to track fruit maturity by Starch Pattern Index (SPI) and to determine optimal harvest time in fruits such as apples.
- It is used in medical screening as Schiller’s test / VILI to differentiate normal glycogen rich cervical cells from abnormal or precancerous cells.
- It is used in Minor test to map sweat gland function and to help diagnose autonomic nervous system disorders such as Horner’s syndrome.
- It is used in microbiology and parasitology as a mordant in Gram staining and as a stain to highlight internal structures of protozoan cysts and parasites in stool sample and also to identify bacteria capable of starch hydrolysis.
- It is used in plant biology to confirm photosynthesis by testing leaves for starch reserves.
- It is used in textile industry to check complete removal of starch during desizing so that fabric dyes evenly.
- It is used in paper and bioplastic production to monitor starch distribution on paper surface and to track breakdown of starch based bioplastics.
- It is used as a chemical indicator to detect end point in iodometric redox titrations.
Limitations of Iodine test
- It is a qualitative test only. It indicates presence or absence of starch but it cannot measure exact amount or concentration.
- The test is not valid in strongly acidic condition because starch undergoes acid hydrolysis and it breaks down.
- The test is also not valid in strongly alkaline condition because iodine reagent is altered to hypoiodite and iodate and it do not react with starch properly.
- The starch-iodine complex is temperature sensitive. On heating the blue-black colour disappears due to dissociation of complex.
- Water miscible organic solvents (ethanol, methanol, isopropyl alcohol) can interfere and it reduces the intensity of colour complex.
- Very dark coloured samples cannot be tested accurately because natural pigments mask the blue-black colour change. In plant leaves chlorophyll can also mask colour change if leaf is not decolorized first.
- It cannot detect simple sugars such as glucose and sucrose because the test is specific for coiled polysaccharide structure.
- In medical screening (Schiller’s test) results may not be fully specific because inflammation, tissue damage or ulceration can also fail to take iodine stain and it can give inaccurate interpretation.
Advantages of Iodine Test
- It is a simple and quick test and result is seen immediately by visual colour change without special equipment.
- It is cost effective and inexpensive to perform.
- It is comparatively safe because extreme pH condition is not required and toxic/heavy metal reagents like some other carbohydrate tests are not used.
- The reaction occurs rapidly at room temperature so additional heating is not required for routine test.
- It is highly specific for starch and it can differentiate starch from simple sugars and rigid polysaccharides like cellulose.
- In clinical use it is used as non-invasive screening test and in some cases it can reduce need of biopsy.
- It is compatible with tissue staining because iodine staining do not interfere much with later histologic staining and microscopic observation.
References
- Angel Starch. (n.d.). Starch in textile industry. https://angelstarch.com/starch-in-textiles/
- Auriga Research. (2021, April 8). What is milk adulteration and how can it be detected?. https://aurigaresearch.com/milk-adulteration-how-to-check-adulteration/
- BioGnost Ltd. (n.d.). Lugol’s solution (Schiller’s test). https://www.biognost.com/wp-content/uploads/2020/02/Lugols-solution-Schillers-test-IFU-V9-EN4.pdf
- Biology Online. (2022, June 16). Iodine test – Definition and examples – Biology online dictionary. https://www.biologyonline.com/dictionary/iodine-test
- Bio Research For Medical Diagnostics. (n.d.). Lugol’s iodine for gram stain. https://www.bioresearch.com.jo/ifu/Biological%20Stain/LUGOS%20IODINE%20FOR%20GRAM%20STAIN.pdf
- Bluecraft Agro. (2024, March 5). Elevating textile excellence: The impact of starch in the textile industry. https://bluecraftagro.com/blog/the-impact-of-starch-in-the-textile-industry/
- Brilliant Biology Student. (n.d.). Iodine test for starch. https://brilliantbiologystudent.weebly.com/iodine-test-for-starch.html
- Chia, Y.-T. (1958). Chemistry of +1 iodine in alkaline solution (UCRL-8311). Lawrence Berkeley National Laboratory, University of California. https://escholarship.org/content/qt71312227/qt71312227.pdf
- Comprehensive analytical report on the starch-iodine test: Biochemical principles, experimental protocols, clinical diagnostics, and industrial applications. (n.d.).
- Croptracker. (n.d.). Starch iodine testing. https://www.croptracker.com/resources/quality-control-resources/starch-iodine-testing.html
- Darby, A. (2021, August 12). Walter Schiller (1887–1960). Embryo Project Encyclopedia. Arizona State University. https://hdl.handle.net/10776/13305
- Farmer, S., & Kennepohl, D. (2024, March 23). 25.9: Polysaccharides and their synthesis. Chemistry LibreTexts. https://chem.libretexts.org/Bookshelves/Organic_Chemistry/Organic_Chemistry_III_(Morsch_et_al.)/25%3A_Carbohydrates/25.09%3A_Polysaccharides_and_Their_Synthesis
- Fleischer, H. (2019). The iodine test for reducing sugars – A safe, quick and easy alternative to copper(II) and silver(I) based reagents. World Journal of Chemical Education, 7(2), 45-52. https://doi.org/10.12691/wjce-7-2-3
- Food Safety and Standards Authority of India. (2012). Quick test for some adulterants in food. https://siesascs.edu.in/docs/notes/Manual%20for%20Adulteration%20in%20Food%20_50001.pdf
- Food Safety and Standards Authority of India. (n.d.). DART: Detect adulteration with rapid test. Eat Right India. https://eatrightindia.gov.in/dart/
- Food Safety and Standards Authority of India. (n.d.). Detect adulteration with rapid test – FSSAI. https://fssai.gov.in/upload/knowledge_hub/1878035b34b558a3b48DART%20Book.pdf
- Gyros Organic Farms. (2025, April 4). Easy turmeric adulteration test: Verify it’s purity at home. https://www.gyros.farm/blogs/know-what-you-eat/turmeric-adulteration-test
- HiMedia Laboratories. (n.d.). Technical data: S019 Lugol’s iodine. https://www.himedialabs.com/media/TD/S019.pdf
- International Agency for Research on Cancer. (n.d.). Histopathology and cytopathology of the uterine cervix – digital atlas – glossary. https://screening.iarc.fr/atlasglossdef.php?key=Schiller%20test&img
- International Agency for Research on Cancer. (n.d.). Testing and reporting the results of visual inspection with Lugol’s iodine (VILI). https://screening.iarc.fr/viavilichap3.php?lang=1
- Internet Pathology Laboratory. (n.d.). Lugol’s iodine. http://www-medlib.med.utah.edu/WebPath/webpath.html
- KClassScienceChannel. (n.d.). Use iodine to test a leaf for starch | Plant Physiology | Biology [Video]. YouTube. https://www.youtube.com/watch?v=0s_xZqvwm_s
- Kiferle, C., Martinelli, M., Salzano, A. M., Gonzali, S., Beltrami, S., Salvadori, P. A., Hora, K., Holwerda, H. T., Scaloni, A., & Perata, P. (2021). Evidences for a nutritional role of iodine in plants. Frontiers in Plant Science, 12, Article 616868. https://doi.org/10.3389/fpls.2021.616868
- Kumar, R. (2020, February 20). Methods for detection of common adulterants in food. Vikaspedia. https://en.vikaspedia.in/viewcontent/health/health-campaigns/beware-of-adulteration/methods-for-detection-of-common-adulterants-in-food
- Lohner, S. (n.d.). Test your foods for starch | STEM activity. Science Buddies. https://www.sciencebuddies.org/stem-activities/starch-food-test
- MecKey. (2025, May 20). Unlocking the industrial uses of starch; A game-changer across multiple sectors. https://www.meckey.com/news/Unlocking-the-Industrial-Uses-of-Starch-A-Game-Changer-Across-Multiple-Sectors-en.html
- MEL Science. (n.d.). Effect of iodine on starch. https://melscience.com/US-en/articles/impact-iodine-starch/
- Mondal, S. (2023, December). Iodine test for polysaccharides [Lecture notes]. ResearchGate. https://doi.org/10.13140/RG.2.2.27551.33442
- Nitya, K., Amberkar, V. S., & Nadar, B. G. (2020). Vital staining- pivotal role in the field of pathology. Annals of Cytology and Pathology, 5(1), 58-63. https://doi.org/10.17352/acp.000017
- Ophardt, C. (2022, July 4). Starch and iodine. Chemistry LibreTexts. https://chem.libretexts.org/Bookshelves/Biological_Chemistry/Supplemental_Modules_(Biological_Chemistry)/Carbohydrates/Case_Studies/Starch_and_Iodine
- Paśko, P., Prochownik, E., Kryczyk-Kozioł, J., Jlassi, M., Yahyaoui, D., Galanty, A., Ben Ahmed, H., & Dobrowolska-Iwanek, J. (2025). Iodine fortification of edible legume sprouts: A pilot biofortification study. Foods, 14(21), 3691. https://doi.org/10.3390/foods14213691
- Pesek, S., & Silaghi-Dumitrescu, R. (2024). The iodine/iodide/starch supramolecular complex. Molecules, 29(3), 641. https://doi.org/10.3390/molecules29030641
- Puri, A., Mohite, P., Ramole, A., Verma, S., Kamble, M., Ranch, K., & Singh, S. (2025). Starch science advancement: Isolation techniques, modification strategies, and multifaceted applications. Macromol, 5(3), 40. https://doi.org/10.3390/macromol5030040
- Sapkota, A. (2024, October 28). Iodine test: Principle, procedure, result, uses. Microbe Notes. https://microbenotes.com/iodine-test/
- Sigma-Aldrich. (n.d.). Lugol solution according to Lugol iodine/potassium iodide solution.
- Sulistyarti, H., Atikah, A., Fardiyah, Q., & Asdauna. (2015, July). The effect of pH on the absorbance of starch-iodine. ResearchGate.
- Taruc, A. E. P. (n.d.). Iodine test for polysaccharides analysis. Scribd. https://www.scribd.com/document/458762038/Iodine-Test-docx
- ThomasTKtungnung. (n.d.). Iodine solution preparation | Lugol’s iodine | Aqueous iodine | 0.1N iodine [Video]. YouTube. https://www.youtube.com/watch?v=wRLKYnQqaYY
- ThomasTKtungnung. (n.d.). Iodine test for starch practical experiment [Video]. YouTube. https://www.youtube.com/watch?v=72zKnccyH-w
- U.S. Food and Drug Administration. (2025, February 27). BAM R40: Lugol’s iodine solution. https://www.fda.gov/food/laboratory-methods-food/bam-r40-lugols-iodine-solution
- Vedantu. (n.d.). Test for starch: Iodine test procedure, colour change & uses. https://www.vedantu.com/biology/test-for-starch
- Wikipedia. (2023, July 21). Schiller’s test. https://en.wikipedia.org/wiki/Schiller%27s_test
- Wikipedia. (2026, January 29). Iodine–starch test. https://en.wikipedia.org/wiki/Iodine%E2%80%93starch_test