What is Indole test?
It is a biochemical test used in microbiology to determine the bacteria is producing enzyme tryptophanase. This enzyme help in breaking down amino acid tryptophan.
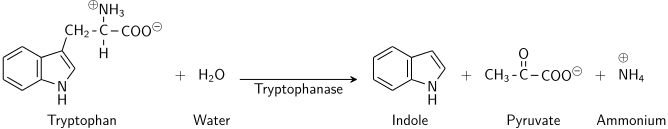
This process occurs when bacteria having tryptophanase utilize tryptophan and the end products is formed. These are indole pyruvic acid and ammonia.
In this test bacteria are cultured in a tryptophan rich medium. After incubation detection reagent is added. It is commonly Kovac’s reagent or Ehrlich’s reagent.
If indole is present it reacts with reagent and a cherry red or pinkish red colour layer is formed at top of the medium. This is referred to as positive result.
If indole is not present no colour change is observed and medium remains yellow. This is referred to as negative result.
It is a part of IMViC tests used for classification of coliform bacteria in Enterobacteriaceae family. It is useful in differentiating indole positive Escherichia coli from indole negative bacteria like Enterobacter and Klebsiella.
Rapid spot test is also used in which result is obtained in seconds from a bacterial colony. In this test blue or blue green colour indicates positive result.
Indole Test Definition
The indole test is a biochemical assay used to determine a bacterium’s ability to metabolize tryptophan into indole through the action of the enzyme tryptophanase, aiding in the differentiation and identification of certain bacterial species.
Purpose of Indol Test (Objectives of Indole Test)
- It is used to determine the bacterial species can produce enzyme tryptophanase.
- It is used to detect the breakdown of amino acid tryptophan and products is formed (indole pyruvic acid and ammonia).
- It is used for identification and differentiation of Enterobacteriaceae family (coliform bacteria) as a part of IMViC tests.
- It is used to differentiate indole positive Escherichia coli from indole negative bacteria like Enterobacter and Klebsiella.
- It is used to establish metabolic character of isolate and it helps in understanding ecological niche and pathogenic potential.
Principle of Indole Test
It is based on detection of bacterial ability to produce enzyme tryptophanase. This enzyme is involved in breakdown of amino acid tryptophan.
This process occurs by reductive deamination and tryptophan is acted by tryptophanase. Coenzyme pyridoxal phosphate is used in this reaction and amine group is removed from tryptophan.
In this step tryptophan is converted into indole pyruvic acid and ammonia.
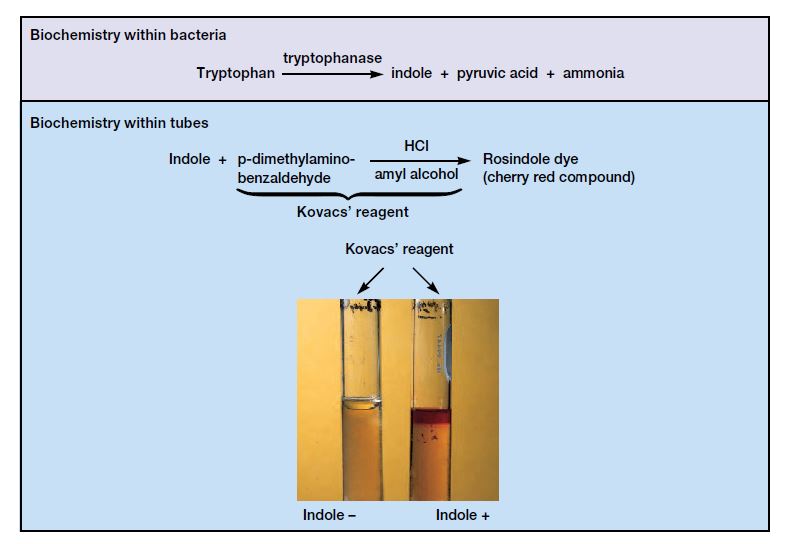
For detection of indole Kovac’s reagent or Ehrlich’s reagent is added to the bacterial culture. These reagent contain p-dimethylaminobenzaldehyde (DMAB) as active ingredient.
In acidic condition DMAB reacts with indole and cherry red or pinkish red compound is formed. This is referred to as rosindole dye.
In Kovac’s reagent amyl alcohol is present which is insoluble in water so the dye is raised and oily ring is formed at surface of liquid medium.
In rapid spot test p-dimethylaminocinnamaldehyde (DMACA) reagent is used and it reacts with indole to produce blue or blue green colour.
Reagents Used in Indole Test
Media
- Tryptone broth (peptone water) – It is the standard liquid medium and it is rich in tryptophan.
- Sulfide-indole-motility (SIM) agar – It is a multi test solid medium used for indole production motility and hydrogen sulfide production.
- Motility-indole-ornithine (MIO) agar – It is a multi test solid medium used for indole motility and ornithine decarboxylation.
- Casein broth – It is a liquid medium and it is used as alternative source of tryptophan.
- Heart infusion or anaerobic medium – It is used with tryptophan for testing anaerobic organisms.
Reagents
- Kovac’s reagent – It is used for aerobic organisms. It contain p-dimethylaminobenzaldehyde (DMAB) amyl or isoamyl alcohol and concentrated hydrochloric acid (HCl).
- Ehrlich’s reagent – It is used for anaerobes and weak indole producers. It contain p-dimethylaminobenzaldehyde (DMAB) ethyl alcohol (ethanol) and concentrated hydrochloric acid (HCl).
- Rapid spot test reagent – It is commonly 1% p-dimethylaminocinnamaldehyde (DMACA) in 10% HCl or 5% p-dimethylaminobenzaldehyde (DMAB) in 10% HCl.
- Extraction solvents – Xylene or ether is used as preliminary extraction step when Ehrlich’s reagent is used.
Supplies
- Inoculating tools – Sterile loop wire or needle or wooden applicator stick is used for picking and transfer of bacterial colony.
- Filter paper – It is used to hold reagent and bacterial smear in rapid spot test.
- Cotton swab – It is used for picking colonies and applying spot test reagent directly.
- Test tubes – It is used for holding broth or agar deep during incubation in tube method.
- Petri dish covers – It is used to hold filter paper during spot test.
Procedure of Indole Test
Conventional tube method
- Step 1- Medium is taken (tryptone broth or peptone water) and test tube is labelled.
- Step 2- Pure bacterial culture is picked by sterile inoculating loop or needle and it is inoculated into the tube containing tryptophan rich medium.
- Step 3- The inoculated tube is incubated aerobically at 35–37°C for 24–48 hours so that growth is obtained and tryptophan breakdown is occurred.
- Step 4- After incubation about 5 drops (or 0.5 ml) detection reagent is added into broth. Kovac’s reagent is used for aerobic bacteria and Ehrlich’s reagent is used for anaerobic bacteria.
- Step 5- Tube is shaken gently and it is kept for few minutes. In this step alcohol based reagent is raised and oily layer is formed at top of medium.
- Step 6- Surface layer is observed for colour change. Cherry red or pinkish red ring indicates positive result and yellow or cloudy ring indicates negative result.
Rapid spot test method
- Step 1- Filter paper is placed in petri dish and it is moistened with rapid spot indole reagent (1% DMACA or 5% DMAB).
- Step 2- Well isolated bacterial colony (18–24 hours old) is picked from non selective tryptophan rich agar plate by sterile loop or wooden applicator stick.
- Step 3- The colony is rubbed on reagent saturated area of filter paper.
- Step 4- Colour change is observed within 20–30 seconds. Blue or blue green colour indicates positive result when DMACA is used and pink to red colour indicates positive result when DMAB is used.
- Step 5- If negative result is present no colour change is seen and it remains colourless or slightly pink.
Alternative spot methods
- Cotton swab is used for picking colony and reagent is dropped directly onto swab.
- Reagent is dropped directly onto bacterial colony growing on agar plate.

Result Interpretation of Indole Test
Conventional tube test (Kovac’s reagent or Ehrlich’s reagent)
- Positive result- Cherry red or pinkish red ring is formed at top liquid layer. It indicates the bacteria is producing enzyme tryptophanase and tryptophan is converted into indole.
- Negative result- No colour change is seen at top layer and it remains yellow or slightly cloudy. It indicates the bacteria do not produce the enzyme.
- Variable result- Orange colour is appeared at surface. It indicates skatole is produced instead of indole and it is a different breakdown product of tryptophan.
Rapid spot test (filter paper or swab method)
- Positive result- Blue or blue green colour is developed within 20–30 seconds when cinnamaldehyde (DMACA) reagent is used. Pink to violet red colour is developed when benzaldehyde (DMAB) reagent is used. Providencia alcalifaciens gives red violet colour even with DMACA.
- Negative result- Test area remains colourless or slightly pink and it indicates indole is not produced.

List of Indole positive And Negative Organisms
| Indole-Positive Organisms | Indole-Negative Organisms |
|---|---|
| Escherichia coli | Klebsiella pneumoniae |
| Proteus vulgaris | Proteus mirabilis and Proteus penneri |
| Klebsiella oxytoca | Enterobacter species (e.g., E. cloacae) |
| Morganella morganii | Salmonella species |
| Edwardsiella tarda (and other Edwardsiella species) | Shigella species |
| Providencia species (e.g., P. rettgeri, P. stuartii) | Citrobacter freundii |
| Citrobacter koseri | Pseudomonas species (e.g., P. aeruginosa) |
| Aeromonas hydrophila and Aeromonas punctata | Serratia species |
| Bacillus alvei | Yersinia species |
| Flavobacterium species | Bacteroides fragilis |
| Haemophilus influenzae | Staphylococcus aureus |
| Pasteurella multocida and Pasteurella pneumotropica | Most Bacillus species |
| Plesiomonas shigelloides | Most Haemophilus species |
| Vibrio species (e.g., V. cholerae) | Actinobacillus species |
| Enterococcus faecalis | Aeromonas salmonicida |
| Fusobacterium species | Alcaligenes species |
| Lactobacillus reuteri | Bordetella species |
| Porphyromonas asaccharolytica | Most Lactobacillus species |
| — | Neisseria species |
| — | Pasteurella haemolytica and Pasteurella ureae |
| — | Klebsiella variicola |
| — | Raoultella terrigena and Raoultella ornithinolytica |
| — | Rhizobium species |
Uses of Indole Test
- It is used for identification and differentiation of Enterobacteriaceae family (coliform bacteria). It is useful in distinguishing indole positive Escherichia coli from indole negative Enterobacter and Klebsiella species.
- It is used to differentiate closely related species within same genus. Proteus vulgaris is positive and Proteus mirabilis is negative. Klebsiella oxytoca is positive and Klebsiella pneumoniae is negative. Citrobacter koseri is positive and Citrobacter freundii is negative.
- It is used for characterization of other pathogens and anaerobes. Vibrio species and Haemophilus influenzae are generally positive. Fusobacterium species is positive and Bacteroides fragilis is negative.
- It is used in food safety and environmental monitoring and it is a confirmatory assay for E. coli detection in standards like ISO and AOAC.
- It is used as rapid secondary screening to confirm E. coli in MUG (4-methylumbelliferyl-β-D-glucuronide) assay and it helps in ruling out false MUG positive caused by indole negative organisms like Salmonella and Shigella.
- It is used to determine indole production and metabolic signature is established which helps in understanding ecological niche signalling mechanism and pathogenic potential.
Quality control organisms for Indole Test
Positive control organisms (Indole producers)
- Escherichia coli ATCC 25922 or NCTC 10418.
- Porphyromonas asaccharolytica ATCC 25260.
- Bacteroides ovatus ATCC 8483.
- Propionibacterium acnes ATCC 29399.
Negative control organisms (Non indole producers)
- Klebsiella pneumoniae ATCC 13883.
- Pseudomonas aeruginosa ATCC 27853.
- Bacteroides fragilis ATCC 25285.
- Proteus mirabilis NCTC 10975 or ATCC 12453.
- Prevotella melaninogenica ATCC 25845.
- Clostridium sordellii ATCC 9714.
Limitations of Indole Test
- Media containing glucose or other acid producing carbohydrate should not be used. Acid production changes the pH and tryptophanase enzyme is inhibited so false negative result is obtained.
- Mueller Hinton agar cannot be used because acid hydrolysis in its manufacture destroys amino acid tryptophan.
- Media containing dyes or indicator (MacConkey agar or EMB agar) can interfere in observation and the colour change is obscured so interpretation is not accurate.
- Performing test on Indole Nitrate media or near a nitrate disc can give false negative result. Nitrite ion reacts with indole and reagent cannot form the red colour.
- Incubation for more than 48 hours can give false negative because indole is degraded by bacteria and it disappears.
- Some anaerobic bacteria (Clostridium species) produce indole but it is broken down rapidly so false negative reaction is seen.
- In spot test on agar plate indole can volatilize and it diffuses from indole positive colony to nearby indole negative colony so false positive result is obtained.
- Some bacteria produce skatole instead of indole and orange reaction is produced which can complicate interpretation.
- Rapid spot test is less sensitive than tube test so negative spot test may require confirmation by tube test.
- In spectrophotometric measurement high concentration of indole can change the complex from pink to yellowish orange and absorbance reading at 530 nm becomes inaccurate.
FAQ
What is the Indole Test?
The Indole Test is a biochemical test used to determine an organism’s ability to convert tryptophan into indole.
Why is the Indole Test important?
The test is crucial for differentiating members of the Enterobacteriaceae family and identifying certain bacterial species based on their metabolic capabilities.
How is a positive result for the Indole Test indicated?
A positive result is indicated by the formation of a pink-red coloration when the reagent is added.
What enzyme is responsible for the conversion of tryptophan to indole?
The enzyme tryptophanase is responsible for this conversion.
Can the Indole Test differentiate between E. coli and Klebsiella pneumoniae?
Yes, E. coli is typically indole positive, while Klebsiella pneumoniae is indole negative.
What reagents are commonly used in the Indole Test?
Kovac’s reagent is commonly used for aerobic organisms, while Ehrlich’s reagent is used for anaerobes and weak indole producers.
Is the spot indole test as sensitive as the conventional tube method?
No, the spot indole test is generally considered less sensitive than the conventional tube method.
Why is tryptophan essential for the Indole Test?
Tryptophan is the substrate that is metabolized to produce indole, making it essential for the test.
Can the Indole Test be used for anaerobic bacteria?
Yes, but it requires specific reagents like Ehrlich’s reagent and specific conditions to ensure accurate results.
Are there any limitations to the Indole Test?
Yes, the conventional method is time-consuming, and there are challenges in differentiating non-fermentative and anaerobic bacteria. Additionally, certain media and conditions can interfere with the results.
References
- Alves, M. S., Dias, R. C. S., de Castro, A. C. D., Riley, L. W., & Moreira, B. M. (2006). Identification of clinical isolates of indole-positive and indole-negative Klebsiella spp. Journal of Clinical Microbiology, 44(10), 3640–3646. https://doi.org/10.1128/JCM.00940-06
- American Society for Microbiology. (2009). Indole test protocol.
- American Society for Microbiology. (2026). Indole test. ASM.org.
- Andonovski, B. S., & Stojković, G. M. (2002). Spectrophotometric study of the reaction of indole with nitrite ions in hydrochloric acid. Bulletin of the Chemists and Technologists of Macedonia, 21(2), 177–185.
- Bio-Rad Laboratories. (2026). Food safety and quality reference guide.
- CDH Fine Chemical. (2023). Kovac’s indole reagent.
- Comprehensive diagnostic and biochemical profiling of the bacterial indole test in clinical and environmental microbiology. (n.d.).
- Çakır, İ., Doğan, H. B., Başpınar, E., Keven, F., & Halkman, A. K. (2002). The need for confirmation in coliform and E. coli enumeration in foods. Turkish Journal of Veterinary & Animal Sciences, 26(5), 1049–1053.
- Dahal, P. (2026). Spot indole test: Principle, procedure, results, uses. Microbe Notes.
- Dalynn Biologicals. (2014). Kovacs reagent.
- Darkoh, C., Chappell, C., Gonzales, C., & Okhuysen, P. (2015). A rapid and specific method for the detection of indole in complex biological samples. Applied and Environmental Microbiology, 81(23), 8093–8097. https://doi.org/10.1128/AEM.02787-15
- Dept. Medical Microbiology and Infectious Diseases. (n.d.). Indole test. Microbe Canvas.
- Faleev, N. G., Tsvetikova, M. A., Gogoleva, O. I., Kulikova, V. V., Revtovich, S. V., & Kochetkov, K. A. (2019). The catalytic mechanisms of the reactions between tryptophan indole-lyase and nonstandard substrates. Acta Naturae, 11(3), 82–88. https://doi.org/10.32607/20758251-2019-11-3-82-88
- Filo. (2025). Examples of indoles negative and indoles positive.
- Hardy Diagnostics. (2020). CRITERION indole nitrate medium.
- Hardy Diagnostics. (2020). Indole test reagents – Kovacs, DMACA, spot test.
- HiMedia Laboratories. (2016). Indole nitrate broth.
- HiMedia Laboratories. (2026). Technical data.
- International Organization for Standardization. (2001). Microbiology of food and animal feeding stuffs — Horizontal method for the detection of Escherichia coli O157 (ISO 16654:2001).
- Lamb, A. C., Federico-Perez, R. A., & Xue, Z.-L. (2015). Product in indole detection by Ehrlich’s reagent. Analytical Biochemistry, 484, 21–23. https://doi.org/10.1016/j.ab.2015.04.033
- Microbiology Class. (2026). Indole test.
- MicrobiologyInfo.com. (n.d.). Indole test- Principle, reagents, procedure, result interpretation and limitations.
- MilliporeSigma. (2026). Kovac’s reagent for indoles for microbiology 100-10-7.
- Palego, L., Betti, L., Rossi, A., & Giannaccini, G. (2016). Tryptophan biochemistry: Structural, nutritional, metabolic, and medical aspects in humans. Journal of Amino Acids, 2016, 8952520. https://doi.org/10.1155/2016/895