Cytomegalovirus (CMV) is a widespread virus whose manifestations in immunocompromised patients with congenital CMV disease range from asymptomatic to severe end-organ dysfunction. Human cytomegalovirus is a member of the herpesviruses, Herpesviridae, or human herpesvirus-5 (HHV-5) family of viruses. Infections with the human cytomegalovirus are frequently associated with the salivary ducts. An infection with CMV may be asymptomatic in healthy individuals, but it can be fatal in immunocompromised patients.
Human Cytomegalovirus
- Cytomegalovirus (CMV) is a widespread virus whose manifestations in immunocompromised patients with congenital CMV disease range from asymptomatic to severe end-organ dysfunction.
- Human cytomegalovirus is a member of the herpesviruses, Herpesviridae, or human herpesvirus-5 (HHV-5) family of viruses.
- Infections with the human cytomegalovirus are frequently associated with the salivary ducts. Infection with CMV may be asymptomatic in healthy individuals, but it can be fatal in immunocompromised patients.
- A congenital infection with cytomegalovirus can cause morbidity and even mortality. CMV often remains dormant after infection, but it can reactivate at any time. Eventually, it leads to mucoepidermoid carcinoma, and it may also induce prostate cancer.
- CMV infects between 60% and 70% of adults in industrialized nations and nearly 100% of adults in developing nations. CMV contains the greatest number of genes dedicated to eluding the host’s innate and adaptive immune responses.
- CMV is associated with antigenic T-cell surveillance and immune dysfunction for a lifetime. Congenital CMV is the primary infectious cause of deafness, intellectual disability, and learning disabilities.
Structure of Human Cytomegalovirus
Enveloped, spherical to pleomorphic, with a diameter of 150-200 nm and T=16 icosahedral symmetry. The capsid is composed of 162 capsomers and is enveloped by an amorphous tegument. Complexes of glycoproteins are embedded in the lipid envelope.
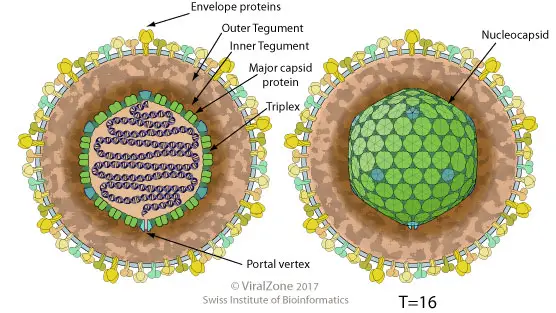
- Human Cytomegalovirus (CMV) is a common virus that can infect people of all ages. It belongs to the family of Herpesviridae and has a complex structure. The virus is composed of an icosahedral protein capsid, which has an average diameter of 100 nm. The capsid is made up of 162 hollow hexagonal and pentagonal capsomeres with an electron-dense core containing the double-stranded DNA genome with 230 kbp nucleotides, forming the nucleocapsid.
- The nucleocapsid is surrounded by a lipoprotein envelope that is derived from the nuclear membrane of the infected host cell. This envelope contains spikes of viral glycoproteins that project from the trilaminar lipid host-derived envelope. These spikes bind to specific host receptors and mediate virus entry into the host cell. Among the glycoproteins, gB is a major surface glycoprotein in CMV that influences virus binding to and entry into the cell and cell-to-cell transmission. On the other hand, gH is a second relatively abundant glycoprotein that is a target for complement independent neutralizing antibodies.
- In mature virus particles, outside the capsid is an amorphous proteinaceous layer called the tegument, which is surrounded by a lipid envelope derived from host cell membranes. The tegument consists of at least 25 proteins, including enzymes such as VP16, which is responsible for subverting cellular proteins and enzymes to involve in viral nucleic acid replication. The tegument also contains VHS (Virion Host Shutoff) protein, which shuts off the host cell protein synthesis in the cytoplasm.
- In summary, the structure of human cytomegalovirus is complex, and it consists of various components such as a protein capsid, nucleocapsid, lipoprotein envelope, glycoproteins, and tegument. Understanding the structure of the virus is crucial in developing effective treatment and prevention strategies.
Genome of Human Cytomegalovirus
A monopartite, linear, approximately 200 kb dsDNA genome. There are both terminal and internal repeated sequences in the genome.
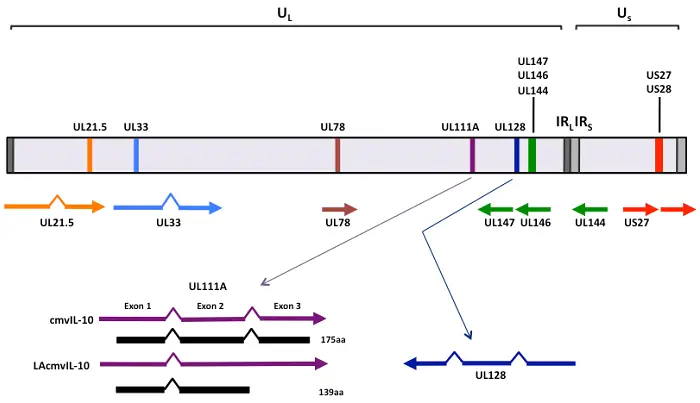
- Human Cytomegalovirus (CMV) has a genome consisting of approximately 230 kilobases of linear double-stranded DNA.
- The genome is monopartite and contains terminal and internal reiterated sequences. It is split into two regions: unique long (UL) and unique short (US) regions, both of which are flanked by inverted sequences.
- The CMV genome contains around 150 open reading frames (ORFs) that encode proteins. Out of these, 41 have been identified as essential and 117 as non-essential for CMV replication.
- The genome plays a crucial role in viral replication, and understanding the genome structure and function can aid in the development of new antiviral treatments.
Replication of Human Cytomegalovirus
- Endocytosis of a virus into a host cell is mediated by the attachment of viral glycoproteins to host receptors.
- Entry into the host cell is still unknown and may depend on the type of host cell, i.e. endocytosis versus plasma membrane fusion.
- The viral DNA is released into the nucleus after the capsid has been transported to the nuclear pore.
- The transcription of immediate early genes, which stimulate the transcription of early genes and protect the virus from the innate host immune system.
- Early viral mRNA transcription by host polymerase II, encoding proteins involved in viral DNA replication.
- Bidirectional replication initiates the first round of circular genome amplification.
- Rolling circle synthesis of linear concatemer copies of viral DNA.
- The transcription of late mRNAs encoding structural proteins by the host polymerase II.
- Assemblage of the virus in nuclear viral factories and branching through the inner lamella of the nuclear membrane, which has been modified by the insertion of herpes glycoproteins, throughout the Golgi, and subsequent release through the plasma membrane.
Latent replication is the replication of a circular viral episome in conjunction with the DNA of the host cell using the host cell’s replication machinery.
Epidemiology of Human Cytomegalovirus
- Human cytomegalovirus infections can be contracted in a variety of methods. As community exposure, CMV is typically transmitted through contact with infected fluids (e.g., sputum, breast milk, blood products) at home and in nursery schools. It is also believed that contact with contaminated urine may play a role in cytomegalovirus transmission.
- Breastfeeding is the most common route of CMV infection in young infants among postnatal contaminations, particularly in populations with high CMV seroprevalence and high rates of breastfeeding from seropositive women during the first 6 months of breastfeeding, with the risk peaking between 4 and 8 weeks after delivery, but persisting for the duration of breastfeeding.
- Even though it is believed that a local reactivation in the mammary glands of mothers with latent CMV infection can result in transmission to the infant, the transmission mechanism has not yet been elucidated.
- As a virus reservoir, these neonates excrete virus in their saliva and urine for extended periods of time, which is then transmitted to other infants, children, and adults. CMV-infected preschoolers can transmit the virus through feces and may infect their parents and instructors in child care centers.
- CMV is transmitted throughout infancy and early adulthood through exposure to saliva and urine. Due to the presence of the virus in seminal and cervical fluids, the virus can also be transmitted sexually.
- Congenital CMV infection (present at birth) occurs via intrauterine transmission of the virus that is believed to be transmitted to the developing fetus in approximately 30% of women undergoing primary infection during pregnancy or by reactivation in women previously immune to CMV at a rate of 1–2% (vertical road).
- In the United States, Canada, Western Europe, and Australia, the prevalence of this infection is estimated to be between 5 and 7 per 1000 live births [5]. Asia and Africa are characterized as having the greatest rates, at 2%.
- Congenital CMV infection contributes to children’s permanent hearing loss, vision impairment, cerebral palsy, and/or cognitive impairment. Approximately 90% of neonates with congenital CMV are asymptomatic, but they are at risk for CMV-associated disabilities [6].
- CMV-related nosocomial infections result from exposure to CMV-containing blood products. Transfusion-acquired CMV infections frequently caused symptomatic illness in children and adults, including hepatitis and thrombocytopenia. Due to the absence of CMV antibodies, newborns who are born to mothers without CMV immunity may develop fatal infections.
- Also susceptible to severe infection are immunocompromised patients who received CMV-infected blood. By using blood products from CMV-seronegative donors, the incidence of CMV infections associated with blood transfusions has decreased dramatically in recent years.
- Infections caused by the transfer of CMV in the allograft are significant causes of morbidity in the early and late posttransplant periods. CMV infection causes long-term graft dysfunction and graft loss, especially in cardiac and lung transplant recipients.
Transmission of Human Cytomegalovirus
- Human Cytomegalovirus (HCMV) is primarily transmitted through bodily fluids such as saliva, urine, blood, breast milk, vaginal secretions, and semen. The virus can spread through close contact with an infected person, such as kissing or sexual contact, or through contact with contaminated objects such as diapers, towels, or toys.
- In addition, the virus can also be transmitted from an infected mother to her unborn child during pregnancy, or during childbirth. This is known as congenital CMV infection and is a significant cause of hearing loss and other health problems in infants.
- People with weakened immune systems, such as those with HIV/AIDS, cancer, or those who have undergone organ transplantation, are at a higher risk of developing severe HCMV infections. The virus can be transmitted through blood transfusions or organ transplantation from an infected donor.
- Preventative measures, such as regular hand washing, avoiding close contact with infected individuals, and practicing safe sex, can help reduce the risk of HCMV transmission. Pregnant women and individuals with weakened immune systems should take extra precautions to avoid exposure to the virus.
Pathogenesis of Human Cytomegalovirus
Human Cytomegalovirus (HCMV) is capable of infecting and replicating in numerous cell types, including epithelial cells, smooth muscle cells, macrophages, and vascular endothelial cells. This broad cell tropism facilitates systemic dissemination within the human body and transmission between hosts. Due to direct contact with infectious secretions from an infected individual, the primary replication of HCMV typically occurs in mucosal epithelium. The virus then spreads via leukocyte-associated viremia, which can last for several months. HCMV can also enter latency in myeloid cells of the bone marrow, resulting in a chronic infection with intermittent reactivation.
In healthy individuals, HCMV infection is typically asymptomatic, but it poses a life-threatening risk for immunocompromised individuals, such as organ transplant recipients and HIV-positive individuals. HCMV is also the primary infectious cause of congenital neurological disease when transmitted through the placenta from the mother to the fetus.
During primary infection, CMV-specific IgM antibodies persist for three to four months, whereas IgG antibodies persist for life. Immunocompetent individuals do not generate IgM antibodies during recurrent infections. IgM and IgG are both produced by the fetus in intrauterine infections, but the fetal IgG response can only be detected after the passively acquired IgG from the mother is catabolized.
It is believed that cell-mediated immunity (CMI) plays a crucial role in the suppression of HCMV infection. CD4+ and CD8+ T cells that recognize HCMV are known to control viral replication and prevent disease progression. In immunocompromised individuals, the absence of CMI permits unrestricted HCMV replication and the development of severe disease.
Clinical manifestations of Human Cytomegalovirus
Human cytomegalovirus (HCMV) infections are generally asymptomatic or result in mild symptoms in healthy individuals. However, in immunocompromised patients, such as organ transplant recipients or people living with HIV, HCMV infection can cause severe and potentially life-threatening diseases. Some of the clinical manifestations of HCMV infection include:
- Retinitis: HCMV can cause inflammation of the retina, leading to vision loss or even blindness.
- Pneumonia: HCMV pneumonia is a serious condition that can occur in immunocompromised individuals and is associated with high morbidity and mortality.
- Gastrointestinal disease: HCMV can cause gastrointestinal ulcers, bleeding, and diarrhea, particularly in patients with AIDS.
- Encephalitis: HCMV encephalitis is a rare but severe condition that can result in seizures, confusion, and coma.
- Congenital infections: HCMV can be transmitted from a pregnant woman to her fetus, leading to congenital HCMV infection. This can cause a range of complications, including hearing loss, intellectual disability, and vision impairment.
- Hepatitis: HCMV hepatitis is a rare but potentially life-threatening complication of HCMV infection in immunocompromised patients.
- Mononucleosis-like syndrome: HCMV infection can cause a syndrome that is similar to infectious mononucleosis, with symptoms such as fever, sore throat, and fatigue.
Laboratory diagnosis of Human Cytomegalovirus
- The laboratory diagnosis of Human Cytomegalovirus (HCMV) infection involves the detection of viral nucleic acid or antigen, as well as the detection of specific antibodies produced in response to the infection.
- Molecular techniques, such as polymerase chain reaction (PCR), are commonly used for the detection of viral DNA or RNA in clinical samples such as blood, urine, saliva, and tissue biopsy. These techniques are highly sensitive and specific, and can be used for the diagnosis of both acute and chronic infections.
- Serological tests, such as enzyme-linked immunosorbent assay (ELISA), are used for the detection of HCMV-specific antibodies in serum or plasma. The presence of IgM antibodies indicates recent infection, while the presence of IgG antibodies indicates past or current infection. IgG avidity assays can also be used to determine the duration of infection.
- In addition, antigen detection assays, such as immunofluorescence assays (IFA) or enzyme immunoassays (EIA), can be used for the detection of viral antigens in clinical samples.
- Culture-based methods, such as cell culture and shell vial culture, are less commonly used due to their lower sensitivity and longer turnaround time compared to molecular and serological assays.
- Overall, the laboratory diagnosis of HCMV infection relies on the use of multiple diagnostic tests and the correlation of clinical and laboratory findings.
Treatment of Human Cytomegalovirus
- There is no specific antiviral treatment available for human cytomegalovirus (HCMV) infection in immunocompetent individuals, as the infection is usually self-limiting and resolves on its own. However, in immunocompromised patients, such as those with HIV or organ transplant recipients, antiviral therapy may be necessary to prevent the progression of the infection.
- The most commonly used antiviral agents for HCMV infection are ganciclovir and valganciclovir, which are nucleoside analogues that interfere with viral DNA replication. Other antiviral agents, such as foscarnet and cidofovir, may be used as second-line treatment options.
- For congenital HCMV infection, antiviral treatment may be considered in cases of symptomatic infection or in infants with evidence of progressive disease. The recommended treatment for congenital HCMV infection is intravenous ganciclovir followed by oral valganciclovir.
- It is important to note that antiviral therapy for HCMV infection may be associated with adverse effects, and careful monitoring of patients is necessary. Additionally, antiviral therapy may not completely eliminate the virus and may only suppress viral replication, which could lead to the development of drug-resistant strains of the virus.
- Preventive measures, such as practicing good hygiene and avoiding contact with bodily fluids of infected individuals, may also help to reduce the risk of HCMV infection. Vaccines for HCMV are currently under development, but none have been approved for use in humans yet.
Prevention and control
Prevention and control measures for Human Cytomegalovirus (HCMV) involve both primary prevention strategies to prevent initial infections and secondary prevention strategies to prevent reactivation or transmission in those who are already infected.
Primary prevention strategies include:
- Hygiene practices: Washing hands frequently with soap and water, avoiding close contact with people who have HCMV infections, avoiding sharing food, utensils, and personal items like towels, and maintaining good personal hygiene can reduce the risk of acquiring HCMV.
- Safe sex practices: Practicing safe sex, including using condoms, can reduce the risk of acquiring HCMV through sexual contact.
- Blood and organ screening: Screening blood and organ donors for HCMV can help prevent transmission of the virus through blood transfusions or organ transplants.
- Education: Educating pregnant women and women of childbearing age about the risks of HCMV and ways to reduce exposure can help prevent congenital infection.
Secondary prevention strategies include:
- Antiviral therapy: Antiviral medications, such as ganciclovir, valganciclovir, foscarnet, and cidofovir, are available for the treatment of HCMV infections in immunocompromised individuals. These drugs can also be used for prophylaxis in high-risk individuals to prevent reactivation of HCMV.
- Immunoglobulin therapy: Administration of intravenous immunoglobulin (IVIG) can be used to prevent or treat HCMV infections in immunocompromised individuals.
- Vaccines: Several vaccines are in development for the prevention of HCMV infections, including live attenuated vaccines and subunit vaccines.
- Infection control: In healthcare settings, strict infection control measures should be implemented to prevent the spread of HCMV between patients, including hand hygiene, wearing gloves, and isolation precautions for patients with active HCMV infection.
In summary, preventing HCMV infections is an important public health concern, especially for vulnerable populations such as pregnant women, neonates, and immunocompromised individuals. Implementation of preventive measures, including hygiene practices, safe sex practices, blood and organ screening, and education, can help reduce the incidence of HCMV infections. In addition, treatment and prevention strategies, including antiviral therapy, immunoglobulin therapy, vaccines, and infection control measures, can help manage and prevent HCMV infections in those who are already infected.
FAQ
What is human cytomegalovirus?
Human cytomegalovirus (HCMV) is a type of herpes virus that can cause mild or severe illness in people of all ages, but it is particularly dangerous for those with weakened immune systems.
How is HCMV transmitted?
HCMV is transmitted through bodily fluids such as blood, saliva, urine, breast milk, and semen. It can be passed from mother to child during pregnancy, childbirth, or breastfeeding. It can also be spread through close personal contact, such as kissing or sexual contact.
What are the symptoms of HCMV?
Many people who are infected with HCMV do not have any symptoms. However, in some people, it can cause symptoms such as fever, fatigue, muscle aches, and swollen glands. In severe cases, it can cause damage to organs such as the liver and brain.
Who is at risk of getting HCMV?
Anyone can get HCMV, but it is more common in people with weakened immune systems, such as those with HIV/AIDS, organ transplant recipients, and infants born to mothers who were infected during pregnancy.
How is HCMV diagnosed?
HCMV can be diagnosed through a blood test that detects antibodies to the virus. A doctor may also perform a test to detect the virus itself in bodily fluids or tissue samples.
Can HCMV be treated?
There are antiviral medications that can be used to treat HCMV, particularly in people with weakened immune systems. However, there is no cure for the virus and it can persist in the body for life.
Is there a vaccine for HCMV?
Currently, there is no licensed vaccine for HCMV, but research is ongoing to develop a safe and effective vaccine.
How can HCMV be prevented?
The best way to prevent HCMV infection is to practice good hygiene, such as washing your hands frequently, avoiding contact with bodily fluids, and not sharing food, drinks, or utensils with others. Pregnant women who have never been infected with HCMV should avoid contact with young children, who are often carriers of the virus.
What are the risks of HCMV during pregnancy?
If a woman becomes infected with HCMV during pregnancy, there is a risk that the virus can be passed to the fetus, which can cause serious health problems such as hearing loss, vision loss, intellectual disability, and developmental delays.
Can HCMV be spread through blood transfusions?
Yes, HCMV can be spread through blood transfusions or organ transplants from an infected donor. However, blood and organ donors are screened for HCMV to reduce the risk of transmission.