pH meter is an instrument used to measure the acidity or alkalinity of a solution. It gives the value of pH directly. It is mainly used for aqueous solution.
It works like a sensitive voltmeter. It measures the voltage difference produced between two electrodes. One is pH glass electrode and another is reference electrode.
The glass electrode is sensitive to hydrogen ion activity. When it is dipped in the solution, a small electrical potential is produced. This potential changes according to the amount of H⁺ ion present in the solution.
The reference electrode gives a constant potential. It does not change much with the sample. The meter compares both electrode potentials and converts the voltage into pH value.
The conversion is based on Nernst equation. The reading is shown on the display as acidic, neutral or alkaline value. Solution below pH 7 is acidic. pH 7 is neutral and above pH 7 is alkaline.
In early time, pH was measured by colour change of indicators. But coloured, cloudy and reactive solutions gave wrong result. Later potentiometric method was used and it became more accurate.
Earlier mercury chloride reference electrode was used for stable potential. Now silver/silver chloride (Ag/AgCl) reference electrode is commonly used. Mercury system is avoided because mercury is toxic and not suitable for high temperature work.
Why pH Meters need to Calibrate?
- pH meter need calibration because it measures pH by voltage change. This voltage is in millivolt. The meter has to convert this voltage into correct pH value.
- The electrode does not always give perfect value. Even new electrode may have little difference from ideal value. So calibration is needed before accurate measurement.
- The glass membrane of pH electrode becomes old with time. Due to this, its response to H⁺ ion slowly changes. Then the reading may become wrong.
- The internal electrolyte of electrode may become weak or less. This also changes the electrode potential. So the meter must be adjusted again with standard buffer.
- Temperature change affects the electrode response. The same solution may give slightly different reading at different temperature. Calibration helps to reduce this error.
- Chemicals present in sample may coat the electrode surface. Dirt, salt, protein or other material may stick on glass membrane. This makes the electrode response slow and inaccurate.
- During calibration, standard buffer solutions of known pH are used. The meter compares its reading with the known buffer value. Then it corrects the slope and offset.
- Thus calibration makes the pH meter reading accurate and reliable. Without calibration, the meter may show wrong acidity or alkalinity of the solution.
Preparation of Standard Buffer for Calibration of a pH Meter
It is based on the fact that each buffer maintain a constant pH when it is dissolved properly and the solution is allowed to mix uniformly. The buffer act as the reference medium during the adjustment of the instrument, and any variations in dissolution or contamination is reflected in the reading.
Preparation of pH 4.00 Buffer
The pH 4.00 buffer at about 20°C is prepared by transferring the capsule powder into a 100 ml volumetric flask. It is observed that purified water help in dissolving the contents, which is necessary for correct calibration. In this step, nearly 80 ml water is added and stirred until the powder is dispersed.
After mixing, the volume is made up to the mark producing an uniform solution. It is the process where small fragments sometimes float before complete dissolution.
Preparation of pH 7.00 Buffer
In this step, the pH 7.00 buffer capsule is poured into the 100 ml flask. It is used to provide the mid-range reference point. About 80 ml purified water is added so that the powder is dissolved forming a homogeneous mixture.
The final volume is adjusted to 100 ml although the alignment of the meniscus is sometimes tricky. Gentle swirling is helpful in reducing the undissolved granules that appear first.
Preparation of pH 9.20 Buffer
The pH 9.20 buffer is prepared next. It is the process where the tablet is placed inside the volumetric flask giving a slightly alkaline solution needed for upper pH calibration. The addition of nearly 80 ml water allow the contents to break down.
Then the volume is taken to 100 ml. It is sometimes seen that slow mixing is preferred because rapid shaking produce micro-bubbles which affect the reading during calibration.
pH Meter Calibration Procedure
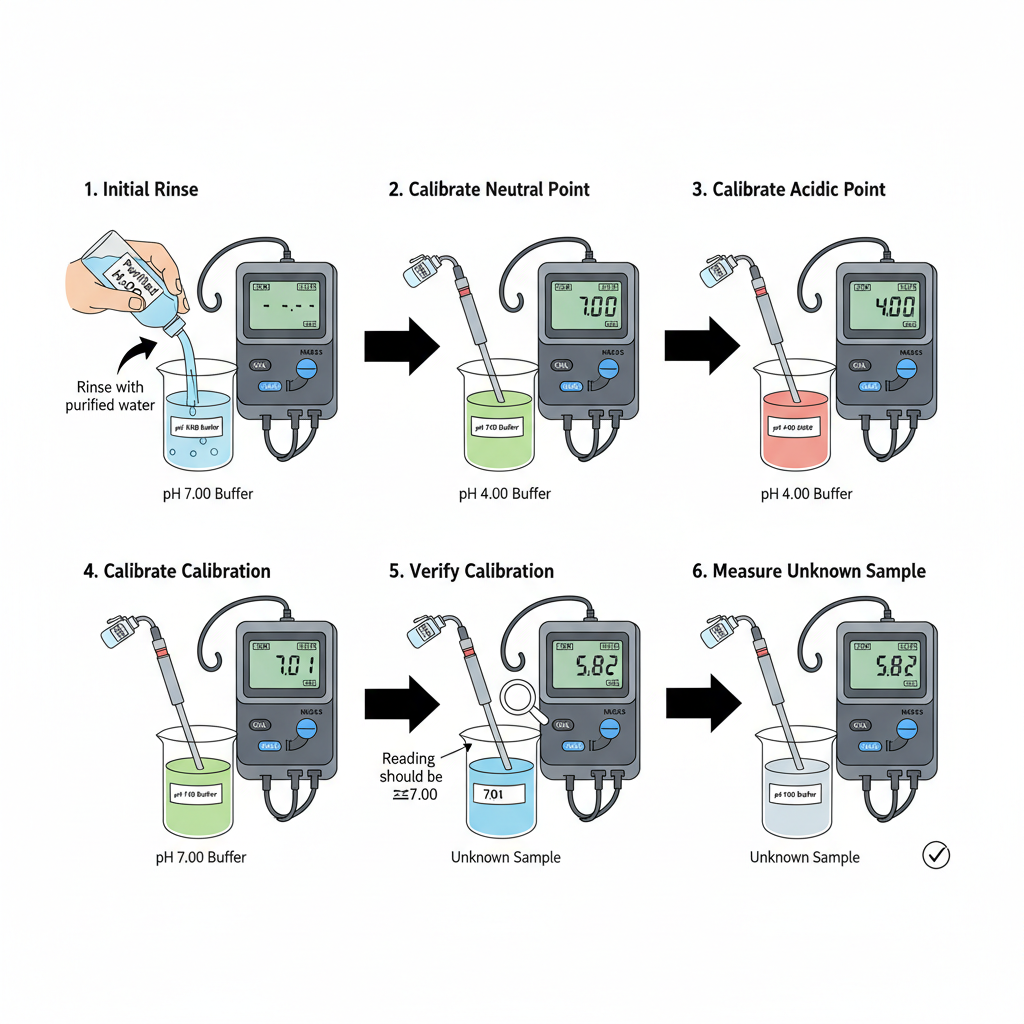
- pH meter is switched on first. It is kept for some time. The display should come to stable condition before calibration.
- The pH electrode is rinsed with purified water. This remove the previous solution from the electrode. Extra water is shaken off gently.
- pH 7.00 buffer is taken first. It is used as the neutral reference point. Fresh buffer is used because contaminated buffer gives wrong reading.
- The electrode is dipped in pH 7.00 buffer. The glass bulb should be fully covered by the buffer. Slight stirring is done for uniform contact of electrode and solution.
- The meter is kept undisturbed for some time. The reading is allowed to stop drifting. After the reading becomes stable, the value is adjusted to 7.00. This is the initial point setting.
- The electrode is taken out from pH 7.00 buffer. It is washed again with purified water. This is done because buffer drops remaining on the electrode may change the next buffer value.
- The electrode is then placed in pH 4.00 buffer. This buffer is slightly acidic. The reading may change slowly at first.
- The value is allowed to become steady. After stabilization, the meter is set to 4.00. This gives the acidic range calibration.
- The electrode is removed from pH 4.00 buffer. It is washed properly with purified water. Acidic residue should not remain on the glass surface.
- The electrode is dipped in pH 9.20 buffer. This is used for upper pH range calibration. Alkaline buffer sometimes take more time to settle.
- The reading is allowed to become stable. After the reading stops drifting, the meter is adjusted to 9.20.
- The electrode is rinsed again with purified water. It is shaken lightly to remove attached droplets. The bulb is not rubbed.
- Sometimes verification is done by putting the electrode again in pH 7.00 buffer. If the meter shows nearly 7.00, the calibration is considered correct.
- After these steps, the pH meter is ready for use. The electrode can now be used for measuring unknown solution.
How Often To Recalibrate pH Meter?
- pH meter should be recalibrated when a new electrode is used. New electrode also may not give exact reading without calibration.
- It should be recalibrated when old electrode is replaced. The response of each electrode is different, so calibration is needed again.
- Recalibration is done after cleaning of electrode. Cleaning may change the surface condition of glass bulb. So the meter should be checked again with buffer.
- It is recalibrated after maintenance work. If electrode or meter is disturbed, the reading may shift from the correct value.
- If the pH meter was kept unused for long time, calibration is done before use. Long storage may make the electrode response slow.
- For high precision laboratory work, calibration is done daily. Sometimes it is done before each batch of sample. In very accurate work, calibration is done before every measurement also.
- For field work, calibration is done before each sampling trip. Portable meter may change reading due to movement, temperature and outdoor condition.
- For routine work, calibration once or twice in a week may be enough. This is used when the sample condition is same and very high accuracy is not required.
- For home use or aquarium use, calibration may be done weekly or monthly. It depends on how often the meter is used.
- More frequent calibration is needed for harsh samples. Strong acids, strong bases, dirty samples, low ion solution and samples with temperature change can affect the electrode. So in these cases, calibration is done more often.
Consequences of Not Calibrating a pH Meter
- pH meter gives inaccurate reading if it is not calibrated. The electrode does not always work like ideal condition. So the shown pH value may be different from the actual value.
- The data becomes unreliable. In laboratory work, environmental testing and quality control, wrong pH reading can affect the whole result. The sample may be considered acidic or alkaline wrongly.
- Electrode drift is not corrected. With time the pH electrode changes its response due to ageing of glass membrane, electrolyte dilution and contamination. Without calibration, this drift remains uncorrected.
- The slope and zero point of electrode are not adjusted. The meter cannot calculate the actual condition of electrode. So even if the meter display looks stable, the reading may still be wrong.
- Damaged electrode may not be identified. During calibration, electrode condition can be checked by slope and offset value. If calibration is not done, a weak or dead electrode may be used unknowingly.
- Temperature error may occur. Temperature affects the solution pH and also electrode response. Without calibration with proper buffer, the meter cannot adjust the reading correctly.
- The experiment may fail due to wrong pH value. Chemical reaction, biological culture, enzyme activity and industrial process may not work properly if the pH is not correct.
- Time and reagent may be wasted. Wrong reading may cause repeated testing and correction. In some cases, the whole sample or batch may need to be discarded.
- Product quality may be affected. In food, pharmaceutical and chemical industries, wrong pH control can change the safety and stability of product. So regular calibration is important.
References
- Atlas Scientific. (2025, July 30). How to calibrate a pH meter correctly. https://atlas-scientific.com/blog/how-to-calibrate-ph-meter/
- Atlas Scientific. (2025, September 26). What is pH drift and how to fix it? A complete troubleshooting guide. https://atlas-scientific.com/blog/ph-drift/
- Chemistry Stack Exchange. (2015, August 9). Solution for storing a glass electrode. https://chemistry.stackexchange.com/questions/34878/solution-for-storing-a-glass-electrode
- Chromservis. (n.d.). Cleaning & reconditioning pH electrodes. https://www.chromservis.eu/en/cleaning-reconditioning-ph-electrodes
- Cole-Parmer. (n.d.). How to store, clean, and recondition pH electrodes. https://www.coleparmer.com/tech-article/how-to-store-clean-and-recondition-ph-electrodes
- Cole-Parmer. (n.d.). pH electrode selection guide. https://www.coleparmer.com/tech-article/ph-electrode-selection-guide
- Cole-Parmer. (n.d.). Troubleshooting your pH tester: What to do, what not to do, and why. https://www.coleparmer.com/tech-article/ph-tester-troubleshooting
- Convergent Water Controls (CWC). (n.d.). Which pH buffer solution should I use first when calibrating? https://blog.cwc.com.au/which-ph-buffer-solution-should-i-use-first-when-calibrating
- Cushman, C. (2019, February 14). Anatomy of pH electrodes. YSI. https://www.ysi.com/ysi-blog/water-blogged-blog/2019/02/anatomy-of-ph-electrodes
- Daly, J. (2023, February 23). How to clean pH electrodes for maximum life. South Fork Instruments. https://www.southforkinst.com/how-to-clean-ph-electrodes-for-maximum-life/
- Environmental Protection Agency (EPA). (n.d.). 9040C pH electrometric measurement. https://www.epa.gov/sites/default/files/2015-12/documents/9040c.pdf
- Fabsits, P. (2007). Checking pH electrode functionality – There are many different industries that rely on the use of a pH meter. Sigma-Aldrich. https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/marketing/global/documents/169/757/labwarenotes_v1_6.pdf
- GlobalSpec. (n.d.). How often should you calibrate your pH sensor? https://insights.globalspec.com/article/24748/how-often-should-you-calibrate-your-ph-sensor
- Goodwin, N. (2025, December 2). Why is my pH reading slow to stabilise? 10 common causes and how to fix them. TPS. https://tps.com.au/blogs/articles-news/why-is-my-ph-readings-slow-to-stabilise
- Hach. (2024, April 17). What buffers should be used to calibrate a pH probe? https://waterqualitygroup.my.site.com/MyHachCustomerService/s/article/KA-en-US-TE483-1000412
- Hamilton Company. (n.d.). pH calibration problems. Knowledge Center. https://www.hamiltoncompany.com/knowledge-base/article/ph-calibration-problems
- Hanna Instruments. (2017, August). pH electrode maintenance guide. https://pages.hannainst.com/hubfs/006-finished-content/pH_Guides/ph-electrode-maintenance-guide–hanna-instruments.pdf
- Hanna Instruments. (2017, August). pH electrode troubleshooting guide. https://pages.hannainst.com/hubfs/006-finished-content/pH_Guides/ph-electrode-troubleshooting-guide–hanna-instruments.pdf
- Hanna Instruments. (n.d.). pH meter calibration and electrode maintenance guide. https://blog.hannainst.com/ph-meter-calibration
- HORIBA. (n.d.). pH electrode care and maintenance procedures. https://www.horiba.com/int/water-quality/support/technical-tips/electrodes/ph-electrode-care-and-maintenance-procedures/
- Li, C. (2026, March 26). pH meter calibration complete guide: Step-by-step procedures. GAIMC. https://www.gaimc.com/Newsinfo/pH-Meter-Calibration-Complete-Guide
- M4 Knick. (2020, May 21). A better pH calibration when switching to automatic mode. https://m4knick.com/a-better-ph-calibration-when-switching-to-automatic-mode/
- M4 Knick. (2023, January 19). 2-point and 3-point calibration for pH measurements. https://m4knick.com/2-point-and-3-point-calibration-for-ph-measurements/
- Mettler Toledo. (2000, June). Instruction manual pH-meter 1120 and pH-meter 1120-X. https://www.mt.com/dam/mt_ext_files/Editorial/Generic/0/BA_pH_PortableMeter_1120_0x000010083e770640400046cf_files/ba_ph_portablemeter1120e.pdf
- Mettler Toledo. (n.d.). pH electrode maintenance guide. GWB. https://gwb.fi/wp-content/uploads/2023/10/pH_Mettler-elektrodin-kayttoonotto-ja-yllapito.pdf
- Morais, J. (n.d.). How to calibrate a pH meter. Joan Morais Cosmetics School. https://joanmorais.com/how-to-calibrate-a-ph-meter/
- Murphy and Dickey Instrumentation and Pumps. (n.d.). Calibration tips for pH | Your guide to perfect pH calibration. https://murphyanddickey.com/calibration-tips-for-ph/
- PASCO. (n.d.). pH electrode maintenance. Knowledge Base. https://www.pasco.com/support/knowledge-base/21
- Paxman, R. (2018, September 26). Care and maintenance of pH meter electrodes. ThermoWorks Help Center. https://help.thermoworks.com/knowledge-base/care-maintenance-ph-meter-electrodes/
- Reddit (r/labrats). (n.d.). Trouble with pH meter. https://www.reddit.com/r/labrats/comments/1qhovzc/trouble_with_ph_meter/
- Reithmayer, K. (2025, April 2). FAQ pH measurement. Xylem Analytics. https://www.xylemanalytics.com/en/company/blog/blog/2025/04/faq-ph-measurement
- ResearchGate. (n.d.). How can I fix or refresh the pH electrodes? https://www.researchgate.net/post/How-can-I-fix-or-refresh-the-pH-electrodes
- ResearchGate. (n.d.). How can I prepare my own electrode filling and storage solution? https://www.researchgate.net/post/How_can_I_prepare_my_own_electrode_filling_and_storage_solution
- Ryan Herco Flow Solutions. (2018). Everything you need to know about pH sensor calibration. RHFS Pulse. https://www.rhfs.com/pulse/everything-you-need-to-know-about-ph-sensor-calibration/
- Simpson, E. (2018, July 4). A guide to taking proper care of your pH electrodes. Westlab. https://www.westlab.com/blog/a-guide-to-taking-proper-care-of-your-ph-electrodes
- Thermo Fisher Scientific. (2008, August). pH calibration procedure for optimal measurement precision. https://tools.thermofisher.com/content/sfs/brochures/TN-ph-calibration-procedure-for-optimal-measurement-precision-T-PHCAL-EN.pdf
- Thermo Fisher Scientific. (2009). pH measurement handbook. https://documents.thermofisher.com/TFS-Assets/LSG/brochures/pH-Measurement-Handbook-S-PHREFBK-E.pdf
- Turtle Tough. (n.d.). FAQ – Calibration procedure for pH sensors 2 point. https://turtletoughsensors.com/images/FAQ/FAQ%20-%20Calibration%20Procedure%20for%20pH%20Sensors_Rev1.1.pdf
- Unknown. (n.d.). Advanced electrochemical protocols for pH meter calibration, validation, and systematic maintenance. [Markdown document].
- Westlab. (2020, December 11). Storing a pH electrode: The correct way. https://www.westlab.com/blog/what-is-the-correct-way-to-store-a-ph-electrode
- YSI. (2015). pH electrode calibration guide. https://www.ysi.com/file%20library/documents/guides/ysi_ph_electrode_calibration_guide_w77_0815.pdf