What is Gerhardt’s test?
Gerhardt’s test is a classical qualitative urine test used for the detection of acetoacetic acid which is a type of ketone body. It is also known as ferric chloride test. It is mainly performed in cases of suspected ketonuria especially in diabetes mellitus.
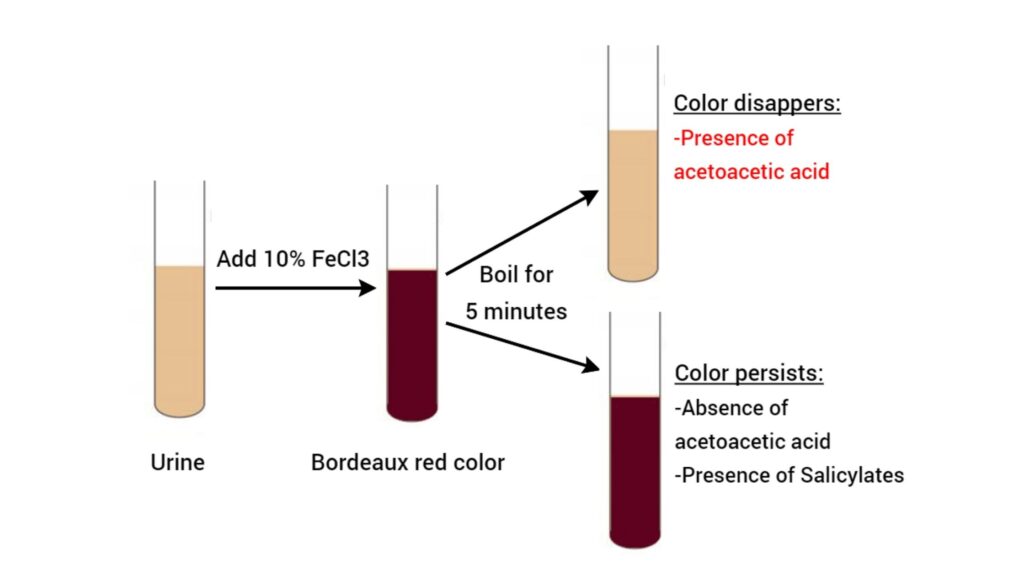
The principle of Gerhardt’s test is based on the reaction between ferric chloride (FeCl₃) and acetoacetic acid present in urine. It is the process in which ferric ions reacts with acetoacetic acid forming a coloured complex. When 10% ferric chloride solution is added dropwise to a fresh urine sample a Bordeaux red or port-wine colour is produced. This colour formation indicates the presence of acetoacetic acid.
However some substances like salicylates (aspirin) also produce similar red colour. So confirmation is required. Acetoacetic acid is heat unstable and it is destroyed by boiling. Therefore the urine sample is boiled for few minutes. If the red colour disappears after boiling it confirms true positive test for ketone bodies. If the red colour persists after boiling it indicates false positive reaction due to other heat stable interfering substances.
Objective
- To detect the presence of ketone bodies within the supplied urine sample.
Gerhardt’s test Principle
The principle of Gerhardt’s test is based on the chemical reaction between ferric chloride (FeCl₃) and acetoacetic acid present in urine. It is the process in which trivalent ferric ions (Fe³⁺) bind with the enol form of acetoacetic acid forming a coordination complex called ferric acetoacetate. This reaction depends on the strong affinity of ferric ions towards the ketone body.
When 10% ferric chloride solution is added to the urine sample a Bordeaux-red or port-wine colour is produced. This colour formation is the initial positive indicator of ketone bodies. The complex formed between ferric ions and acetoacetic acid is responsible for this characteristic colour.
However some other substances like salicylates and phenols also give similar red colour. So a confirmatory boiling step is essential. Acetoacetic acid is thermally unstable and it breaks down into carbon dioxide and acetone on heating. Acetone does not react with ferric chloride. Therefore if the red colour disappears after boiling it confirms the presence of acetoacetic acid. If the red colour persists after boiling it indicates false positive reaction due to other heat stable compounds.
Gerhardt’s test reaction
Gerhardt’s test reaction is based on the chemical interaction between ferric chloride (FeCl₃) and acetoacetic acid present in urine. It is the reaction in which ferric ions (Fe³⁺) combine with the enol form of acetoacetic acid forming a coordination complex called ferric acetoacetate. This complex formation is responsible for the colour development.
When 10% ferric chloride solution is added to the urine sample a Bordeaux red or port-wine colour is produced. This colour is taken as the initial positive reaction for ketone bodies. The reaction can be represented as follows–
Acetoacetic acid + Fe³⁺ → Ferric acetoacetate complex (Bordeaux red colour)
However the test is prone to false positive reactions. Substances like salicylates (aspirin) also reacts with ferric chloride producing similar red colour. So boiling step is necessary. When the coloured solution is boiled for 3–5 minutes acetoacetic acid being thermally unstable degrades into carbon dioxide and acetone. Acetone does not react with ferric chloride and therefore the red colour disappears in true positive case.
If the red colour persists after boiling it indicates false positive reaction due to heat stable interfering substances. It should be noted that this test detects only acetoacetic acid and it does not react with other ketone bodies like acetone or β-hydroxybutyrate.
Requirements For Gerhardt’s test
- Fresh urine sample – 5–10 mL of freshly voided urine is required. The sample should be tested immediately because acetoacetic acid is unstable and it degrades into acetone at room temperature. If delay occurs it is kept in refrigerator in a closed container.
- 10% Ferric chloride (FeCl₃) solution – It is the chemical reagent used in this test. The solution is added dropwise to the urine sample to observe colour reaction.
- Clean glass test tubes – These are required to hold and mix the urine and ferric chloride solution.
- Heating source – A flame or boiling water bath is required. The solution is boiled for 3–5 minutes. This step is mandatory to confirm that the red colour is due to acetoacetic acid and not due to heat stable drugs like aspirin (salicylates).
- Filter paper and funnel (optional) – It is used if white precipitate of phosphates is formed after adding ferric chloride. The solution is filtered to see the colour change clearly.
Gerhardt’s test Procedure
- Fresh urine sample is taken – About 3–10 mL of freshly voided urine is transferred into a clean and dry glass test tube.
- Addition of ferric chloride – 10% ferric chloride (FeCl₃) solution is added drop by drop to the urine sample and mixed properly after each addition.
- Removal of phosphates – After adding first few drops a thick white precipitate of ferric phosphate is usually formed. Ferric chloride is added slowly until the white precipitate no longer forms.
- Filtration (if required) – If the precipitate is dense and obscures the solution the mixture is filtered to obtain a clear filtrate.
- Observation of initial colour – Few more drops of ferric chloride solution is added to the clear filtrate. If Bordeaux-red or port-wine colour develops it is taken as presumptive positive result for acetoacetic acid.
- Confirmatory boiling step – The test tube is heated over flame or in boiling water bath for 3–5 minutes. This step is essential because other substances like salicylates may also produce red colour.
- Interpretation of result – If the red colour disappears or turns yellowish brown after boiling it confirms the presence of acetoacetic acid (true positive). If the red colour persists or becomes intense after boiling it indicates false positive reaction due to heat stable interfering substances like aspirin.
Observations and Results of Gerhardt’s test
- Initial white precipitate – On adding first few drops of ferric chloride a thick white precipitate of ferric phosphate may be formed. This precipitate may dissolve on further addition of reagent or it is removed by filtration.
- Negative result – No colour change is observed and the solution retains the usual yellow colour of ferric chloride reagent.
- Presumptive positive result – The solution develops Bordeaux-red or port-wine or burgundy colour. This is the initial indicator of acetoacetic acid in urine.
- True positive (after boiling) – When the solution is boiled for 3–5 minutes the red colour disappears or becomes yellowish brown. It confirms the presence of acetoacetic acid.
- False positive (after boiling) – The red colour persists or becomes more intense after boiling. It indicates the presence of heat stable interfering substances like salicylates (aspirin) or antipyrine.
- Atypical colour interferences –
- Pink or purple colour may indicate presence of phenothiazines.
- Deep green colour indicates phenylpyruvic acid (in phenylketonuria) or bilirubin or urobilin.
- Blue or green transient colour indicates homogentisic acid (in alkaptonuria).
- Grey or black colour indicates presence of melanin.
- Grading of positive results (based on colour intensity) –
- 1+ (Small) – Faint red or reddish brown colour.
- 2+ (Moderate) – Clear Bordeaux red colour.
- 3+ (Large) – Deep intense port-wine colour.
- 4+ (Very large) – Extremely dark nearly opaque red colour.
Uses of Gerhardt’s test
- Detection of ketone bodies – It is classically used for the detection of acetoacetic acid in urine. It helps in assessing ketonuria and ketosis in patients.
- Evaluation of diabetic ketoacidosis (DKA) – The test is used in diabetic patients for monitoring ketone level. A positive result indicates high level of ketone bodies and severe metabolic decompensation.
- Identification of other metabolic crises – In non diabetic individuals a positive test may indicate severe starvation, nutritional depletion, alcoholic ketoacidosis or hyperemesis gravidarum.
- Screening for salicylate toxicity – Since salicylates (aspirin) reacts with ferric chloride producing red colour the test is also used as rapid bedside method to detect salicylate overdose and intoxication.
- Diagnosis of phenylketonuria (PKU) – Historically the test was used as screening method for PKU. It produces green colour when it reacts with phenylpyruvic acid present in urine.
- Chemical analysis of phenols – In laboratory setting it is used to detect presence of phenols, enols, oximes and sulfinic acids in chemical samples or plant extracts.
Limitations of Gerhardt’s test
- Low sensitivity – The test is less sensitive as compared to other ketone tests like Rothera’s test. High concentration of acetoacetate (about 25–80 mg/dL) is required to produce positive result.
- Incomplete ketone detection – It detects only acetoacetic acid. It completely fails to detect other ketone bodies like acetone and β-hydroxybutyrate.
- High rate of false positive – Many drugs like salicylates (aspirin), phenothiazines, L-dopa and antipyrine reacts with ferric chloride giving similar red or purple colour.
- Requirement of confirmatory boiling step – Due to frequent drug interference boiling step is mandatory. It is done to differentiate heat labile acetoacetate from heat stable drug metabolites.
- Effect of pH and technique errors – Excessively acidic urine or addition of too much ferric chloride may cause false negative result. Highly basic urine may form brown orange precipitate which creates confusion.
- Sample instability – Acetoacetate is unstable and it spontaneously degrades into acetone at room temperature. Delayed or unpreserved samples may give false negative result.
- Colour interference – Substances like bilirubin, high bicarbonates or metabolites of phenylketonuria (PKU) may produce green, brown or blue colour which mask the actual result.
- Urine concentration variation – Highly concentrated urine may intensify the colour reaction whereas very dilute urine may mask mild ketosis and give false negative result.
Precautions of Gerhardt’s test
- Freshness of specimen – Urine sample should be tested immediately after voiding. If delay occurs it is kept in refrigerator in tightly closed container. Acetoacetic acid is chemically unstable and it may break down into acetone at room temperature or be destroyed by bacterial action.
- Use of proper preservatives – If 24 hour urine collection is required suitable preservatives should be used. It helps in maintaining integrity of ketone bodies.
- Storage of reagent – 10% ferric chloride reagent should be stored in amber coloured glass bottle to prevent photochemical degradation. Before use the reagent should be clear and free from heavy sediment.
- Careful addition of reagent – Ferric chloride solution should be added slowly drop by drop. Urinary phosphates should be completely precipitated and filtered if they obscure the colour. Excess addition of ferric chloride should be avoided as it may give false negative result.
- Mandatory boiling step – If red colour develops boiling for 3–5 minutes must be done. It is essential to differentiate true positive from false positive caused by heat stable drugs like salicylates (aspirin) or phenothiazines.
- Attention to pH – The pH of the solution should be considered. If excessively acidic the colour complex may not form giving false negative. If highly basic brown orange precipitate of ferric hydroxide may be formed and obscure the result.
- Consider urine concentration – Specific gravity of urine should be taken into account. Highly concentrated urine may intensify colour reaction whereas very dilute urine may mask mild ketosis and give false negative result.
FAQ
What is Gerhardt’s test?
Gerhardt’s test is a laboratory test used to qualitatively detect the presence of ketone bodies in urine.
What are ketone bodies?
Ketone bodies are a byproduct of fat metabolism and include acetone, acetoacetic acid, and beta-hydroxybutyrate.
What can cause ketonuria?
Ketonuria, the presence of excess ketone bodies in urine, can be caused by conditions such as starvation, uncontrolled diabetes, persistent vomiting, and a high-fat, low-carbohydrate diet.
How does Gerhardt’s test work?
Gerhardt’s test is a type of nitroprusside test that detects acetoacetic acid in urine. It is not considered very sensitive, as it can only detect levels of acetoacetic acid between 25 to 50 mg/dl.
What is the relationship between ketosis and diabetic ketoacidosis?
Ketosis is the presence of ketone bodies in the body, while diabetic ketoacidosis is a condition where the body produces excess ketone bodies due to uncontrolled diabetes. Ketosis can also occur due to starvation, a high-fat, low-carbohydrate diet, or persistent vomiting.
What is the color formed when ferric chloride reacts with acetoacetic acid in Gerhardt’s test?
When ferric chloride reacts with acetoacetic acid in Gerhardt’s test, a port wine or Bordeaux red color is formed.
Can acetone be detected using Gerhardt’s test?
No, acetone cannot be detected using Gerhardt’s test as it does not react with ferric chloride.
What happens to acetoacetic acid when it is boiled during Gerhardt’s test?
When acetoacetic acid is boiled during Gerhardt’s test, it loses carbon dioxide and transforms into acetone.
What does the disappearance of the color during Gerhardt’s test indicate?
The disappearance of the color during Gerhardt’s test indicates that acetoacetic acid is present within the sample being tested.
What is the significance of the color disappearance in a urine test?
The disappearance of color in a urine test indicates the presence of Acetoacetic acid in the urine sample.
What does a persistent color mean in a urine test?
The persistence of color in a urine test indicates the absence of Acetoacetic acid in the urine sample.
Can Acetoacetic acid be found in a negative urine test result?
No, Acetoacetic acid cannot be found in a negative urine test result, as the persistent color indicates its absence.
Is the disappearance of color the only indicator of Acetoacetic acid in a urine test?
Yes, the disappearance of color is the only indicator of Acetoacetic acid in a urine test, as per the given text.
What are the possible reasons for a false negative result in a urine test?
The possible reasons for a false negative result in a urine test could be an error in the testing process, such as not following the instructions correctly, or a low concentration of Acetoacetic acid in the urine sample.
References
- Comprehensive analysis of the Gerhardt ferric chloride test: Biochemical principles, clinical protocols, and diagnostic interferences in metabolic monitoring. (n.d.).
- Comstock, J. P., & Garber, A. J. (1990). Ketonuria. In H. K. Walker, W. D. Hall, & J. W. Hurst (Eds.), Clinical methods: The history, physical, and laboratory examinations (3rd ed.). Butterworths.
- Ferric chloride test. (2025, April 18). In Wikipedia.
- Grech, J. L. (n.d.). The examination of urine. The Chest-Piece, 1(2), 25–33.
- Labpedia.net. (2025, February 6). Urine for ketones, and ketone bodies (ketonuria).
- Labpedia.net. (2025, November 6). Diabetes mellitus:- Part 7 – Diabetic ketoacidosis and ketone bodies.
- Messner, D. A. (2012, May 17). On the scent: The discovery of PKU. Distillations. Science History Institute.
- Vissa, S., & Rao, B. S. S. (n.d.). Test for ketone bodies in urine. Histopathology.guru.
- Wisniak, J. (2006). Charles Fréderic Gerhardt. Educación Química, 17(3), 343–356. https://doi.org/10.22201/fq.18708404e.2006.3.66037
Thanks ,the content has been helpful to writing my report and being on top