Bial’s test is a chemical test used for detection of pentoses (5-carbon sugars) and it is used to distinguish pentoses from hexoses. It was developed by German physician Manfred Bial. It is also used in laboratory for estimation of RNA because RNA contains pentose sugar ribose.
It works on principle of acid dehydration of sugars. When sample containing pentose is mixed with Bial’s reagent and heated the concentrated hydrochloric acid dehydrates pentose to form furfural. This furfural is then condensed with orcinol in presence of ferric ions (Fe3+) and a distinct blue or green coloured complex is formed which indicates positive test. When hexose is present it is dehydrated to hydroxymethylfurfural and it reacts to give muddy-brown yellow or grey colour which is considered negative for pentose.
Bial’s reagent is composed of orcinol concentrated hydrochloric acid (HCl) and ferric chloride (FeCl3). In procedure a small amount of sample is taken in test tube reagent is added and the mixture is heated for few minutes (generally in boiling water bath) and then colour is observed.
If blue or green colour is produced pentose is present. If muddy-brown yellow or grey colour is produced it indicates hexose and negative for pentose. This test is used for detection of pentose sugars in carbohydrate sample and for RNA detection and it has also been adapted for detection of aqueous formaldehyde.
Objectives of Bial’s Test
Objectives of Bial’s Test are-
- To detect presence of pentoses and pentosans in given sample.
- To distinguish pentoses from hexoses (six-carbon sugars) and other carbohydrate derivatives.
- To quantify concentration of pentose sugars which is commonly used for estimation of RNA present in a solution.
- To detect and quantify aqueous formaldehyde as a rapid low-cost method for monitoring this compound in industrial and medical applications.
Principle of Bial’s test
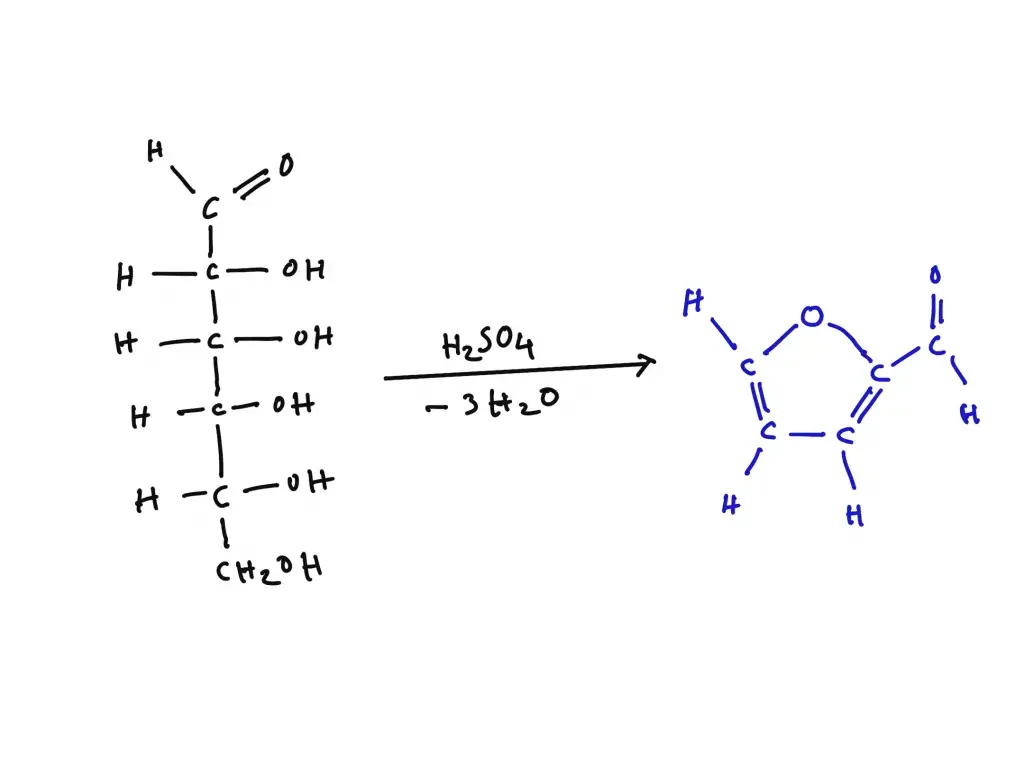
Principle of Bial’s test is based on acid catalyzed dehydration of pentose sugars. When sample is heated with Bial’s reagent (hydrochloric acid, orcinol and ferric chloride) the hydrochloric acid dehydrates pentoses present in sample to form furfural. This furfural is then condensed with orcinol and iron ions and a distinct blue or blue-green coloured complex is formed. This colour change is considered as positive test for pentose.
When hexose sugars are present the acid dehydrates them to hydroxymethylfurfural. This compound reacts with orcinol to form muddy-brown yellow or grey condensation product and it is considered negative for pentose. This difference in colour helps in distinguishing pentose and hexose sugars.
Requirements for Bial’s Test
Requirements for Bial’s Test are-
- Test tubes test tube rack and test tube caps.
- Pipette and beakers.
- Bunsen burner mantle heater or hot water bath for heating the reaction mixture.
- Vortex mixer or glass rod for mixing of solution.
- UV spectrophotometer or colorimeter (only required for quantitative analysis).
- Standard lab supplies like ice bath tissue paper and wash bottles.
- Bial’s reagent– It is composed of orcinol concentrated hydrochloric acid (HCl) and ferric chloride (FeCl3) (sometimes ethanol is also used depending on protocol).
- Sample solution– Unknown carbohydrate or compound to be tested.
- Control solution– Known pentose sugar (ribose or xylose) as positive control and distilled water as blank or negative control.
Procedure of Bial’s test
Detail step by step Procedure of Bial’s test are-
- Step 1- A clean and dry test tube is taken. About 1 to 2 ml of carbohydrate sample solution (or about 10 drops) is added into the test tube.
- Step 2- Equal volume of Bial’s reagent is added into the same test tube (about 2 to 3 ml). The content is mixed properly by swirling or mixing with glass rod.
- Step 3- The test tube is placed in boiling water bath or it is heated gently over Bunsen burner. The heating time is maintained for about 1 to 5 minutes depending on protocol.
- Step 4- After heating the test tube is removed from heat source and it is allowed to cool to room temperature.
- Step 5- The colour change is observed visually against a white background. Blue or green colour indicates positive test for pentose. If colour is not clear some water is added into the test tube to clarify the result.
Result and Interpretation of Bial’s Test
Result and Interpretation of Bial’s Test are-
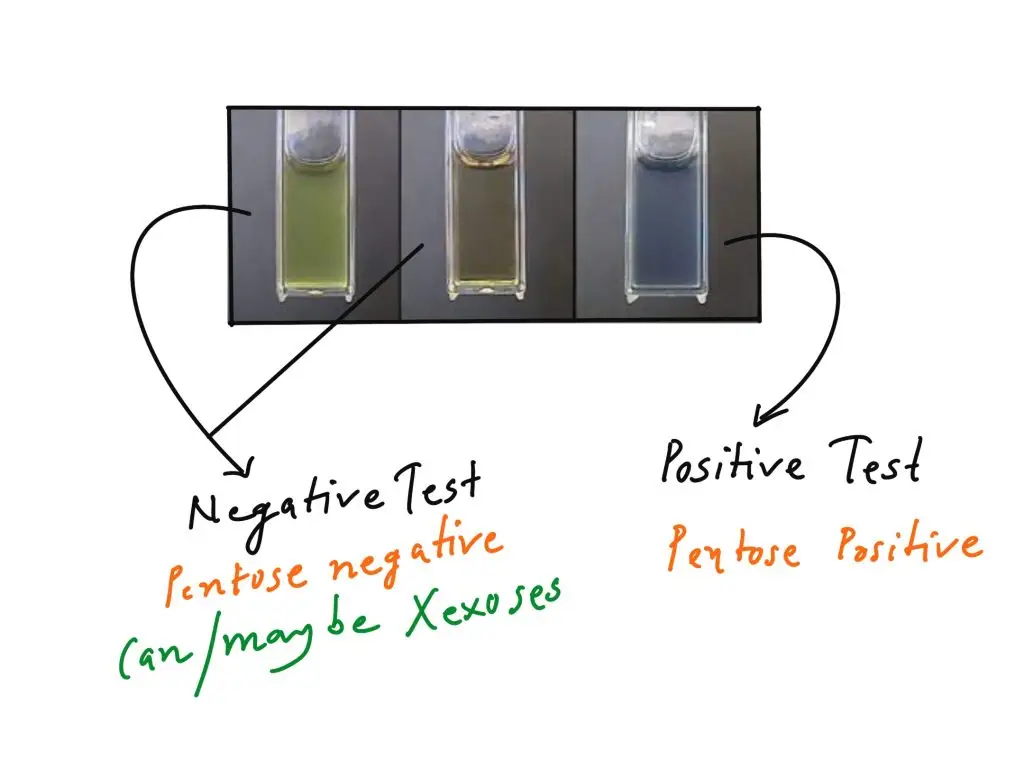
Positive result– Distinct blue or blue-green colour is developed. It indicates presence of pentoses (five-carbon sugars) like ribose or xylose in sample.
Negative result– Muddy-brown yellow or grey colour is developed. It indicates negative for pentoses and it generally signifies presence of hexoses (six-carbon sugars) like glucose or fructose.
Quantitative interpretation– The intensity of blue-green colour is directly proportional to concentration of pentose sugars in solution. For exact quantification the colour intensity is measured by spectrophotometer (generally at 620 nm).
False positive– Prolonged heating should be avoided because some compounds like glucuronates can produce blue-green precipitate leading to false positive interpretation.
Uses of Bial’s Test
Uses of Bial’s Test are-
- It is primarily used to detect pentose sugars (five-carbon sugars) and pentosans and to distinguish them from hexoses and other carbohydrate derivatives.
- It is used for diagnosing pentosuria by detecting pentose sugar in urine and it helps in avoiding misdiagnosis with diabetes mellitus.
- It is used for quantification of RNA (orcinol test) because it reacts with ribose sugar of RNA and it does not react with deoxyribose of DNA.
- It is used for detection of aqueous formaldehyde and it works as a rapid low-cost photometric method for monitoring formaldehyde in industrial wastewater food products and medical stabilizers.
- It is used in food quality control for saccharide profiling (detecting adulteration in fruit juices and honey) and for monitoring sugar consumption during fermentation and for measuring hemicellulose derived additives.
- It is used in pharmaceutical analysis for evaluating pentose sugar concentration in medicines and for verifying carbohydrate composition of vaccines and glycosylated drugs.
- It is used for quantifying glycoproteins and glycolipids by measuring total covalently bound carbohydrates and for monitoring overall glycosylation levels in biopharmaceutical manufacturing.
- It is used in environmental monitoring such as soil science to estimate microbial biomass by measuring RNA derived pentoses in soil extracts and to track environmental pollution and ecosystem health.
Limitations of Bial’s Test
Limitations of Bial’s Test are-
- Narrow specificity– It is mainly designed for pentose sugars and it will not detect other types of carbohydrates.
- False positive from glucuronates– If sample is heated for too long some compounds like glucuronates can break down and blue-green precipitate is formed which looks same as positive pentose result.
- Interference from hexoses– This test depends on timed heating and if boiling is continued more than about 5 minutes high concentration of hexoses can give olive-green or dark grey colour which can be mistaken as weak positive.
- Interference from reducing sugars– Presence of reducing sugars can affect accuracy of test and complementary tests (like Benedict’s test) is required for confirmation.
- Absorbance saturation– At very high carbohydrate concentration the intensity of blue-green colour may not remain directly proportional with amount of sugar present so sample dilution is required for accurate quantitative measurement.
- Reagent sensitivity– Fresh reagent is needed and if orcinol is old or oxidized it can give baseline brown or green tint which masks low concentration of pentoses.
- Sample compatibility– It is not suitable for every carbohydrate solution and some modifications is needed depending on characteristic of sample.
Precautions of Bial’s Test
Precautions of Bial’s Test are-
- Wear personal protective equipment (PPE) like lab coat safety goggles and gloves while handling chemicals.
- Bial’s reagent contains concentrated hydrochloric acid (sometimes sulphuric acid in some protocol) so inhalation and direct skin contact should be avoided.
- Heating step should be done in fume hood or under proper ventilation because boiling can produce corrosive acid fumes.
- Prolonged heating should be avoided. Heating more than about 5 minutes can cause hexoses or glucuronates to react and false positive blue-green colour can be produced.
- Fresh reagent should be used. Orcinol is light sensitive and it can get oxidized so reagent is prepared freshly and it is used within 24 hours.
- Solutions should be mixed properly before and after heating to distribute reaction products evenly and to avoid localized colour pockets.
- Glasswares should be clean and free from contamination. If RNA is quantified then glasswares should be treated properly to remove RNases (e.g. DEPC treated).
- Chemical waste and acids should be disposed properly according to laboratory safety and environmental regulations.

References
- Allen, J., Anderson, A., Nguyen, D., & Sanborn, E. (n.d.). Flintstones vs..
- Asghar, F. S. (2023, December 19). Bial’s test for pentose detection. Scribd..
- Asghar, F. S. (n.d.). Bial’s test for quantifying pentose sugar. Scribd..
- Balmori, C. R. H. (n.d.). Orcinol method for RNA estimation. Scribd..
- BenchChem Technical Support Team. (2025, December). Orcinol | High-purity reagent for research. BenchChem..
- Bhusal, A., Bhandari, S., & Sah, R. P. (2022). Can HPLC be used as an ideal methodology instead of Hb electrophoresis for the diagnosis of hemoglobinopathies in a routine clinical laboratory of under-resource country like Nepal? Is the change necessary?. Annals of Medicine and Surgery, 82, 104367..
- Bial’s test. (2025, August 24). In Wikipedia..
- Claudio, D. G. M. (n.d.). Bial’s test and carbohydrate analysis. Scribd..
- Comprehensive analytical evaluation of Bial’s chromogenic assay: Evolution, chemical mechanism, and multidisciplinary applications in molecular diagnostics. (n.d.)..
- Creative Biolabs. (n.d.). Pentose and hexose: Identification guide..
- Dela Pena, V. (n.d.). Bial’s orcinol test. Scribd..
- Determination of RNA:DNA ratio in bovine liver tissue introduction. (n.d.). SUNY Cortland..
- EssayCompany. (2015, March 23). Qualitative tests used for carbohydrates biology essay..
- Exp.10 assay of carbohydrate (part II) introduction: D. Bial’s test distinguishing pentoses from hexoses. (n.d.)..
- Explain the use of Bial’s reagent in carbohydrate testing. (n.d.). Proprep..
- Faisal, E., Nassr, D., & Farahan, I. (n.d.). Bial`s test)) ((. Al-Mustaqbal University College..
- Hamza, H., Ali, K., & Kadhim, K. (n.d.). Seventh lecture: Carbohydrates tests. Al-Mustaqbal University..
- HiMedia Laboratories. (n.d.). HiPer® RNA estimation teaching kit..
- Khan, S. (2025, August 11). LAB 9 – Tests for carbohydrates. Chemistry LibreTexts..
- King Saud University. (n.d.). Color tests for specific carbohydrates: Ketoses, pentoses, and polysaccharides..
- Mahesha, H. B. (n.d.). Estimation of RNA by orcinol reaction. Yuvaraja’s College, University of Mysore..
- Mechanism for Bial’s test. (2019, February 20). Chemistry Stack Exchange..
- Nooraldeen, H. (n.d.). Bial’s test. Academia.edu..
- Omran, A. P., & Coughlin, C. B. (2020). Bial’s test, a simple method for formaldehyde detection. Biomedical Journal of Scientific & Technical Research, 27(3), 20860-20862..
- OPRL Biosciences Private Limited. (n.d.). Pro TechEx-estimation of RNA by orcinol method teaching kit..
- Sapkota, A. (n.d.). Bial’s test- Definition, principle, procedure, result, uses. Microbe Notes..
- Shaker, M. A. (Ed.). (2019). Practical biochemistry for third class students. Mustansiriyah University College of Science..
- Singh, A., & Askiitians Tutor Team. (n.d.). What is Bial’s test?. askIITians..
- Tests for carbohydrates | Types, principles, apparatus. (n.d.). A Level Biology Revision..