SXT (Sulfamethoxazole-Trimethoprim) test is a microbiological test used to differentiate and presumptively identify different groups of beta hemolytic streptococci and also to determine the susceptibility of bacteria to the antibiotic combination of sulfamethoxazole and trimethoprim. It is commonly performed along with bacitracin disc test for grouping of beta hemolytic streptococci in diagnostic laboratories.
In this test a paper disc impregnated with a fixed ratio of trimethoprim and sulfamethoxazole is placed on agar plate which is already swabbed with the test organism. These two drugs act synergistically and they block successive steps of folic acid synthesis pathway in bacteria. Due to this DNA and protein synthesis is affected and bacterial growth is inhibited.
After incubation the plate is observed for zone of inhibition around the SXT disc. If a clear zone is present then the organism is susceptible. If growth occurs up to the edge of disc then the organism is resistant.
In traditional grouping pattern Group A streptococci are susceptible to bacitracin but resistant to SXT. Group B streptococci are resistant to both bacitracin and SXT. Groups C F and G are resistant to bacitracin but susceptible to SXT. It is also noted that earlier Group A streptococci appeared resistant to SXT due to high thymidine in older media but in low thymidine media S. pyogenes is actually susceptible to SXT.
Objectives of SXT (Sulfamethoxazole-Trimethoprim) Test
- To differentiate different groups of beta hemolytic streptococci (Group A Group B and other groups like C F and G) based on susceptibility or resistance pattern.
- To increase the accuracy of bacitracin susceptibility test by using SXT test along with it for presumptive identification of Group A streptococci.
- It is used as an alternative method for presumptive grouping of streptococci in laboratories where serological grouping facility is not available.
- It is used for selective isolation of Group A and Group B streptococci from mixed cultures by using SXT containing media (SXT blood agar) which inhibits competing commensal bacteria.
- To determine in vitro susceptibility of bacterial isolates to SXT antibiotic combination for guiding clinical treatment decisions.
Principle of SXT (sulfamethoxazole-trimethoprim) Test
SXT test is based on the synergistic bacteriostatic effect of two antifolate drugs sulfamethoxazole and trimethoprim used in a fixed ratio (5:1). Unlike mammalian cells bacteria must synthesize their own folate for formation of precursors like purines thymidine and methionine which are required for DNA and protein synthesis.
This drug combination inhibits folate synthesis by sequential inhibition of enzymes. Sulfamethoxazole competitively inhibits dihydropteroate synthase in the initial step and trimethoprim reversibly inhibits dihydrofolate reductase in the next step. By blocking these two successive steps the bacteria is starved of folate intermediates and replication is stopped.
In laboratory a paper disc containing a precise amount of SXT (23.75 µg sulfamethoxazole and 1.25 µg trimethoprim) is placed on agar plate inoculated with test organism. The antibiotics diffuse in the medium creating a concentration gradient. If organism is susceptible then growth is inhibited and a clear zone of inhibition is formed around the disc. If organism is resistant then growth occurs up to the edge of the disc.
Materials Required for SXT Test
- Culture medium
Mueller-Hinton agar (MHA) plates with low thymidine level. For streptococci MHA is supplemented with 5% defibrinated sheep blood. - Inoculum preparation
Tryptic soy broth (TSB) or Mueller-Hinton broth or sterile saline (for preparing bacterial suspension). - Turbidity standard
0.5 McFarland standard (for adjusting inoculum density about 1×10^8 to 2×10^8 CFU/mL). - Applicators
Sterile cotton swabs (for lawn culture on agar surface). - SXT discs
SXT antibiotic discs containing trimethoprim (1.25 µg) and sulfamethoxazole (23.75 µg). - Supplementary disc (optional)
Bacitracin disc (0.04 units) for differentiation of beta hemolytic streptococci. - Disc placing tools
Sterile forceps (may be sterilized by ethanol and flame) or disc dispenser. - Incubation equipment
Incubator at 35°C to 37°C. For streptococci CO2 incubator (5% to 10% CO2) may be used. - Measuring device
Ruler or sliding caliper (to measure zone of inhibition in mm).
Procedure of SXT (sulfamethoxazole-trimethoprim) Test
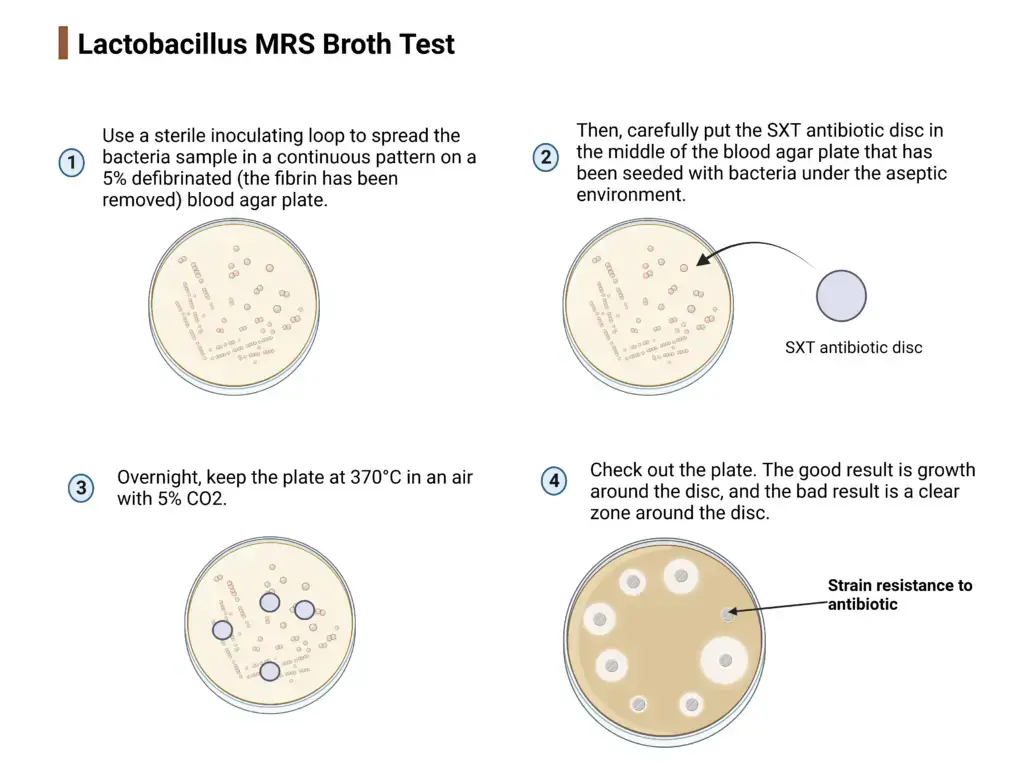
- Select 3 to 5 well isolated colonies from overnight culture and prepare suspension in sterile broth or sterile saline.
- Adjust the turbidity of suspension to 0.5 McFarland standard to obtain standard inoculum density.
- Within 15 minutes dip a sterile cotton swab into the suspension and rotate it against the inner wall of tube to remove excess fluid.
- Inoculate Mueller-Hinton agar plate by swabbing evenly over entire surface. Rotate the plate about 60 degree and repeat swabbing two more times for uniform lawn culture. (For streptococci MHA with 5% sheep blood is used).
- Allow the plate surface to dry at room temperature for about 3 to 15 minutes.
- Place SXT disc on the agar surface using sterile forceps or disc dispenser. The disc contains trimethoprim (1.25 µg) and sulfamethoxazole (23.75 µg). Press the disc gently for proper contact and disc should not be moved after touching agar.
- Within 15 minutes of disc placement invert the plate and incubate at 35°C to 37°C for 18 to 24 hours. For streptococci incubation is done in 5% to 10% CO2.
- After incubation measure the diameter of zone of inhibition in mm using ruler or caliper. Faint haze or slight growth (about 20% or less) is ignored and reading is taken at the obvious margin of complete inhibition.
- Compare the measured zone diameter with standard guideline and interpret as susceptible intermediate or resistant to SXT combination.

Result of SXT Test
- Susceptible
A clear zone of inhibition is formed around the SXT disc. This indicates the organism is susceptible to SXT. The zone diameter should meet the required cut off value (e.g. ≥16 mm or ≥19 mm depending on organism). - Intermediate
A smaller zone of inhibition is present (e.g. 11–15 mm or 16–18 mm depending on organism). This indicates intermediate response and higher dose may be required at infection site. - Resistant
No clear zone is formed and growth occurs up to the edge of the disc. This indicates the organism is resistant to SXT.
Presumptive identification with bacitracin disc
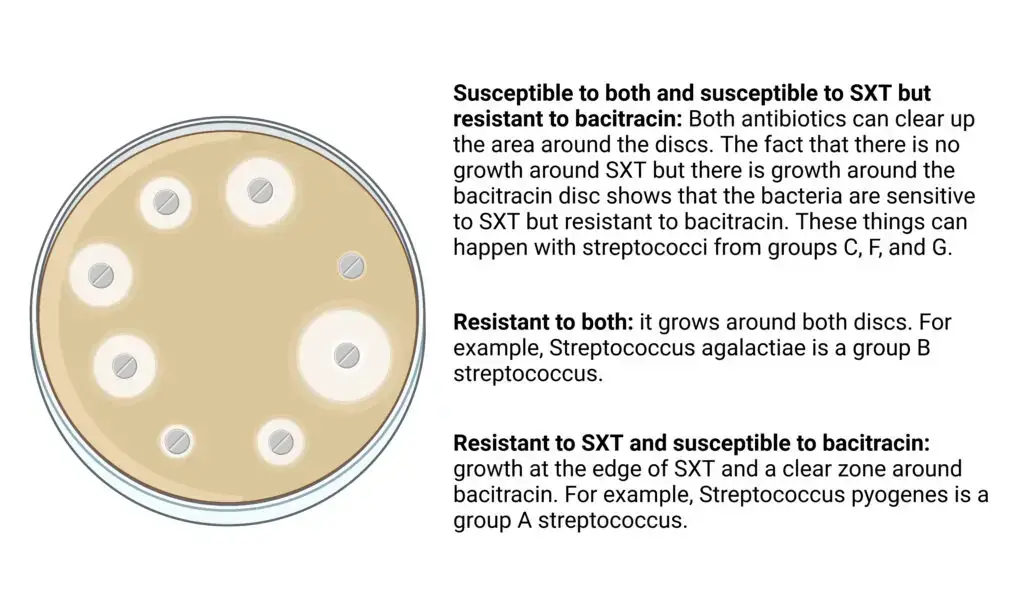
- Group A beta hemolytic streptococci
SXT resistant and bacitracin susceptible (in traditional grouping). - Group B beta hemolytic streptococci
SXT resistant and bacitracin resistant. - Groups C F and G beta hemolytic streptococci
SXT susceptible and bacitracin resistant. - Other non Group A or B streptococci
If susceptible to both SXT and bacitracin then it is presumed not Group A or B and serological test is required.
List of organisms which gives positive result in SXT (Sulfamethoxazole–Trimethoprim) Test
- Group C beta hemolytic streptococci.
- Group F beta hemolytic streptococci.
- Group G beta hemolytic streptococci.
- Group A beta hemolytic streptococci (Streptococcus pyogenes) (in low thymidine media it shows susceptibility).
- Staphylococcus aureus including MRSA.
Quality control strains (susceptible)
- Escherichia coli (ATCC 25922).
- Haemophilus influenzae (ATCC 49247).
- Streptococcus pneumoniae (ATCC 49619).
Other susceptible organisms
- Stenotrophomonas maltophilia.
- Achromobacter xylosoxidans.
- Burkholderia pseudomallei.
- Aeromonas spp.
Quality control of SXT (Sulfamethoxazole–Trimethoprim) Test
- Escherichia coli (ATCC 25922)
- Haemophilus influenzae (ATCC 49247)
- Streptococcus pneumoniae (ATCC 49619)
- Enterococcus faecalis (ATCC 29212 or ATCC 33186) — This organism is specifically used to test new batches of Mueller-Hinton agar to ensure the medium has sufficiently low levels of thymine and thymidine, which can interfere with the test results.
Uses of SXT (Sulfamethoxazole–Trimethoprim) Test
- It is used for presumptive identification and differentiation of beta hemolytic streptococci by performing SXT test along with bacitracin test to classify Group A Group B and other groups like C F and G.
- It is used for selective isolation of Group A and Group B streptococci from mixed clinical specimens by using SXT in primary culture media (SXT blood agar) which inhibits normal flora.
- It is used as a practical and cost effective alternative to serological grouping in laboratories where serologic testing facility is not available.
- It is used to determine in vitro susceptibility of bacterial isolates to SXT antibiotic combination for guiding treatment decisions such as in skin and soft tissue infections.
Advantages of SXT Test
- It increases diagnostic accuracy for presumptive identification and differentiation of Lancefield groups of beta hemolytic streptococci when it is performed along with bacitracin test. It reduces the error of using bacitracin alone because some non Group A strains may also show bacitracin sensitivity.
- It is simple reliable and inexpensive method and it is useful in laboratories where serological grouping facility is not available.
- The drug combination shows synergistic action by blocking successive steps of folate synthesis pathway. This makes the bacteria more sensitive to drug and chances of resistance by single mutation is reduced.
- It helps in selective isolation when SXT is incorporated in primary culture media (SXT blood agar) because it suppresses normal throat flora and allows Group A and Group B streptococci to be isolated from mixed specimen.
- It is used to determine in vitro susceptibility of isolate to SXT combination and it helps clinician in treatment decisions for infections like skin and soft tissue infection where S. pyogenes and MRSA may be present.
Limitations of SXT (Sulfamethoxazole–Trimethoprim) Test
- High level of thymine or thymidine in culture medium can interfere and bacteria can bypass folate pathway inhibition giving false resistant result.
- The test is sensitive to agar properties and Mueller-Hinton agar depth should be about 4 mm. Shallow plates can give false susceptible due to faster diffusion and deep plates can give false resistant due to restricted diffusion.
- Reading of zone may be difficult because faint haze or slight growth (up to about 20% lawn) can occur within zone due to antagonists present in medium.
- For fastidious organisms like streptococci agar is supplemented with 5% sheep blood but blood can cause indistinct zone or thin film growth inside zone for SXT disc.
- It is only presumptive test when used with bacitracin for Lancefield grouping (about 94% accurate) and definitive identification requires serological or molecular tests.
- Selective isolation by SXT blood agar is not perfectly specific and some non Group A or B streptococci (especially Group C and G) may also grow on the medium.
- In vitro susceptibility result does not always guarantee in vivo clinical effectiveness in patient.
Precautions of SXT (Sulfamethoxazole–Trimethoprim) Test
- Mueller-Hinton agar should have low thymine and thymidine level otherwise false resistant result can be obtained. New batch of agar can be tested using Enterococcus faecalis for thymidine level.
- Agar depth should be maintained at about 4 mm. Shallow plates gives false susceptible and deep plates gives false resistant due to diffusion changes.
- Inoculum density should be standardized to 0.5 McFarland. Heavy inoculum can narrow the zone and give false resistant while light inoculum can give larger zone and false susceptible.
- pH of agar should be checked and it should be about 7.2 to 7.4 at room temperature.
- Pure culture should be used and it should be confirmed by Gram staining before test. If distinct colonies are seen within clear zone then purity is questioned and test should be repeated.
- Agar surface moisture should be controlled and plate should be dried for 3 to 15 minutes before applying disc.
- SXT disc should be placed properly and pressed gently for complete contact with agar. Disc should not be moved after touching agar because diffusion starts immediately.
- Discs should be stored properly (about -20°C to +8°C) protected from moisture and direct light to prevent loss of potency which can show false resistance.
- While reading result faint haze or slight growth (20% or less) should be ignored and zone should be measured at the clear margin of complete inhibition.
References
- Bacitracin Susceptibility Test – Principle, Procedure, Uses and Interpretation. (n.d.).
- BACITRACIN DISKS. (2014).
- Becton, Dickinson and Company. (n.d.). Mueller Hinton II Agar Mueller Hinton Agar with 5% Sheep Blood – SECO.
- Bowen, A. C. (2014). The Skin Sore Trial: Exploring a better treatment option for impetigo in Indigenous children living in remote Australia [Doctoral thesis, Charles Darwin University].
- Bowen, A., Lilliebridge, R., Tong, S., Baird, R., Ward, P., McDonald, M., Currie, B., & Carapetis, J. (2012). Is Streptococcus pyogenes Resistant or Susceptible to Trimethoprim-Sulfamethoxazole? Journal of Clinical Microbiology, 50(12), 4067-4072. https://doi.org/10.1128/JCM.02195-12
- Charlie Rose, LLC. (2026). Can Bactrim Treat Streptococcus pyogenes? IDStewardship.
- Clinical and Laboratory Standards Institute (CLSI). (2020). Performance Standards for Antimicrobial Susceptibility Testing (30th ed., CLSI supplement M100).
- Comprehensive Technical Analysis of the Sulfamethoxazole-Trimethoprim (SXT) Susceptibility Test in Diagnostic Microbiology. (n.d.).
- Creative Biolabs. (2026). Disk Diffusion Method for Antibiotic Susceptibility Test.
- European Committee on Antimicrobial Susceptibility Testing (EUCAST). (n.d.). Avläsning av zoner – Exempel och praktiska tips – NICD.
- Gendron, A. (2026). Trimethoprim–Sulfamethoxazole Susceptibility in Beta-Hemolytic Streptococci at a Canadian Tertiary Center (Version 1) [Data set]. Mendeley Data. https://doi.org/10.17632/4c4ymdhp9j.1
- Hardy Diagnostics. (2020). HardyDisk™ Bacitracin Differentiation Disks – for group A Strep Identification – streptococcus pyogenes.
- Hardy Diagnostics. (n.d.). HardyDisks – disk diffusion susceptibility test procedure – Kirby Bauer.
- Julia’s Science Lab. (n.d.). Lancefield Groupings [Video]. YouTube.
- Mueller Hinton Agar – Technical Data. (n.d.).
- Mutual Pharmaceutical Company, Inc. (2012). BACTRIM™ sulfamethoxazole and trimethoprim DS (double strength) tablets and tablets USP.,
- Nippon Becton Dickinson Company, Ltd. (n.d.). BD BBL™ Mueller Hinton II Agar.
- Remel. (2008). SXT Blood Agar.
- Reynolds, J. (2024). 9: Kirby-Bauer (Antibiotic Sensitivity). Biology LibreTexts.,
- SXT Test – Principle, Procedure, Uses and Interpretation. (n.d.).
- Yajko, D. M., Lawrence, J., Nassos, P., & Young, J. (1986). Clinical trial comparing bacitracin with Strep-A-Chek for accuracy and turnaround time in the presumptive identification of Streptococcus pyogenes. Journal of Clinical Microbiology, 24(3), 431–434. https://doi.org/10.1128/jcm.24.3.431-434.1986