What is Coliform Presence/Absence Test?
Coliform Presence/Absence (P-A) test is a rapid simple and inexpensive qualitative method which is used mainly for monitoring treated drinking water and distribution system for fecal indicator bacteria (coliforms). It is the modification of traditional multiple-tube fermentation technique where a single large water sample volume is inoculated in one culture bottle. It is based on public health principle that 100 mL sample of safe drinking water should not contain any coliform bacteria.
In this test a specialized broth is used which contains lactose and pH indicator (bromcresol purple). If coliform bacteria are present in water sample then lactose is fermented during incubation and acid is produced (sometimes gas is also formed). Due to acid production pH of broth is lowered and colour change is observed usually from purple to yellow. This colour change is referred to as presumptive positive result and it is needed further confirmation.
It is advantageous for routine water quality monitoring because minimal technical training is required and larger number of samples are processed quickly. It also helps in maximizing detection of coliform even when background noncoliform bacteria are present in high number and it may interfere other standard test like membrane filtration.
Principle of Presence/Absence Test
Presence/Absence (P-A) test is based on public health principle that no coliforms or other pollution indicator bacteria should be present in 100 mL sample of drinking water. In this test 100 mL water sample is inoculated into a single culture bottle containing a special nutrient broth. The broth is formulated with lactose as fermentable carbohydrate for energy and selective agents like sodium lauryl sulfate is present for inhibiting growth of non-coliform bacteria.
The key feature of this test is inclusion of pH indicator such as bromcresol purple which gives initial purple colour to medium. If coliform bacteria are present in sample then lactose is fermented during incubation and organic acids is produced which lowers pH of broth. Due to acidic reaction indicator dye is changed in colour from purple to yellow and gas may also be produced which is visible as bubbles or foam. Any colour change to yellow is referred to as presumptive positive result and it indicates coliforms are present and further confirmatory test is required.
Objective of Presence/Absence Test
Requirement
Media and Reagents
- Presence-Absence (P-A) broth – It is the primary culture medium and it is purchased in dehydrated powder or sterile concentrated liquid form. It contains nutrients (beef extract, peptones) lactose selective agent (sodium lauryl sulfate) and pH indicator (bromcresol purple).
- Distilled or deionized water – It is used for dissolving and preparing dehydrated P-A broth.
- Sodium thiosulfate – It is used as dechlorinating reagent and it is added in sample container when water sample contains residual chlorine.
- Brilliant Green Lactose Bile (BGLB) broth – It is used in confirmed phase for verifying presumptive positive result from P-A broth.
Instruments and Apparatus
- Culture bottles – Sterile 250 mL screw-cap milk dilution bottle (or equivalent container) is used and it should hold 50 mL prepared triple strength broth with 100 mL water sample.
- Incubator – It is used for maintaining 35 ± 0.5°C for 24 to 48 hours during testing period.
- Autoclave – It is used for sterilizing laboratory prepared P-A broth before use and for decontaminating biohazard waste after test.
- Sample collection containers – Sterile glass or plastic bottles or sterile sampling bags (Whirl-Pak bag) are used for collecting water sample.
- Stirring device – It is used for dissolving dehydrated P-A broth completely in water without using heat.
- Inoculating loop or wooden applicator stick – Sterile 3.0 to 3.5 mm loop or stick is used for transferring culture from positive P-A bottle into BGLB broth for confirmation.
PA Broth Preparation
| Ingredients | Gms / Litre |
| Peptic digest of animal tissue | 5.000 |
| Tryptose | 9.830 |
| Beef extract | 3.000 |
| Lactose | 7.460 |
| Sodium chloride | 2.460 |
| Dipotassium phosphate | 1.350 |
| Monopotassium phosphate | 1.350 |
| Sodium lauryl sulphate | 0.050 |
| Bromo cresol purple | 0.0085 |
Preparation of P-A Broth (3X concentrate)
- It is prepared as 3X concentrate for routine use.
- 91.5 grammes of medium is weighed and it is dissolved in 1 litre deionized water.
- For complete dissolving it is brought to boil while stirring.
- After dissolving it is dispensed into suitable containers.
- Sterilization is done at 121°C for 12 minutes.
Procedure of PA Test
Sample collection
- Sterile glass or plastic container is used (Whirl-Pak® bag or bottle). Sodium thiosulfate is added when residual chlorine is present. If no residual disinfection is present sodium thiosulfate is not required.
- Sample container is opened just before collection and it is closed soon after. Cap is not kept down and container lip or inner surface is not touched. Container is not rinsed before use.
- For potable water sample from faucet spigot hydrant or pump water is allowed to flow at moderate rate for 2 to 3 minutes. Filters and aerators is removed. Leaky or swivelling faucet is not used.
- For non-potable water sample from river lake or reservoir cap is removed while submerged or container mouth is pushed down in water to avoid surface scum. Container is filled completely and mouth is kept in current.
- At least 100 mL sample is collected and about 2.5 cm (1 inch) air space is maintained in container.
- Sample details is written on container and analysis is started immediately.
- If analysis cannot be started sample is maintained at or below 10°C for up to 8 hours. Sample is not freezed.
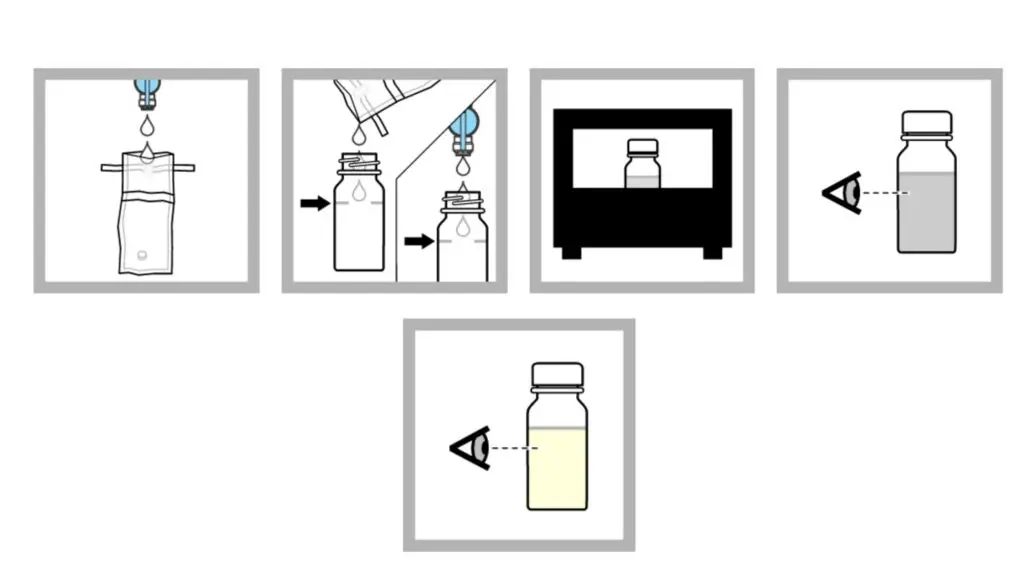
P/A procedure (Method 1 – with bottles)
- 100 mL sample is collected in sterile container and aseptic approach is used to prevent contamination. If sample is disinfected dechlorinating agent containing container is used.
- Sample is added to fill line of P/A bottle. Non-disinfected sample can be added directly from faucet or spout.
- It is incubated at 35 ± 0.5°C for 24 hours.
- After 24 hours colour change is observed. If no colour change is seen incubation is continued for further 24 hours.
- If after 48 hours no colour change is present result is negative.
- If colour change is observed result is presumptively positive.
P/A procedure (Method 2 – with ampules)
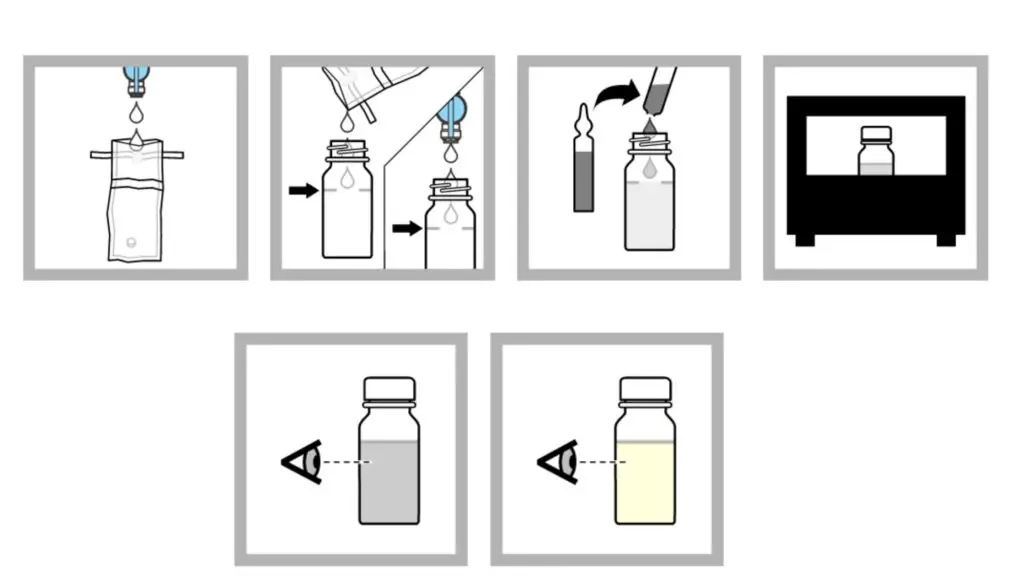
- 100 mL sample is collected in sterile container and aseptic approach is used. If sample is disinfected dechlorinating agent containing container is used.
- Sample is added to fill line of sterile sampling vial. Non-disinfected sample can be added directly from faucet or spout.
- Broth ampule is opened aseptically using ampule breaker and ampule contents is added to bottle.
- It is incubated at 35 ± 0.5°C for 24 hours.
- After 24 hours colour change is observed. If no colour change is seen incubation is continued for further 24 hours.
- If after 48 hours no colour change is present result is negative.
- If colour change is observed result is presumptively positive.
Confirmation procedure
All presumptive positive P/A test results is needed to be confirmed using confirmation medium.
Confirmation media inhibits growth of non-target organisms and it promotes growth of target organisms.
If presumptive medium contains MUG then UV confirmation technique is used.
Method 1 – Confirmation by confirmation medium
- A sterile inoculating loop is used and a portion of culture is transferred from presumptive bottle to confirmation medium.
- The inoculated confirmation medium is incubated for suitable time and temperature.
- After incubation inner tube is observed for gas production and turbidity.
- If gas and turbidity is present it indicates positive result and coliform bacteria is present in sample.
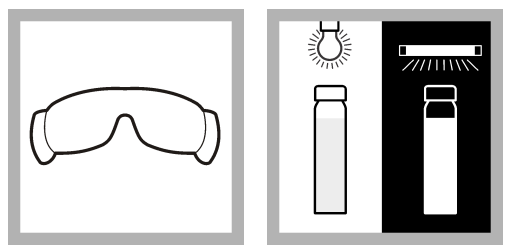
Method 2 – UV confirmation procedure (for MUG medium)
- UV protective glasses is worn.
- Incubated sample containing MUG broth is illuminated with long-wave UV lamp (around 365 nm).
- If fluorescence (glow) is observed then E. coli is confirmed and no further confirmation procedure is required.
- Sample container may show little fluorescence so E. coli Fluorescence Standard is used for comparison and detection.

Result of Coliforms Presence/Absence (PA) Test
- Negative result (Coliforms absent)
- After incubation up to 48 hours the broth remains purple.
- No gas formation or foaming is seen (even after gentle shaking).
- It indicates coliform bacteria are absent in 100 mL water sample.
- Presumptive positive result
- Broth colour is changed from purple to distinct yellow due to acid production (lactose fermentation).
- Gas may be produced and it is seen as bubbles or foaming when bottle is gently shaken.
- This result is referred to as presumptive positive within 24 to 48 hours.
- Confirmed positive result (Total coliforms)
- Presumptive positive culture is transferred to confirmatory medium like Brilliant Green Lactose Bile (BGLB) broth.
- If gas is produced in confirmatory medium within 48 hours then total coliform presence is confirmed.
- Positive for fecal coliforms / E. coli
- Presumptive positive sample is transferred to EC broth and incubated at higher temperature (44.5°C). If gas is produced it indicates fecal coliforms are present.
- If medium contains MUG (EC-MUG) and bright blue fluorescence is seen under long-wave UV light then E. coli is confirmed.
- Invalid result
- Broth becomes turbid (cloudy) but colour remains purple and no gas is produced.
- It may occur due to heavy interference by non-coliform background bacteria so result is taken as invalid.
- New sample is collected and test is repeated.
- Final reporting
- Final result is reported only as “Presence” or “Absence” per 100 mL water sample (total coliforms and/or E. coli).
Uses of Coliform Presence/Absence (PA) Test
- It is used for routine examination of drinking water and public water supplies for safety of consumption.
- It is used for evaluating efficiency of water treatment plant and how effectively water is treated.
- It is used for monitoring distribution system (pipes mains) to detect breach regrowth problem or contamination entry pathway.
- It is used as primary screening test for fecal contamination and fecal indicator bacteria (E. coli and other coliforms) so possible pathogens presence is indicated.
- It is used for testing clean water sources like groundwater well water and chlorinated piped supply where coliforms are expected to be absent.
- It helps in maximizing detection of coliform in sample having high background non-coliform bacteria which may overgrow and interfere other method (membrane filtration).
- It is used in resource limited monitoring because it is simple inexpensive and minimal equipment is required so it is suitable in developing countries and limited setting.
Limitations of Coliform Presence/Absence (PA) Test
- It is a qualitative test and only presence or absence is reported so bacterial count or density is not provided.
- It is a presumptive screening test so positive result (acid and/or gas) is needed confirmatory testing (subculture in Brilliant Green Lactose Bile (BGLB) broth).
- False presumptive positive may occur because some non-coliform organisms (Aeromonas or Clostridium spp.) may ferment lactose or survive selective agents.
- Interference may occur when very high background non-coliform heterotrophic bacteria are present (about 100000 to 1000000 CFU/100 mL) and overgrowth is done so coliform detection is affected.
- It is mainly intended for treated drinking water or distribution system where zero coliform is expected so it is not useful for raw untreated or highly polluted water.
- Weak or borderline reaction may occur (slight brownish-yellow colour with very little gas) so interpretation becomes difficult and further analysis is required.
Advantages of Coliforms Presence/Absence (PA) Test
- It is simple and easy to use because bottle-only format is used and only single 100 mL sample is inoculated in one culture bottle.
- It requires very less technical knowledge so it is suitable for small water system or rural area where laboratory infrastructure is limited.
- It is less labour intensive as compared to quantitative method so larger number of sample are processed in shorter time.
- Sample handling time is less (about few minutes) and it is faster than other procedures.
- It is cost effective method because less laboratory equipment is required and overall equipment cost is low.
- It helps in maximizing detection of coliform in sample having large number of background non-coliform bacteria which may interfere membrane filtration test.
- It allows recovery of injured organisms because nutrient rich liquid medium helps bacteria stressed by chlorine to repair metabolic damage and multiply so sensitivity is high.
- It can be further screened from same bottle for other pollution indicator organisms (Pseudomonas Staphylococcus fecal streptococcus).
Controls for coliform bacteria tests
- Positive control organisms (Expected positive – medium is changed to yellow)
- Escherichia coli (ATCC 25922, ATCC 11775, ATCC 13762) – Good growth is observed and lactose is fermented to produce acid so medium is turned to yellow (gas may or may not be produced).
- Klebsiella aerogenes (formerly Enterobacter aerogenes) (ATCC 13048) – Good to luxuriant growth is observed and medium is turned to light yellow.
- Klebsiella pneumoniae (ATCC 13883) – Good to luxuriant growth is observed and medium is turned to yellow.
- Negative control organisms (Expected negative – medium remains purple / no significant colour change)
- Enterococcus faecalis (ATCC 29212) – Growth is partially or completely inhibited by selective agent so colour change is not significant.
- Pseudomonas aeruginosa (ATCC 27853) – Partial inhibition to moderate growth may occur but lactose is not fermented so medium remains purple.
- Salmonella Typhimurium (ATCC 14028) – Good growth may be seen but it is non-lactose fermenter so negative reaction is produced and medium remains purple.
References
- American Public Health Association, American Water Works Association, & Water Environment Federation. (1992). Standard methods for the examination of water and wastewater (18th ed.). American Public Health Association.
- Becton, Dickinson and Company. (n.d.). Difco™ presence-absence broth.
- Hach Company. (n.d.). Coliforms, presence/absence DOC316.53.01191.
- Hardy Diagnostics. (2020). CRITERION presence-absence broth: Instructions for use.
- HiMedia Laboratories. (2019). Coliform PA broth M1051: Technical data.
- Jacobs, N. J., Zeigler, W. L., Reed, F. C., Stukel, T. A., & Rice, E. W. (1986). Comparison of membrane filter, multiple-fermentation-tube, and presence-absence techniques for detecting total coliforms in small community water systems. Applied and Environmental Microbiology, 51(5), 1007–1012.
- Neogen Corporation. (2010). Presence-absence broth.
- Upadhyaya, S., Mishra, D. B., & Vats, M. K. (2022). The socio biological need of rapid, specific and sensitive tests for detection of coliforms in food and drinking water. International Journal of Creative Research Thoughts, 10(3), 454–466.