pH meter is an instrument used to measure pH of a solution. It measures the hydrogen ion activity in water based solution. It is used to know whether solution is acidic or alkaline.
It works on potentiometry principle. It measures the difference in electrical potential between a measuring electrode and a reference electrode. The measuring electrode is pH sensitive and generally glass electrode is used. The reference electrode remains stable during measurement. The potential difference is produced and then it is amplified by the meter.
The meter converts this signal into a numerical value. The value is shown on the display unit. The pH scale is generally from 0 to 14. pH below 7 is acidic. pH 7 is neutral. pH above 7 is basic or alkaline.
pH meter gives quick reading and more accurate reading. It is used in laboratories for routine pH checking. It is used in agriculture, water treatment and food processing to maintain proper chemical balance.
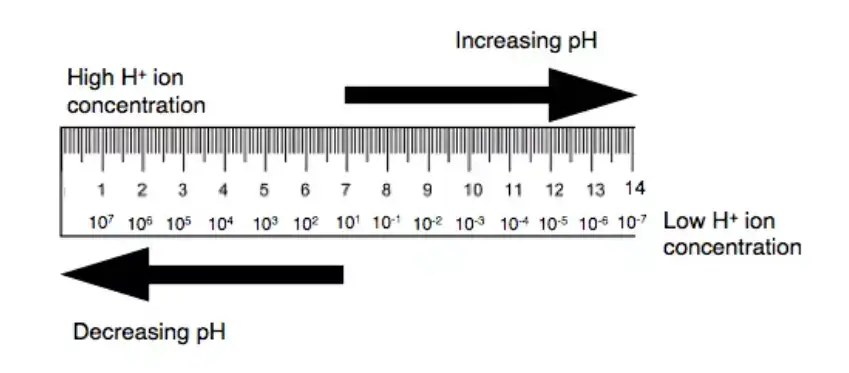
Definition of pH Meter
A pH meter is a scientific instrument used to measure the acidity or alkalinity of aqueous solutions by determining the hydrogen-ion activity, expressed as pH. It operates based on the difference in electrical potential between a pH electrode and a reference electrode.
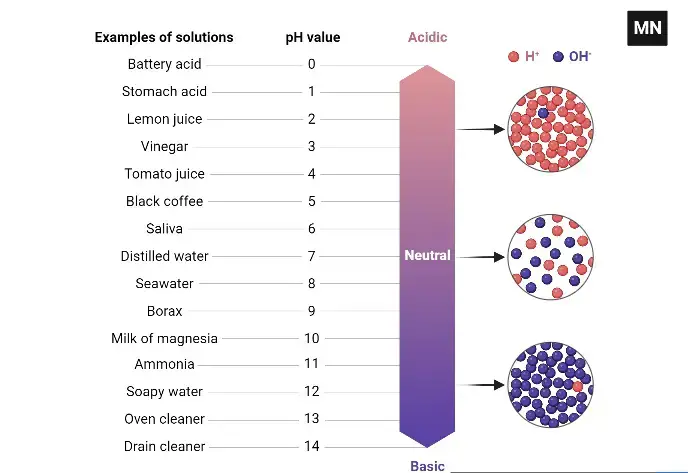
Principle of pH Meter
Principle of pH meter is based on potentiometry. It measures the electrical potential difference between two electrodes. One is measuring electrode (pH sensitive) and other is reference electrode (stable). When both electrodes are dipped in the sample, an electrical circuit is completed.
The measuring electrode is designed to detect hydrogen ion activity. When it contacts the solution, ion exchange occurs between hydrogen ions of sample and hydrated gel layer present on the glass membrane. Due to this, a small voltage is developed across the membrane. The reference electrode remains in a stable electrolyte solution and it maintains constant voltage which acts as baseline.
The meter measures the potential difference between measuring and reference electrode. This relationship follows Nernst equation. The voltage produced is proportional to hydrogen ion concentration of the solution. At 25°C, change of 1 pH unit gives about 59.16 mV change in potential. The weak millivolt signal is amplified inside the meter and it is converted into numerical pH value on the display.
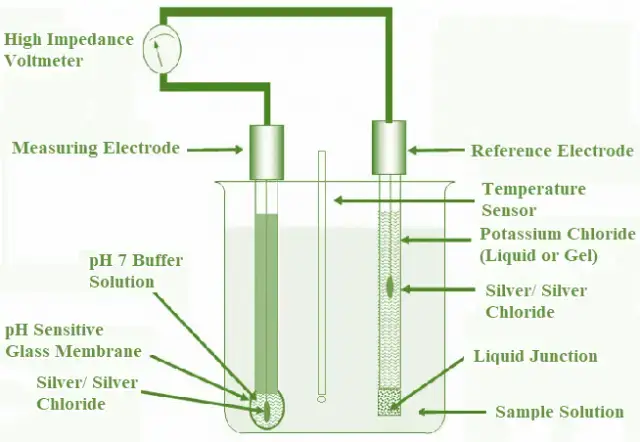
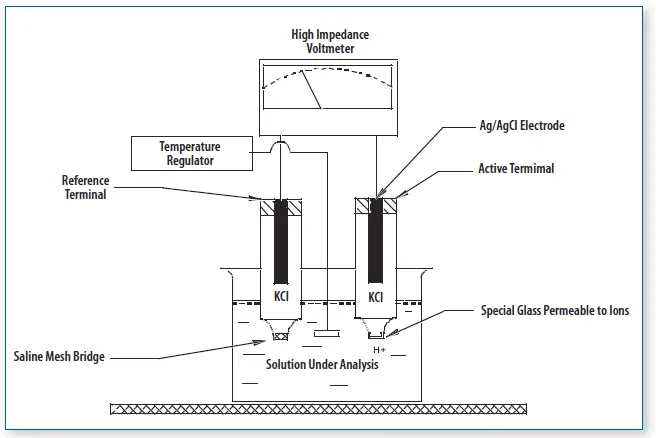
Parts of a pH meter
- Glass electrode (measuring/indicator electrode)- It is the main sensing electrode. It detects hydrogen ion activity of the sample. It has a thin pH sensitive glass bulb. It contains internal reference solution and a silver chloride wire.
- Reference electrode- It gives a constant stable potential. It is used for comparison with measuring electrode. It may be silver/silver chloride type or calomel type. In many meters both are combined as a combination electrode.
- Temperature sensor- It measures the sample temperature. It is used for temperature compensation because pH reading changes with temperature.
- Amplifier circuit- It amplifies the weak millivolt signal produced by the electrodes. The signal is high impedance so amplification is required for proper reading.
- Display unit- It shows the pH value. It may be digital display or analog scale type.
- Calibration system- It is used to standardize the meter. Calibration is done using buffer solutions of known pH before taking sample reading.
pH Meter Operating Procedure
- Calibration– Switch on the pH meter. Calibration is done using standard buffer solutions. If refillable electrode is used, the fill hole is kept open during measurement.
- Rinse electrode- The electrode is rinsed with distilled or deionized water. It is blotted dry using lint free tissue. The glass bulb is not rubbed.
- Sample preparation- The sample solution is taken in a clean beaker. Enough volume is taken for proper dipping.
- Electrode immersion- The electrode is dipped into the sample. The pH bulb and reference junction should remain completely inside the solution.
- Stirring- The sample is stirred gently at uniform rate. It keeps the solution homogeneous during reading.
- Stable reading- The reading is allowed to stabilize. The value is noted only after fluctuation stops.
- Recording- Final pH value is recorded. Temperature of the sample is also noted.
- Cleaning and storage– The electrode is removed and rinsed with distilled water. The electrode is stored in recommended storage solution. It is not stored in distilled or deionized water.
Types of pH Meter
Based on portability
- Pen tester- It is small and portable type. Meter, display and electrode remain in one unit. It is used for quick checking in hydroponics, food work and pool water.
- Handheld meter- It is bigger than pen type. Electrode is separate from meter and can be changed as per need. It is used in agriculture, environmental work and water treatment.
- Benchtop meter- It is large and used on desk or fixed place. It gives higher accuracy as compared to portable type. It is used in laboratories, food processing and monitoring work.
Based on usage
- Laboratory pH meter- It gives wide range and high accuracy. It is used for detailed analysis and quality control work.
- Industrial (online) pH meter- It is used for continuous monitoring. It is fixed with process line and gives real time pH. It can have alarm and control function for industrial use.
Based on advanced level
- Economic pH meter- It gives basic function with low cost. It is used where high precision is not required.
- Intelligent pH meter- It has extra features like automatic temperature compensation and data logging. It is used in water conditioning, aquarium, food processing and laboratory work.
- Precision pH meter- It is used where very high accuracy is needed. It may be pointer type or digital type.
- Pointer pH meter- It uses needle and dial scale. Reading depends on user observation.
- Digital pH meter- It shows pH value in number form. Reading is easier and more accurate.
Based on reading method
- Analog pH meter- It uses needle on scale. It gives reliable reading but careful observation is needed. It is used for basic field work.
- Digital pH meter- It shows value directly on screen. It is easy to read and gives better accuracy. It is used in laboratory and field both.
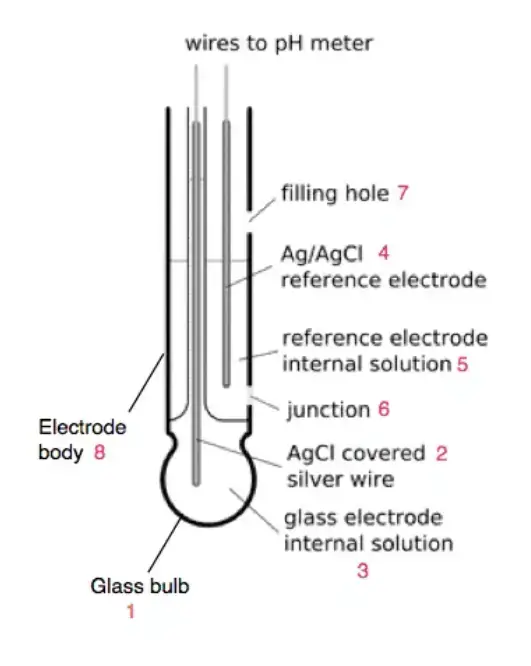
What is pH Electrode?
- pH electrode is the main sensing part of a pH meter. It is used to detect and measure hydrogen ion activity of an aqueous solution. It is used to know acidity or alkalinity of the solution.
- It works like an electrochemical galvanic cell. It converts the chemical property of solution into an electrical millivolt signal. The relationship follows Nernst equation and the signal changes with hydrogen ion concentration.
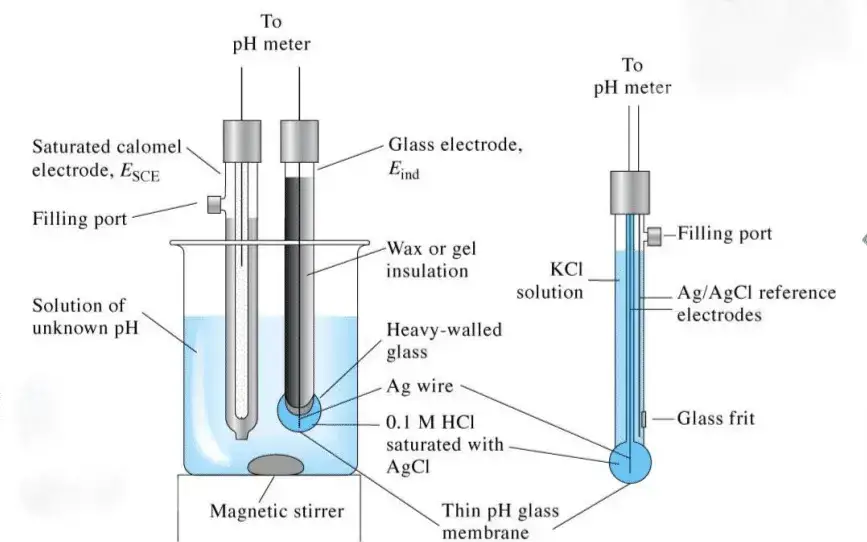
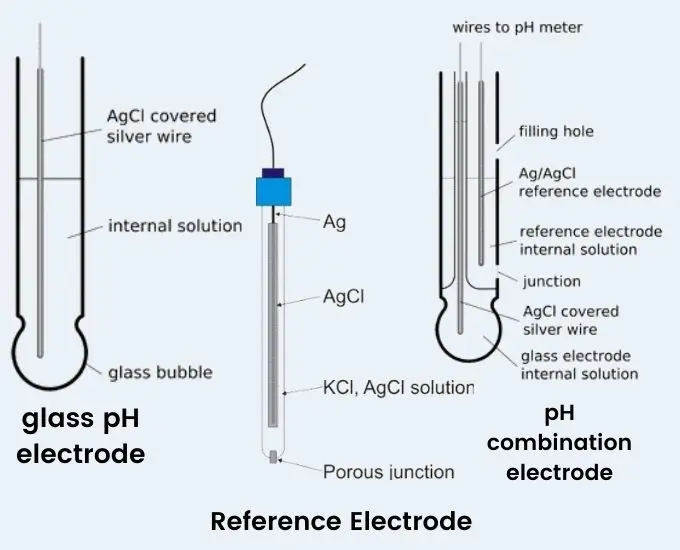
- The measuring (indicator) electrode is generally a glass tube with a thin pH sensitive glass membrane (bulb) at the tip. It contains an internal buffer solution (mostly pH 7) and a silver/silver chloride (Ag/AgCl) wire. It develops a voltage which changes according to the sample pH.
- The reference electrode gives a constant potential as baseline. It has Ag/AgCl wire kept in potassium chloride (KCl) electrolyte solution. It makes contact with sample through a porous junction or diaphragm. In many modern meters, both measuring and reference parts are combined in one unit and it is called combination electrode.
- Some electrodes are non-glass type also. ISFET electrode and dual enamel type electrode are used in some cases. These are used where glass bulb may break and for harsh conditions like food processing or mining.
How pH Electrodes Works?
- Immersion and hydration- When the pH electrode is dipped in the sample, the glass membrane surface contacts water. A thin hydrated gel layer is formed on the outer surface.
- Ion exchange- Hydrogen ions present in the sample exchange with alkali metal ions present in the hydrated gel layer of glass membrane.
- Voltage generation- Due to this ion exchange, charge separation occurs. A small electrical potential is developed across the glass membrane. This potential changes according to hydrogen ion concentration of the sample.
- Reference comparison- A reference electrode gives constant stable potential. In combination electrode, reference part remains in same body and acts as baseline.
- Circuit completion- The liquid junction allows small flow of internal electrolyte from reference side into the sample. This completes the electrical circuit between measuring and reference part.
- Measurement and conversion- The pH meter measures the potential difference between glass electrode and reference electrode. Using calibration and Nernst equation principle, the weak millivolt signal is amplified and converted into readable pH value.
Types of pH Electrodes
- Glass indicator (measuring) electrode- It is the main pH sensing electrode. It is selective for hydrogen ions. It has a thin pH sensitive glass bulb.
- Reference electrode- It gives constant stable potential. It may be silver/silver chloride type or calomel type. It is used as reference for comparison.
- Combination electrode- It contains both glass indicator and reference electrode in one body. It is commonly used in routine work.
- ISFET (non-glass) electrode- It uses semiconductor silicon chip instead of glass bulb. It is durable and not easily breakable. It is used in food processing and rough environment.
- Differential pH electrode- It uses two ion sensitive electrodes instead of normal reference junction. It reduces reference poisoning and fouling problems in industrial samples.
- Antimony electrode- It is a special non-glass type. It is used in rough condition and for solutions containing hydrofluoric acid (HF) where glass membrane dissolves.
- Spear tip (conical) electrode- It has a pointed strong tip. It is used for solid, semi solid or slurry samples like meat, cheese, fruit and agar.
- Flat surface electrode- It has flat membrane. It is used for surface pH measurement of moist surface, thick gels, paste and small volume samples.
- Micro and semi micro electrode- It is very thin and small size electrode. It is used for very small volume sample and narrow space like microtiter plate and NMR tube.
Effect of temperature on pH measurement
- Electrode slope changes- Temperature affects the Nernst slope of pH electrode. When temperature changes, the electrode response changes. Around 0.2 mV per pH unit change occurs for every 1°C change.
- Actual pH of sample changes- Temperature affects ion activity and equilibrium in the solution. On heating, dissociation may increase and hydrogen ion concentration increases. So pH may become lower even if same sample is taken.
- Buffer pH changes- Calibration buffers do not have fixed pH at all temperature. Their pH value changes with temperature. If buffer temperature is different, calibration value also shifts.
- Electrode damage and error- Very high or very low temperature can affect electrode physically. Internal fill solution may boil or freeze. Glass tip may crack. Electrode ageing becomes fast. Asymmetry potential may develop and zero point shifts.
- Need of temperature compensation- Automatic temperature compensation (ATC) is used to correct slope change. But ATC cannot correct actual chemical change in sample pH due to temperature. So pH reading should be noted along with sample temperature.
pH Meter Calibration method
Step 1- Buffer preparation and setup
Fresh buffer solutions are taken in clean beakers. pH 7.00 buffer is taken first. pH 4.00 buffer is taken for acidic range or pH 10.00 (or 9.18) buffer is taken for alkaline range. If refillable electrode is used, the fill hole is kept open.
Step 2- Rinsing of electrode
The electrode is rinsed with distilled or deionized water. It is blotted dry with lint free tissue. The glass bulb is not rubbed or wiped.
Step 3- First point calibration (pH 7.00)
The electrode is dipped in pH 7.00 buffer. Glass bulb and reference junction should remain inside the buffer. The electrode should not touch beaker wall or bottom.
Step 4- Stabilization and setting
The buffer is stirred gently at uniform rate. The reading is allowed to stabilize (generally 1 to 2 minutes). After stable reading, pH 7.00 point is accepted or set on the meter.
Step 5- Rinse again
The electrode is removed from pH 7.00 buffer. It is rinsed again with distilled water. It is blotted dry gently.
Step 6- Second point calibration (slope)
The electrode is dipped in second buffer. pH 4.00 is used for acidic samples. pH 10.00 (or 9.18) is used for alkaline samples.
Step 7- Stabilization and setting
The buffer is stirred gently. The reading is allowed to stabilize. After stable reading, second calibration point is accepted on the meter.
Step 8- Third point calibration (optional)
For better accuracy, third buffer is also used. The electrode is rinsed and then dipped in third buffer and reading is set after stabilization.
Step 9- Check and save calibration
The calibration result is checked on the display. Slope should be around 92% to 102% (or 95% to 103%) and offset should be around -15 mV to +15 mV. Calibration is saved and meter is ready for sample reading.
Application of pH Meter
- Food and beverage industry- It is used to check pH of raw material and product. It is used in brewing and wine making to maintain flavour. It is used in dairy products like cheese and yogurt. It is used in meat processing for safety and shelf life checking.
- Water and wastewater treatment- It is used to monitor drinking water pH. It is used to check municipal and industrial effluent discharge. It is used for environmental compliance and process control.
- Pharmaceuticals and biotechnology- It is used in drug formulation work. It is used for stability testing of product. It is used to control drug release and buffer condition. It is used in cell culture and fermentation process monitoring.
- Agriculture and hydroponics- It is used to test soil pH for fertilizer planning. It helps in improving crop yield. It is used to check pH of nutrient solution in hydroponic system.
- Cosmetics and personal care- It is used to make pH balanced skincare and haircare product. It is used to match body pH and avoid irritation. It is used during formulation and quality checking.
- Chemical and petrochemical manufacturing- It is used to control acid base reaction. It is used to prevent corrosion. It is used in plating, neutralization and other process work.
- Oil and gas extraction- It is used to monitor drilling fluid pH. It is used in gas sweetening process for H2S removal monitoring. It is used for pipeline corrosion control and refinery reaction monitoring.
- Textile industry- It is used in dyeing process for proper dye absorption. It is used in finishing and bleaching process. Proper pH is required for colour fastness and stain removal.
- Automotive industry- It is used to check battery electrolyte condition. It is used to monitor coolant system pH to prevent rust and scale formation.
- Education and research- It is used in school and college laboratory practical. It is used in research and R&D for chemical analysis and experimental study.
Advantages of pH Meter
- Quick reading- It gives fast pH value. It saves time in routine checking.
- Accurate and precise- It gives reliable reading with good accuracy. It is used when exact pH is required.
- Easy to use- Digital display makes reading easy. Result is seen directly in number form.
- Wide use- It is used in food and beverage, pharmaceuticals, agriculture and water treatment. Same meter can be used in many fields.
- Low maintenance and long life- It needs less maintenance in normal use. It works for long time if electrode is handled properly. Some ISFET type electrodes are strong and not easily breakable and can be stored dry.
- Standard and reliable data- It gives dependable result for industrial testing. It supports quality control and standards like ASTM and ISO.
- Automatic temperature compensation (ATC)- Many meters have ATC. It corrects temperature effect on reading and reduces error.
- Continuous monitoring in process- Inline pH meter gives real time reading in process stream. It is used for automatic data collection and quick adjustment without manual sampling.
Limitations of pH Meter
- Glass probe breakage- Glass electrode is fragile. It can break easily during handling. It breaks more when sample has hard particles.
- Short electrode life- Electrode does not last very long. Generally 6 to 12 months life is seen. After that response becomes poor and replacement is needed.
- Contamination problem- Glass membrane and reference junction gets coated or fouled. Chemical poisoning can also occur in some samples. Due to this response becomes slow and reading drifts.
- Alkaline (sodium) error- In very basic solution (above pH 12) glass may respond to sodium ions also. It gives false low pH reading.
- Acid error- In very strong acid (below pH 1) glass membrane absorbs acid molecules. It gives false high pH reading.
- Chemical damage- Some harsh chemicals damage glass electrode. Hydrofluoric acid (HF) can dissolve glass rapidly and electrode becomes useless.
- Non-aqueous limitation- pH concept is mainly for water based solutions. In solvents, alcohols or oils true pH is not obtained. Only apparent reading is obtained.
- Temperature effect- Reading changes with temperature. Meter can compensate slope change but actual pH change inside sample cannot be corrected fully. So error may remain when temperature fluctuates.
- High maintenance- Frequent recalibration is needed with fresh buffers. Regular cleaning is required. Electrode should be stored wet in proper storage solution, otherwise glass dehydrates and reading becomes unstable.
Precautions of pH Meter
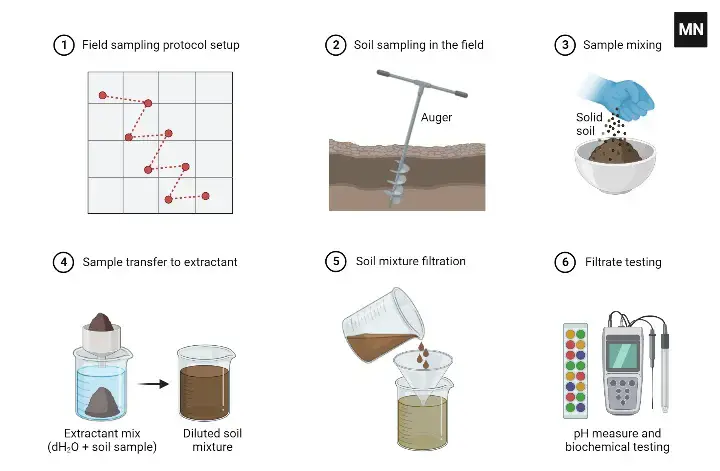
- Glass bulb handling- Glass bulb is delicate. It is not rubbed or wiped with tissue or cloth. It is only blotted gently for drying.
- Do not touch beaker wall or bottom- Electrode should not rest on wall or bottom of beaker. It can scratch or break the tip.
- Proper immersion- Glass bulb and reference junction should be fully dipped in sample or buffer. Half dipping gives wrong reading.
- Rinse between readings- Electrode is rinsed with distilled or deionized water after every reading. It prevents cross contamination of samples and buffers.
- Stir carefully- Sample is stirred gently with uniform speed. Vortex is not formed. Vigorous stirring brings bubbles and CO2 and reading becomes unstable.
- Remove air bubbles- Air bubbles near sensor gives erratic reading. Electrode can be tapped gently or swung down to remove trapped bubbles.
- Keep electrode moist- Electrode is not allowed to dry. Hydrated gel layer is required for ion exchange. Dry electrode gives slow and drifting reading.
- Proper storage solution- Electrode is stored in recommended storage solution (3M or 4M KCl) or suitable buffer. Storage cap is kept filled.
- Not stored in distilled water- Distilled or deionized water is not used for storage. Electrolyte leaches out and electrode becomes sluggish.
- Avoid extreme temperature- Very high or very low temperature is avoided for long time. Internal fill solution may boil or freeze and glass may crack.
- Cleaning precaution- Abrasive cleaning is not done. If needed, soft brush like toothbrush is used with proper cleaning solution. Hard tool damages membrane.
- Fresh buffer for calibration- Calibration is done with fresh standard buffers. Expired buffer gives wrong calibration.
- Do not reuse buffer- Used buffer is not poured back into bottle. Single use buffer is not reused. Contamination occurs fast.
- Match temperature- Buffer and sample should be at same temperature during measurement. Temperature difference gives error in pH reading.
pH Meter Working Animation Video
References
- Advanced Sensor Technologies, Inc. (n.d.). Solid-state pH sensor case studies for mining slurry media.
- Alluvial Soil Lab. (n.d.). Top 7 pH testers for home gardeners: A comprehensive review.
- AlpHa Measurement Solutions. (2023, March 23). Practical pH: Theory and use.
- Atlas Scientific. (2024, March 18). What are the applications of pH in different industries?
- Atlas Scientific. (2025, July 30). How to calibrate a pH meter correctly.
- Barton, K., & Ryan, J. (2026, April 17). The best pH meters for tracking your soil’s health, tested and reviewed. Bob Vila.
- Bryant, S. (n.d.). What is sodium error or Na error in a pH electrode? Camlab.
- Cadence PCB Education. (2025, September 29). Types of pH sensors: Simulation and design. Advanced PCB Design Blog | Cadence.
- Camlab. (n.d.). Meter & electrode selection.
- Campbell Scientific. (n.d.). What are the advantages of using an ISFET chip pH sensor instead of a glass bulb pH sensor?
- Chirdo, T. (2025, November 10). Analog vs. digital sensors: Differences in pH measuring devices? Endress+Hauser.
- Choudhary, A. (2026, April 12). Principle and working of pH meter | pH probes explained. Pharmaguideline.
- Cole-Parmer. (n.d.). How to calibrate a pH meter. Antylia Scientific Blog.
- Cole-Parmer. (n.d.). pH electrode selection guide.
- Comprehensive technical report on potentiometric pH measurement: Electrochemical theory, instrumentation, and industrial applications in 2025. (2025).
- Endress+Hauser. (n.d.). pH measurement – A practical guide to the installation, operation, maintenance and calibration of pH electrodes.
- GAO Tek. (n.d.). Applications of pH meters in oil and gas extraction industry.
- Hamilton Company. (n.d.). Alkaline error. Knowledge Center.
- Hamilton Company. (n.d.). Comparing glass membrane versus optical pH sensors. Knowledge Center.
- Hamilton Company. (n.d.). Just a moment… Knowledge Center.
- Hamilton Company. (n.d.). The Nernst equation. Knowledge Center.
- Hanna Instruments. (n.d.). What is alkaline error?
- Hinotek. (n.d.). How a pH meter works & a guide to pH meter calibration.
- Holme Research Group. (n.d.). pH meter – Calibration. Iowa State University.
- Holme Research Group. (n.d.). pH meter – Instrument. Iowa State University.
- Kremesti, R. E. (n.d.). Glass pH electrode vs ISFET pH electrode.
- Li, C. (2025, October 24). 10 best water quality meters 2025: Home & professional guide. GAIMC.
- Loughnan, L. (2025, October 7). Choosing the right pH meter for your food factory. Klipspringer.
- Malhotra, G. (2025, June 17). What is a pH meter? Learn its principle, types & internal diagram. Presto Group.
- Mason, M. (2017, November 13). What causes damage to pH sensor glass electrodes? Emerson Exchange 365.
- Meier, L. (2024, June 17). How to calibrate a pH meter. Metrohm.
- Pacorr Testing Instruments. (2025, October 6). Complete guide to pH meter: Principle, diagram, and applications.
- Pulse Instruments. (n.d.). ISFET non glass pH technology.
- Royal Brinkman. (n.d.). How to calibrate a pH meter?
- Samp, J. (2025, August 18). Inline vs. laboratory pH measurement: Which is best for industrial processes? Sensorex.
- Schenkel, S., & Kalkman, I. (n.d.). pH-measurement – Monograph. Metrohm.
- Selena. (2023, June 13). What is the difference between digital pH sensor and analog pH sensor? Daruifuno.
- Thermo Fisher Scientific. (n.d.). pH measurement handbook.
- Vernier. (2018, April 6). The theory behind pH measurements.
- Wikipedia contributors. (2026, March 14). Nernst equation. Wikipedia, The Free Encyclopedia.
- Wikipedia contributors. (2026, April 14). pH meter. Wikipedia, The Free Encyclopedia.
- Yokogawa. (n.d.). Understanding differential pH measurement and its benefits.