What is Pauly’s Test?
Pauly’s test is a biochemical colour test which is used for detection of tyrosine and histidine amino acid in proteins. It is a specific colourimetric assay. It was given by Hermann Pauly (German chemist) in 1904.
It is based on azo-coupling reaction. In this process sulfanilic acid is diazotized in cold acidic condition and diazonium salt is formed. This diazonium compound is then added to sample.
In alkaline medium the diazonium compound couples with phenol group of tyrosine or imidazole ring of histidine. Due to this reaction a coloured complex is produced.
Positive test is indicated by formation of dark red or cherry red colour. This confirms the presence of tyrosine or histidine in given sample.
It is also used in clinical applications. It is utilized for detection of histidine rich proteins secreted by malaria parasite Plasmodium falciparum.
Purposes of Pauly’s Test
- It is used for detecting specific amino acids like tyrosine and histidine in protein sample during qualitative analysis.
- It is used for differentiating tyrosine and histidine from other standard amino acids by their specific chemical reaction.
- It is used for diagnosing malaria by detecting histidine rich proteins (PfHRP2 and PfHRP3) secreted by Plasmodium falciparum in human blood sample.
- It is used in food safety for analysing histamine level in fish and seafood products (often with Thin Layer Chromatography–TLC) for spoilage and food poisoning prevention.
- It is used for detecting illegal meat additives like salbutamol in swine meat when combined with advanced techniques like Surface Enhanced Resonance Raman Scattering (SERRS).
- It is used for verifying transgenic plant proteins by confirming expression of genetically engineered proteins containing histidine rich motifs in agricultural biotechnology.
Principle of Pauly’s Test
Principle of Pauly’s test is based on two step chemical reaction. These are diazotization and azo-coupling reaction. It is used for detection of tyrosine and histidine amino acid.
In first step sulfanilic acid is reacted with sodium nitrite and hydrochloric acid in cold condition. Due to this a reactive intermediate is formed which is referred to as diazonium salt (p-phenyldiazosulphonate).
In second step alkaline medium is produced by adding sodium carbonate. This activates phenolic group of tyrosine and imidazole ring of histidine for electrophilic attack. The diazonium salt is then coupled with these activated ring structures.
In this reaction two moles of diazonium compound reacts with each amino acid molecule. Due to azo-coupling a bis-azo dye complex is formed. It gives dark red or cherry coloured solution and indicates positive test.
Reaction of Pauly’s Test
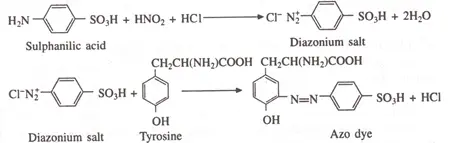
Sulphanilic acid + HNO2 + HCl → Diazonium salt + 2H2O
Diazonium salt + Tyrosine → Azo dye (red color)
Requirements for Pauly’s Test
Reagents
- 1% Sulfanilic acid dissolved in 10% hydrochloric acid (HCl) (it must be kept chilled).
- 5% Sodium nitrite solution in water (it should be pre chilled).
- 10% Sodium carbonate solution in water (it is used as alkalizing agent during coupling phase).
- Chilled sample solution (0.1% or 1% solution of amino acid or protein like tyrosine or histidine).
Materials and Equipment
- Ice bath (it is mandatory for maintaining 0–5°C during preparation because diazonium salt intermediate is unstable at room temperature).
- Test tubes and test tube stand.
- Pipettes for transferring reagent solutions.
- Vortex for gentle mixing of solutions during reaction.
Procedure of Pauly’s Test
- Step 1- Ice bath is prepared and all reagent is chilled properly (1% sulfanilic acid, 5% sodium nitrite and sample solution).
- Step 2- Take a clean test tube and add 1 mL of chilled sulfanilic acid (dissolved in 10% hydrochloric acid).
- Step 3- Add few drops or up to 1 mL of pre chilled sodium nitrite solution in the same test tube.
- Step 4- The mixture is shaken gently or vortex is done and it is kept in ice bath for 1–3 minutes for complete formation of diazonium salt (it is reactive and temperature sensitive).
- Step 5- Immediately add 1–2 mL of chilled amino acid or protein sample to diazonium solution and mix properly (sample should be cold to avoid temperature rise which can spoil intermediate compound).
- Step 6- Add 10% sodium carbonate solution dropwise to make alkaline medium and coupling reaction is initiated.
- Step 7- Observe colour change. Dark red or cherry red colour indicates positive test for presence of tyrosine or histidine.
Result and Interpretation of Pauly’s Test
Positive Result
A dark red or cherry red or deep orange red coloured complex/solution is formed.
Interpretation of Positive Result – It confirms the presence of tyrosine or histidine amino acid in given sample.
Differentiation between Tyrosine and Histidine– Pauly’s test detect both tyrosine and histidine so further observation is required.
- For tyrosine- The cherry red complex changes to yellow colour on dilution. It turns bronze yellow colour when the solution is acidified.
- For histidine- The red colour intensity is decreased and it changes to orange shade on acidification.
Negative Result
Red coloured solution is not formed. Sometimes faint yellow tint can be seen due to reagent.
Interpretation of Negative Result– It indicates absence of both tyrosine and histidine in the sample.
Potential Interferences (False Positive)
Some other molecules having activated aromatic ring or heterocyclic ring can also react and give positive result. Exogenous phenolic compounds and certain drugs and other imidazole containing molecules (like carnosine) can also produce red colour. So chromatographic separation can be required for accurate identification.

Limitations of Pauly’s Test
- Diazonium salt intermediate is unstable so strict cold condition is required. Ice bath is needed and higher temperature causes decomposition of compound.
- Pauly’s reagent should be freshly prepared and it should be protected from light and used immediately. Delay in processing can decrease colour intensity or the test is failed.
- It gives positive result for both tyrosine and histidine so it cannot differentiate them alone. Secondary test (like Millon’s test) can be required for confirmation.
- Cross reactivity can occur and false positive can be obtained. Strong acids or nitrites can affect the test. Other molecules having activated aromatic ring or heterocyclic ring like exogenous phenols (detergents) aromatic drug metabolites and imidazole containing compounds (carnosine) can also give red colour.
- Procedure is complex and multiple steps are involved like diazotization coupling and strict pH and temperature control. So human error chances is increased as compared to simple tests.
- It is less reliable for exact quantitative analysis because colour development is variable and coupling efficiency is inconsistent and reagent is unstable.
Uses of Pauly’s Test
- It is used for detecting specific amino acids. Presence of tyrosine and histidine is identified in different protein samples during biochemical analysis.
- It is used for diagnosing malaria in clinical testing. Histidine rich proteins (PfHRP2) secreted by Plasmodium falciparum is detected in human blood.
- It is used for monitoring seafood safety. Histamine level in fish and fishery products is analysed for freshness and spoilage and prevention of histamine food poisoning.
- It is used for detecting illegal meat additives. It is combined with advanced techniques like Surface Enhanced Resonance Raman Scattering (SERRS) for detecting trace amount of banned substance like salbutamol in swine meat.
- It is used for verifying transgenic plant proteins. Expression of genetically engineered proteins having histidine rich motifs is confirmed (micronutrient biofortification in crops like rice and barley).
- It is used in research for investigating structural biological materials. Natural histidine rich proteins (like proteins for porous underwater mussel glue formation) is studied.
FAQ
What is Pauly’s test?
Pauly’s test is a biochemical test used to detect the presence of tyrosine and histidine-containing proteins. It is named after the German chemist Hermann Pauly who discovered the test.
What is the principle behind Pauly’s test?
Pauly’s test is based on the principle of coupling between the amino acids and the diazonium ion formed in the reagent. The pauly’s reagent consists of sulphanilic acid dissolved in concentrated hydrochloric acid. The sulfanilic acid undergoes diazotization in the presence of sodium nitrate and hydrochloric acid. The result of the diazotization reaction is the diazonium salt (p-phenyldiazosulphonate).
How is Pauly’s test performed?
To perform Pauly’s test, 1 ml of chilled sulfanilic acid is added to a test tube, and a few drops of pre-chilled sodium nitrite are mixed in a vortex. One ml of amino acid sample is immediately added to the test tube and mixed in a vortex. Few drops of sodium carbonate are added to the test tube drop by drop until color begins to appear. A positive result is demonstrated by the appearance of a red-colored complex, indicating the presence of histidine and tyrosine in the solution.
What are the uses of Pauly’s test?
Pauly’s test is used to detect the presence of tyrosine and histidine-containing proteins. The test also allows the differentiation of histidine and tyrosine from other amino acids.
What are the limitations of Pauly’s test?
The formation of diazonium salt occurs at cold temperatures; thus, the test should be performed in the presence of ice. The test doesn’t allow the differentiation between histidine and tyrosine. Millon’s Test can be performed as histidine gives a negative result in Millon’s test.
References
- AMINO ACIDS AND PROTEINS. (n.d.)..
- Color test for proteins and amino acids. (n.d.). Mustansyriah University, College of Medicine..
- Filo. (2025, November 13). Disadvantage of pauly reaction over Milton’s test..
- Qualitative analysis of amino acid. (n.d.). Virtual Labs..
- Sahal, D., Rangiah, K., Sinha, A., & Babbarwal, V. K. (2002). Specific and instantaneous one-step chemodetection of histidine-rich proteins by Pauly’s stain. Analytical Biochemistry, 308(2), 405-408. https://doi.org/10.1016/S0003-2697(02)00210-5.
- Sapkota, A. (2022, September 5). Pauly’s test- Definition, principle, procedure, result, uses. Microbe Notes..
- Supriya, N. (n.d.). Qualitative analysis of amino acids – Definition & methods. Biology Reader..
- The analytical evolution of Pauly’s test: A comprehensive study of diazotization, azo-coupling, and the histochemical detection of tyrosine and histidine. (n.d.)..
- Wikipedia contributors. (2022, February 3). Pauly reaction. In Wikipedia, The Free Encyclopedia..
- Wikipedia contributors. (2026, March 1). Hermann Pauly. In Wikipedia, The Free Encyclopedia..
- Yu, S., Liu, Z., Zhang, J., Li, H., Xu, N., Yuan, X., & Wu, Y. (2018). An azo-coupling reaction-based surface enhanced resonance Raman scattering approach for ultrasensitive detection of salbutamol. RSC Advances, 8(10), 5536–5541. https://doi.org/10.1039/c7ra12927k.