Nitrite Reduction Test is a biochemical test, which is used to determine that bacteria can reduce nitrite (NO2−) into other nitrogenous compounds. The nitrite is further reduced to nitric oxide (NO), nitrous oxide (N2O), nitrogen gas (N2) or ammonia (NH3). This test mainly indicates the ability of organism to remove nitrite from medium.
In this test bacteria are heavily inoculated into nitrite broth. It is a liquid medium which contains potassium nitrite (KNO2) as the substrate. The inoculated broth is incubated for a specific time period so that nitrite reduction can occurs.
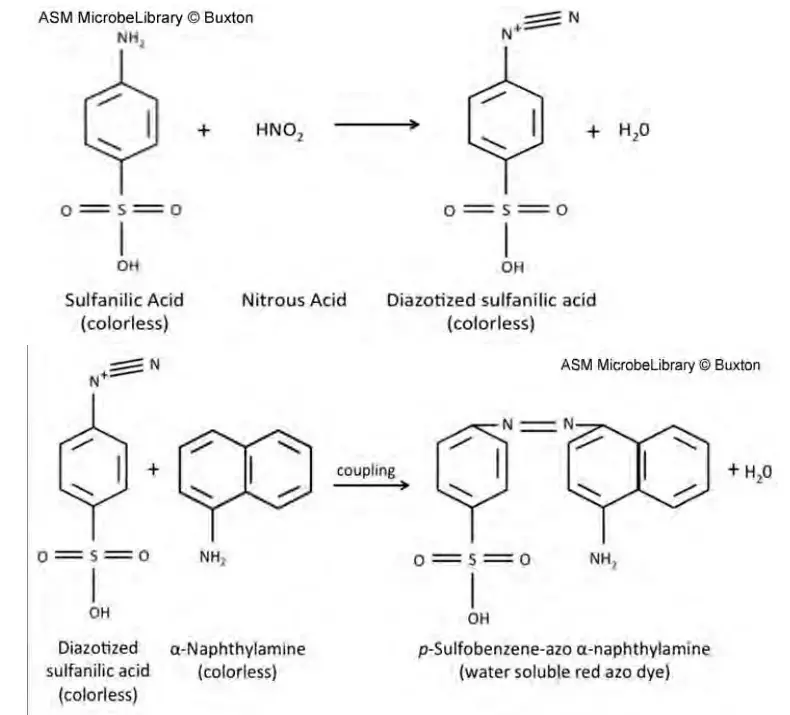
After incubation two chemical detectors are added. These are Reagent A (sulfanilic acid) and Reagent B (alpha-naphthylamine or N,N-dimethyl-alpha-naphthylamine). The reaction is based on presence or absence of remaining nitrite in the tube.
If red or pink colour is developed after adding reagents then test is negative. It is due to the reagents reacting with unreduced nitrite which is still present in the broth and a red azo dye is formed.
If no colour is formed and medium remains colourless then test is positive. It indicates that nitrite has been reduced completely by organism and no nitrite is left to react with reagents.
In many cases an inverted Durham tube is placed inside the broth. It is used to trap displaced air. Gas bubbles formation in Durham tube gives additional confirmation that nitrite is reduced to nitrogen gas.
Objective of Nitrite Reduction Test
- It is used to determine that organism produces nitrate reductase enzyme and nitrate (NO3−) is reduced to nitrite (NO2−).
- It is used to identify that organism can further reduce nitrite into other end products like nitrogen gas (N2), nitric oxide (NO), nitrous oxide (N2O) or ammonia (NH3).
- It is used as biochemical marker for differentiation and identification of bacterial species.
- It is useful for identifying members of Enterobacteriaceae family.
- It is used for separating Neisseria species from Moraxella and Kingella species (example– nitrate negative N. gonorrhoeae and nitrate positive K. denitrificans).
- It is used for classification and differentiation of Mycobacterium and Corynebacterium species.
- It is used to determine that bacteria uses nitrate as final electron acceptor during anaerobic respiration.
- It is used to verify that bacteria possess gene which codes nitrate reductase enzyme.
Principle of Nitrite Reduction Test
It is based on enzymatic reduction of nitrate and nitrite by bacteria. In anaerobic respiration the enzyme nitrate reductase is produced by some organism and nitrate (NO3−) is reduced to nitrite (NO2−). In some bacteria this nitrite is further reduced into nitric oxide (NO), nitrous oxide (N2O), nitrogen gas (N2) or ammonia (NH3).
The main principle is detection of nitrite ion in the culture medium. The presence or absence of nitrite is confirmed by adding chemical reagents after incubation. This is referred to as Griess reaction.
After incubation Reagent A (sulfanilic acid) and Reagent B (α-naphthylamine or related derivative) is added in the broth. If nitrite is present it reacts with sulfanilic acid and diazonium salt is formed and then it couples with Reagent B. A red water soluble azo dye (prontosil) is formed. Red colour indicates that nitrate has been reduced to nitrite.
If no red colour is developed then it indicates nitrite is absent in the medium. It may be due to nitrate is not reduced by organism or nitrate is reduced to nitrite and then nitrite is completely reduced into gases or ammonia.
To differentiate this colourless result zinc powder is added. Zinc acts as chemical catalyst and it reduces any unreduced nitrate into nitrite rapidly. If red colour is developed after zinc addition then test is negative because nitrate was present and it was not utilized by bacteria.
If broth remains colourless after adding zinc then it indicates that no nitrate is left for zinc reaction. It shows that bacteria has completely reduced nitrate and nitrite into other nitrogenous compounds.
A small inverted Durham tube is also placed in broth in many cases. It is used for trapping the nitrogen gas. Gas bubbles formation provides visual evidence that complete nitrite reduction (denitrification) is occurred.
Requirement for Nitrite Reduction Test
Media-
- Nitrite reduction medium (nitrite broth) is required.
- It is a liquid broth which contains potassium nitrite (KNO2) as substrate.
- The composition is as follows- beef (meat) extract (3.0 g), gelatin peptone (5.0 g), potassium nitrite (1.0 g) and deionized water (1000 ml).
Reagents-
- Griess reagents are required for detection of nitrite.
- Reagent A is required (0.6 ml of N,N-Dimethyl-α-naphthylamine in 100 ml of 5N acetic acid).
- Reagent B is required (0.8 g of sulfanilic acid in 100 ml of 5N acetic acid).
- Zinc dust is not required in nitrite reduction test because it is used for nitrate reduction.
Materials and Equipment-
- Test tubes are required for holding nitrite broth during incubation.
- Small test tubes (12 mm × 75 mm) can also be used for mixing small aliquots of cultured broth with reagents.
- Durham tubes are required and it is placed inverted inside the broth for trapping nitrogenous gas.
- Sterile inoculating loop or swab is required for heavy inoculation of bacterial isolate.
- Pasteur pipette or dropper is required for adding chemical reagents in culture.
- Incubator is required for incubation (generally 35°C for 12 to 48 hours).
- Standard microbiological equipment are required like incinerator or Bunsen burner for sterilization of inoculating loop.
Composition of Nitrite reduction medium
- Beef (meat) extract – 3.0 g
- Gelatin peptone – 5.0 g
- Potassium nitrite (KNO2) – 1.0 g
- Deionized water – 1000 ml
Procedure of Nitrite Reduction Test
- A tube of nitrite reduction medium is taken. It is a broth containing potassium nitrite.
- The test organism is taken from well isolated colonies and medium is heavily inoculated by sterile loop.
- The inoculated tube is incubated at 35°C for 12 to 24 hours.
- In rare cases prolonged incubation upto 5 days is required.
- After incubation the broth is observed for gas production.
- The surface of broth and inverted Durham tube is examined for gas bubbles.
- In a separate small test tube (12 mm × 75 mm) the reagents are prepared.
- Two drops of Reagent A (sulfanilic acid) and two drops of Reagent B (α-naphthylamine or dimethyl derivative) is mixed.
- About 1 ml of incubated broth culture is added into the small test tube containing mixed reagents.
- It is shaken well and colour change is observed.
- If no colour is developed and mixture remains colourless then test is positive. It indicates that nitrite substrate is reduced completely into other nitrogenous products. Gas in Durham tube also supports this result.
- If red or pink colour is developed then test is negative. It indicates that unreduced nitrite is still present and organism is unable to reduce it.
- Zinc dust is not added in nitrite reduction test. It is not required unlike nitrate reduction test.
Result of Nitrite Reduction Test
Positive result (colourless)-
When Reagent A (sulfanilic acid) and Reagent B (α-naphthylamine or its derivative) is added the broth remains colourless. It indicates that nitrite present in medium has been completely reduced by bacteria into other nitrogenous products.
Positive result for denitrification (gas production)-
Gas bubbles are seen in inverted Durham tube or on the surface of broth. It gives confirmation that nitrite is reduced into nitrogen gas. Absence of gas does not rule out positive result because other end products can also be formed.
Negative result (red colour)-
When reagents are added the broth turns red. It indicates that original nitrite is still present in tube and it is not reduced by organism.
Partial reduction (light pink colour)-
If light or faint pink colour is developed then only some nitrite is reduced and some amount remains in the tube. In this case the culture can be re incubated and test can be done again next day.
No zinc required-
Zinc dust is not added in nitrite reduction test. It is not required for evaluation of nitrite broth reaction.

Quality Control of Nitrite Reduction Test
- Positive control for nitrite production (partial reduction)– Escherichia coli (ATCC 25922), Enterobacter aerogenes (ATCC 13048) or Salmonella typhimurium (ATCC 14028) are used. These organism confirms reduction of nitrate to nitrite. After adding Reagent A and Reagent B deep red colour should be developed immediately.
- Positive control for denitrification (complete reduction)– Pseudomonas aeruginosa (ATCC 27853) is used. The medium should remain colourless after adding Reagent A and Reagent B. It should remain colourless after adding zinc dust and gas bubbles should be seen in Durham tube.
- Negative control– Acinetobacter baumannii (ATCC 19606) or Acinetobacter calcoaceticus (ATCC 19606) are used. The broth remains colourless after adding Reagent A and Reagent B but red colour is developed after adding zinc dust.
- Fastidious organism controls– Kingella denitrificans (CDC 10,236) is used for positive result and it turns red after adding Reagent A and Reagent B. Neisseria gonorrhoeae (ATCC 43069) is used for negative result and it turns red only after zinc is added.
- Uninoculated control (blank)– An uninoculated tube of nitrate broth is tested along with cultures. It confirms sterility of medium. It also proves zinc powder works properly and it ensures there is no pre existing nitrite contamination in water broth or glassware. The uninoculated tube should not change colour after adding Reagent A and Reagent B but it must turn pink or red after adding zinc dust.
Limitations of Nitrite Reduction Test
- It is only an aid for phenotypic identification and it is not confirmatory alone. Supplementary biochemical, morphological and serological tests are required for complete identification.
- It is a culture based method so time is required. The result can take 24 hours to 5 days for reporting negative result. If test is done early then enough reduction may not be detected and if test is done late then all nitrite can be further reduced or assimilated.
- Growth of organism in broth is not easily noticed so visual limitation is present.
- The colour reaction can fade rapidly so interpretation should be done immediately after adding reagents. Sometimes non specific faint pink colour can be seen and it should be differentiated from deep red colour.
- The media should be completely free from nitrite. Trace nitrite contamination in dehydrated media distilled water or glassware can give false positive result.
- Air bubbles can get trapped in Durham tube during autoclaving and it can be mistaken as nitrogen gas. It may lead to false positive interpretation for denitrification.
- Vigorous bacterial growth is required. Inadequate growth produces low enzyme and it remains below detection limit of reagents. High oxygen tension can repress synthesis of nitrate reductase enzyme and it may give false negative in aerobic cultures.
- It is required to know that organism is glucose fermenter with gas production before test. Gas bubbles in Durham tube can create confusion in interpretation.
- Zinc dust verification is required in some cases to prevent false negative when nitrite is further reduced. Excess zinc dust can give false result because nitrate may be reduced rapidly to ammonia and nitrite stage is bypassed.
- Griess reagents are sensitive to light and temperature. Reagent B can darken and become turbid with time and it can obscure the positive colour.
- Some strong reducing organism can produce brown precipitate immediately after reagents are added and atypical reaction is obtained.
Uses of Nitrite Reduction Test
- It is used for differentiating Gram negative bacilli. It is used in biochemical test battery for separating Enterobacteriaceae family from other Gram negative bacilli.
- It is used for distinguishing specific genera. It helps in identification of Neisseria and it separates it from Moraxella and Kingella species.
- It is used for identification of Gram positive bacteria. It is useful for species level identification of Corynebacterium and other asporogenous Gram positive bacilli.
- It is used for determining metabolic capability of organism. It evaluates that organism can use nitrite as electron acceptor in anaerobic respiration and it reduces it into nitrogen gas, nitric oxide, nitrous oxide or ammonia.
- It is used in study of environmental nitrogen cycling. It is used for identifying bacteria involved in denitrification and dissimilatory reduction. It also indicates nitrogen loss in agricultural soils and it is useful for removing excess nitrogen in wastewater treatment plants.
- It is used for assessing food and consumer safety. It helps to detect nitrate and nitrite reducing bacteria in processed meats foods and beverages. The reduction products can react with amines and carcinogenic N-nitrosamines is formed.
References
- Almeida, S., Sousa, C., Abreu, V., Diniz, C., Dorneles, E. M. S., Lage, A. P., Barh, D., & Azevedo, V. (2017). Exploration of nitrate reductase metabolic pathway in Corynebacterium pseudotuberculosis. International Journal of Genomics, 2017, 9481756. https://doi.org/10.1155/2017/9481756
- Aryal, S. (2022, August 10). Nitrate reduction test – Principle, procedure, uses and interpretation. Microbiology Info.
- Biolab Diagnostics Laboratory Inc. (2016, October 11). Nitrate broth technical sheet.
- Biotium. (2020, April 1). Griess reagent kit – Product information.
- Buxton, R. (2011, November 1). Nitrate and nitrite reduction test protocols. American Society for Microbiology.
- Centers for Disease Control and Prevention. (2013, December 10). Nitrate reduction test – Gonorrhea.
- Central Drug House (P) Ltd. (n.d.). Technical information – Nitrate broth Product Code: DM 1439S.
- Comprehensive analysis of the nitrate and nitrite reduction tests: Biochemical mechanisms, clinical diagnostic applications, and enzymatic regulation in prokaryotic nitrogen metabolism. (n.d.).
- Cooper, C. R., Jr. (2019). The nitrate reduction test. Youngstown State University.
- Dahal, P. (2024, February 18). Nitrate reduction test- Principle, procedure, types, results, uses. Microbe Notes.
- Das, A., Sanakal, S. I., Sivakumar, G., Babu, A., & Maji, S. (2025). Development of a colorimetric sensor utilizing itaconic acid-mediated Griess reaction for sensitive detection of nitrite and nitrate in agricultural products. Materials Advances, 6, 5045-5055. https://doi.org/10.1039/D5MA00314H
- Difco. (n.d.). Nitrate broth. Grosseron.
- GMP Plastic. (2025, March 24). Nitrate reduction test: Principle, procedure, and interpretation.
- Hardy Diagnostics. (2020). CRITERION nitrate broth instructions for use.
- Hardy Diagnostics. (2020). Nitrate reagent instructions for use.
- HiMedia Laboratories. (2015). Nitrate broth M439 technical data.
- Interlab. (n.d.). Nitrate broth – Manual Difco.
- Lassiter, D. V. (1973). Hazard review of alpha-naphthylamine (1-NA) (1-naphthylamine) (PB87210878). National Institute for Occupational Safety and Health. https://stacks.cdc.gov/view/cdc/179675
- Micro Master Laboratories Private Limited. (n.d.). Product specification sheet: Nitrate Reagent-II / α-Naphthylamine solution (IR005).
- Moreno-Vivián, C., Cabello, P., Martínez-Luque, M., Blasco, R., & Castillo, F. (1999). Prokaryotic nitrate reduction: Molecular properties and functional distinction among bacterial nitrate reductases. Journal of Bacteriology, 181(21), 6573–6584. https://doi.org/10.1128/jb.181.21.6573-6584.1999
- Purchase, I. F., Kalinowski, A. E., Ishmael, J., Wilson, J., Gore, C. W., & Chart, I. S. (1981). Lifetime carcinogenicity study of 1- and 2-naphthylamine in dogs. British Journal of Cancer, 44(6), 892–901. https://doi.org/10.1038/bjc.1981.289
- Remel. (2009, June 24). Nitrate reagent B. ThermoFisher.
- SciSpace. (n.d.). Microbial nitrate reductase: Classification and determination.
- Sigma-Aldrich. (n.d.). 38497 Nitrate reagent A (1-naphthylamine solution).
- Sigma-Aldrich. (n.d.). 72548 Nitrate broth.
- Sigma-Aldrich. (n.d.). 73426 Nitrate reduction test.
- Sparacino-Watkins, C., Stolz, J. F., & Basu, P. (2014). Nitrate and periplasmic nitrate reductases. Chemical Society Reviews, 43(2), 676–706. https://doi.org/10.1039/c3cs60249d
- Sun, J., Zhang, X., Broderick, M., & Fein, H. (2003). Measurement of nitric oxide production in biological systems by using Griess reaction assay. Sensors, 3(8), 276-284. https://doi.org/10.3390/s30800276
- Thermo Fisher Scientific. (n.d.). BD Difco nitrate broth 500 g. Fisher Scientific.
- Van Gray, J. (2025, March 20). 5.6: Nitrate reduction. In Microbiology Lab SP25. Biology LibreTexts.
- Varlamova, A. A., Gyulkhandanyan, E. M., Nikolsky, V. M., & Gridchin, S. N. (2017). Nitrate test paper (Patent No. RU169694U1).
- Werkman, C. H. (1930). Dimethyl-alpha-naphthylamine for the determination of bacterial reduction of nitrates. Proceedings of the Iowa Academy of Science, 37(1), 53-55. https://scholarworks.uni.edu/pias/vol37/iss1/7
- Wikipedia contributors. (2024, December 30). Nitrate reductase test. In Wikipedia, The Free Encyclopedia.
- Wikipedia contributors. (2025, April 10). Griess test. In Wikipedia, The Free Encyclopedia.