Millon’s Test is an analytical biochemical test used to detect soluble proteins by detecting tyrosine. Tyrosine is an amino acid that contains phenol functional group and it is mainly responsible for giving positive reaction in this test.
This test is carried out using Millon’s reagent. Millon’s reagent is a strongly acidic solution of mercuric nitrate and mercurous nitrate dissolved in concentrated nitric acid and water.
When Millon’s reagent is added to protein sample and heated the phenol group of tyrosine is nitrated by nitric acid. Then the nitrated tyrosine combines with mercury ions and forms a complex.
A positive Millon’s test is indicated by formation of pink red or brick-red coloured precipitate or solution. If no red colour or precipitate is formed then test is negative showing absence of tyrosine or tyrosine containing proteins.
This test is not completely specific for proteins because any compound containing phenolic group (phenol salicylic acid etc.) can also give similar colour. So it is confirmed along with other protein tests like Biuret test or Ninhydrin test.
Objectives of Millon’s Test
Objectives of Millon’s Test
- To detect the presence of tyrosine containing proteins in the given sample.
- To detect phenol containing compounds by observing colour formation.
- To differentiate tyrosine from other amino acids.
Principle of Millon’s Test
Millon’s test is an analytical test used to detect tyrosine. Tyrosine is the only amino acid that contains a phenol group. Hence proteins containing tyrosine can also give positive result.
The principle of this test is based on reaction of phenolic group of tyrosine with Millon’s reagent. Millon’s reagent is a mixture of mercurous nitrate and mercuric nitrate dissolved in concentrated nitric acid.
In first step the phenol group of tyrosine is nitrated by nitric acid present in reagent. In next step this nitrated tyrosine combines with mercury ions and a coordination complex is formed.
The formation of this complex produces a pink to brick-red coloured precipitate or solution. This colour indicates the presence of tyrosine (or protein containing tyrosine) in the given sample.
Requirements for Millon’s Test
Chemicals and Reagents
- Millon’s reagent (mixture of mercuric nitrate and mercurous nitrate dissolved in concentrated nitric acid and distilled water).
- Test sample (1% tyrosine solution egg albumin or any protein/food sample solution).
- Distilled water (negative control).
- Sodium nitrite (1% solution) (required only for modified Millon’s test).
- Modified Millon’s reagent (Cole’s modification) (mercuric sulphate dissolved in sulphuric acid) (if modified test is performed).
Materials and Equipment
- Clean dry test tubes.
- Test tube stand.
- Test tube holder.
- Pipette or dropper.
- Heat source (boiling water bath or alcohol lamp).
Procedure of Millon’s Test
- Take a clean dry test tube.
- Add 1 to 2 ml of the test sample in the test tube (or take 1% tyrosine solution as positive control).
- Add Millon’s reagent in the test tube (few drops to 2 ml depending on laboratory protocol).
- Mix the contents properly by gently shaking or swirling.
- If red colour or precipitate is not formed immediately then keep the test tube in boiling water bath.
- Heat for about 2 to 10 minutes.
- Observe the colour change. Formation of pink red or brick-red coloured precipitate or solution indicates positive test for tyrosine.
Result and Interpretation of Millon’s Test
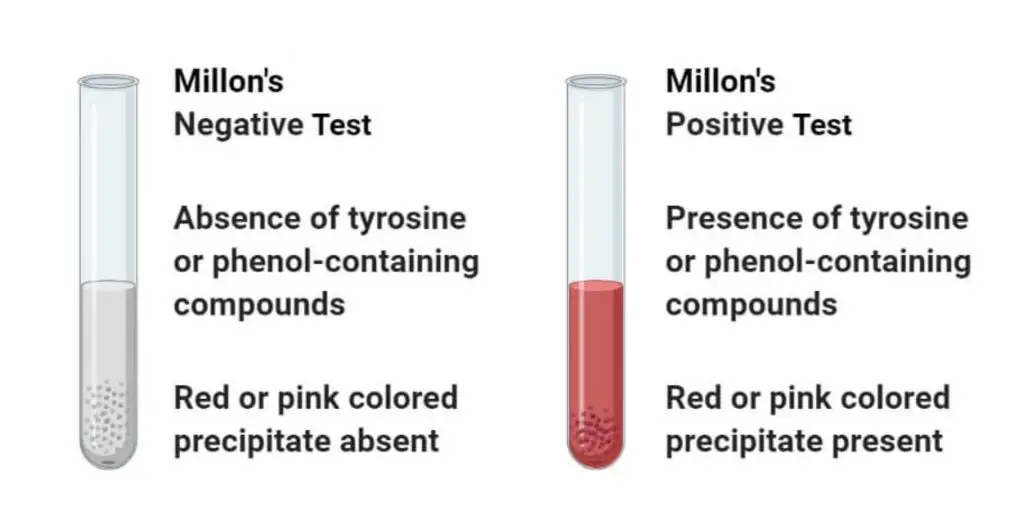
Positive result
Light pink red or brick-red coloured precipitate or solution is formed. This indicates presence of tyrosine or tyrosine containing proteins (or other phenolic compounds) in the given sample.
Negative result
No pink or red coloured precipitate is formed and solution remains colourless (or shows colour other than red). This indicates absence of tyrosine or tyrosine containing proteins (or phenolic compounds) in the given sample.
Uses of Millon’s Test
- It is used to detect tyrosine in the given sample.
- It is used to detect proteins containing tyrosine.
- It helps in differentiating tyrosine from other amino acids.
- It is used to identify phenolic compounds (monohydroxy benzene derivatives) like phenol β-naphthol and salicylic acid.
- It is used in food quality control to identify casein (tyrosine rich protein) in milk butter and cheese.
- It is used to detect genuine animal proteins in raw and processed meat and to distinguish from plant based fillers.
- It is used in agricultural screening to identify protein content and nutritional value of plant seeds and botanical extracts.
- It is used in art and cultural conservation to identify protein based binders (egg yolk egg tempera animal glue) in old paintings and artifacts.
- It is used in forensic science for identification of biological stains on paper or cloth (along with microscopic analysis).
Advantages of Millon’s Test
- It is a specific test for tyrosine and other phenol (monohydroxy benzene) derivatives and helps in differentiating tyrosine from other amino acids.
- It is a rapid test and cost effective screening method when advanced instruments is not available.
- The result is easily observed by pink to brick-red colour precipitate or solution which gives clear visual confirmation.
- It is used in different applied fields like academic research forensic science and food quality control (casein detection in dairy and protein detection in meat).
- Modified Millon’s test (Cole’s modification) is less affected by inorganic salts like chlorides and helps in reducing false negative results.
Limitations of Millon’s Test
- It is not completely specific for proteins because any compound having phenol group (phenol salicylic acid β-naphthol etc.) can give positive result so it cannot be used alone and should be confirmed with other tests like Biuret test or Ninhydrin test.
- Chloride ions can interfere in this test. Chlorides reacts with mercury and forms insoluble mercuric chloride and mercury cannot react with tyrosine so false negative result is obtained.
- If the solution is too alkaline then mercury can precipitate as yellow mercuric oxide which inhibits the reaction and gives false negative result.
- The test depends on concentration. Highly diluted protein sample or proteins having low tyrosine content (like gelatin) may not give visible red precipitate.
- Presence of ammonium salts in sample can also interfere with the reaction.
- On adding Millon’s reagent a white or yellow precipitate can be formed due to denaturation of proteins by mercury ions and it should not be confused with final result which appears only after heating.
- Millon’s reagent contains concentrated nitric acid and mercury salts so it is corrosive and highly toxic and it is also an environmental pollutant so proper safety and hazardous waste disposal is required.
References
- Alareqe, N. A., Roslan, S., Nordin, M. S., Ahmad, N. A., & Taresh, S. M. (2021). Psychometric properties of the Millon Clinical Multiaxial Inventory–III in an Arabic clinical sample compared with American, Italian, and Dutch cultures. Frontiers in Psychology, 12, 562619. https://doi.org/10.3389/fpsyg.2021.562619
- Al-Sharifi, N. (2021). Biochemistry lab 6: Xanthoproteic test, Millon’s test, Hopkin-Cole’s test. AL-RASHEED PHARMACY Dpt. https://www.alrasheedcol.edu.iq/lect-download-1680
- Amino acids and proteins. (n.d.). http://14.139.61.83/BioChemicalEstimations/amino_acids_and_proteins.htm
- Amrita Vishwa Vidyapeetham Virtual Lab. (2011). Qualitative analysis of amino acid (theory). https://vlab.amrita.edu/index.php?sub=3&brch=63&sim=1094&cnt=1
- Biology Online. (2021, June 28). Millons test definition and examples. Biology Online Dictionary. https://www.biologyonline.com/dictionary/millons-test
- BYJU’S. (n.d.). Test for protein. https://byjus.com/biology/test-for-protein/
- CDH Fine Chemical. (2023, December 30). Millon’s reagent (for protein). https://www.cdhfinechemical.com/images/product/specs/39_1794386524_MILLONSREAGENT(FORPROTEIN)_866270.pdf
- Choca, J. P., Shanley, L. A., & Van Denburg, E. (1992). Interpretative guide to the Millon Clinical Multiaxial Inventory. American Psychological Association. https://dokumen.pub/interpretative-guide-to-the-millon-clinical-multiaxial-inventory.html
- Color tests for proteins and amino acids. (n.d.). https://faculty.ksu.edu.sa/sites/default/files/CLS%20281%20-%20Exp2.%20Color%20Tests%20for%20Specific%20Proteins%20and%20Amino%20Acids%20%20.pdf
- Comprehensive analytical assessment of Millon’s test: Chemical principles, methodological protocols, and applications in protein biochemistry. (n.d.).
- Doménech-Carbó, M. T., & Doménech-Carbó, A. (2021). Spot tests: Past and present. ChemTexts, 8(1), 4. https://doi.org/10.1007/s40828-021-00152-z
- Fisher Scientific. (2006, March 22). Material safety data sheet – Millon’s reagent. Cole-Parmer. https://pim-resources.coleparmer.com/sds/45366.pdf
- Fisher Scientific. (2025, December 20). Safety data sheet: Millon’s reagent. https://www.fishersci.com/store/msds?partNumber=AC458541000&productDescription=MILLON+S+REAGENT+FOR+TH+100ML&vendorId=VN00032119&countryCode=US&language=en
- HiMedia Laboratories. (2022). R097 Millon’s reagent. https://www.himedialabs.com/media/TD/R097.pdf
- Khalil, H., & Basam, S. (2023). Lab 2: Bio chemistry – Amino acids. https://www.uomustansiriyah.edu.iq/media/lectures/6/6_2023_06_30!09_30_40_PM.pdf
- Navora, B. T. (2021, September 7). Experiment #3 title: Millon’s test. Scribd. https://www.scribd.com/document/546256658/Millon-s-Test
- Oxford Lab Fine Chem LLP. (n.d.). Material safety data sheet: Millon’s reagent. https://www.oxfordlabchem.com/msds/%28M-06010%29%20MILLIONS%20REAGENT.pdf
- Romero, A. R. M., & Araullo, S. (n.d.). Millon’s test for tyrosine detection. Scribd. https://www.scribd.com/presentation/231076613/Millon-Test
- Sapkota, A. (2022, September 5). Hopkin’s Cole test- definition, principle, procedure, result, uses. Microbe Notes. https://microbenotes.com/hopkins-cole-test/
- Sapkota, A. (n.d.). Millon’s test- definition, principle, procedure, result, uses. Microbe Notes. https://microbenotes.com/millons-test/
- S D Fine-Chem Limited. (2018, June 16). GHS safety data sheet: Millon’s reagent. https://sdfine.com/media/catalog/product/attachment/29085MSDS.pdf
- Scholar Chemistry. (2012, September 22). Material safety data sheet: Millon’s test solution. Columbus Chemical Industries, Inc. https://bpb-us-w2.wpmucdn.com/sites.broward.edu/dist/9/20/files/2021/01/Millons_Test_Solution-SDS.pdf
- Singla, R. (n.d.). Millon’s reagent: Preparation, properties & applications. Vedantu. https://www.vedantu.com/chemistry/millons-reagent
- Subroto, E., Lembong, E., Filianty, F., Indiarto, R., Primalia, G., Putri, M. S. K. Z., Theodora, H. C., & Junar, S. (2020). The analysis techniques of amino acid and protein in food and agricultural products. International Journal of Scientific & Technology Research, 9(2). https://www.ijstr.org/final-print/oct2020/The-Analysis-Techniques-Of-Amino-Acid-And-Protein-In-Food-And-Agricultural-Products.pdf
- ThomasTKtungnung. (n.d.). Millon’s test practical experiment [Video]. YouTube. https://www.youtube.com/watch?v=cOQehG4hRMA
- Wikipedia. (2025, August 24). Millon Clinical Multiaxial Inventory. https://en.wikipedia.org/w/index.php?title=Millon_Clinical_Multiaxial_Inventory&oldid=1307523599
- Wikipedia. (2025, December 8). Millon’s reagent. https://en.wikipedia.org/w/index.php?title=Millon%27s_reagent&oldid=1326389637
- Wikipedia. (2026, March 10). Ninhydrin. https://en.wikipedia.org/w/index.php?title=Ninhydrin&oldid=1342723659