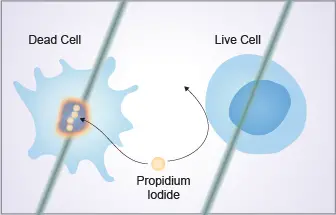
Cell viability analysis using Propidium Iodide is a simple method in which the dye is used to distinguish between living and dead cells. It is the process where Propidium Iodide (PI) is added to a cell suspension and the stained cells is then examined usually by flow cytometer. PI is a red-fluorescent dye that binds with nucleic acid. It is important that viable cells have an intact membrane, so the dye cannot enter inside these cells.
Dead cells lose this membrane integrity and this is the condition when PI can pass through the disordered membrane. Once the dye enters, it binds or intercalates with the DNA present in the nucleus and the dead cells emits red fluorescence.
This is referred to as the basic principle of this analysis. It is applied on cells obtained from blood, bone marrow or cultured cells prepared in a suspension (1×10⁶ cells/ml).
The dye is added usually at 2 µg/ml and incubated for about 15 minutes in ice and in dark, and then the cells is examined. It is important to handle PI carefully because the dye is considered a suspected carcinogen.
Aim
To distinguish between living and dead cells.
Principle of Viability staining using propidium iodide
The principle of viability staining using Propidium Iodide is based on the integrity of the cell membrane and the ability of the dye to bind with nucleic acid.
Propidium Iodide (PI) is a red-fluorescent dye and it cannot cross the intact membrane of viable cells. It is the process where the dye only enters those cells in which the membrane is damaged or disrupted. When PI passes through this disordered membrane, it moves inside the nucleus and intercalates with the DNA.
The stained dead cells then emit red fluorescence which is detected. This differential entry of dye makes it possible to distinguish the living and dead cells. The method is mainly applied on cells in suspension prepared from whole blood, bone marrow or cultured cells.
Requirement
- Cells in suspension obtained from whole blood, bone marrow or cultured cells.
- FACS tubes or 5 ml round-bottom polypropylene test tubes.
- Pipettes, microtips, centrifuge and vortex.
- PBS (1X) or HBSS buffer for washing the cells.
- Staining buffer such as PBS + 2% FBS or PBS containing BSA.
- Propidium Iodide solution prepared either as a stock (1 mg/ml) or as working PI staining solution.
- Final PI concentration required is 2 µg/ml in the cell suspension.
- Trypsin or Accutase when adherent cells is used.
- Washed cells are required before staining.
- Incubation of the stained cells for about 15 minutes on ice and kept in dark.
- The stained cells is examined directly without further washing.
- Flow cytometer with proper setting for PI fluorescence detection.
- Negative control tube without PI and positive control tube containing PI.
- Proper safety precautions because PI is considered a suspected carcinogen.
Procedure of Viability test by propidium iodide
Propidium iodide is a fluorescent molecule that intercalates with nucleic acids. It cannot pass through the intact membrane, so the viable cells exclude the dye but the non-viable cells take it up. The reaction is detected in the flow cytometer where PI fluorescence is measured in specific channels. It is the basis for determining the percentage of dead cells in a sample.
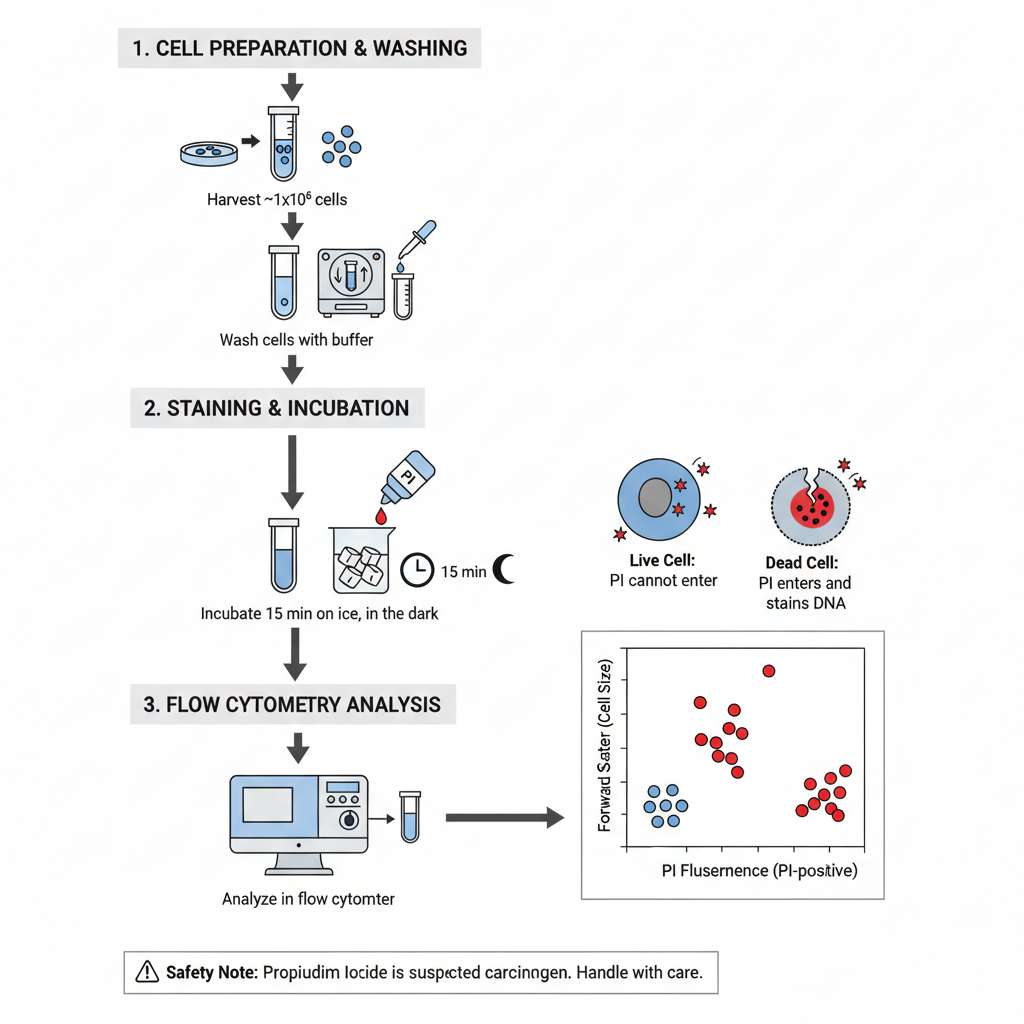
I. Cell Preparation and Washing
The cell suspension is prepared first and then washed to remove unwanted components.
1. Harvest and aliquot cells
- Cells are harvested and around 1×10⁶ cells is taken in 100 µL volume in FACS tubes.
- For suspension cells, these are resuspended in 1 mL PBS + 2% FBS and aliquoted in polypropylene tube.
- Adherent cells need extra steps. It is the process where cells are trypsinized at 37°C (around 10 min), then medium with serum is added to stop trypsin action. The suspension is pipetted up and down so that clumps are separated. The cells are then transferred to tubes and spun. Some detachment agents like Accutase enzyme can also be used.
2. Wash cells– Cells are washed by adding about 2 mL PBS or HBSS and centrifuged at 300×g for 5 min. The buffer is decanted and the washing is repeated two times. Suspension cells are washed two times with PBS + 2% FBS.
3. Resuspend– After washing, the pellet is resuspended in around 100 µL Flow Cytometry Staining Buffer.
II. Staining and Incubation
In this step PI stain is added to the washed cell suspension.
1. Add PI stain– It is added in a final concentration of 2 µg/mL. Sometimes 5–10 µL of 10 µg/mL PI stock solution in PBS is added just before analysis.
2. Incubation– The stained samples are kept for 15 min on ice in the dark. It is important that strong light is avoided once PI is added.
3. Do not wash– The cells are not washed after PI staining because this will remove the dye from damaged cells.
III. Flow Cytometry Analysis
After incubation the sample is directly loaded into the flow cytometer.
1. Analyze sample– The samples are run in the flow cytometer.
2. Determine PI fluorescence– PI fluorescence is commonly read in FL-2 or FL-3.
- If PI alone is used, the FL-2 channel is taken.
- If FITC- or PE-conjugated antibodies are also used, then PI is collected in FL-3.
3. Set stop count– The stop count is set based on viable cells seen on dot-plot of forward scatter versus PI.
4. Acquire data– Data is acquired for experimental samples and also for the controls like unstained cells and positive controls.
Important Notes
Pre-staining condition– Surface antigen staining with antibody can be performed before adding PI, but PI cannot be used for intracellular staining procedures.
Safety requirement– Propidium iodide is suspected carcinogen. It is advised that proper protective measures are used, and all PI wastes are disposed safely.
Result of Viability Test by Propidium Iodide (PI)
The result of the viability test is the percentage of viable and non-viable cells obtained after flow cytometric examination of PI-stained samples. It is based on the membrane integrity of the cells.
Non-viable cells take up the dye because the membrane is damaged. It is the process where PI enters the dead cells and bind with the double-stranded DNA, causing strong red fluorescence. These are observed as PI-positive cells in the cytometer.
Viable cells exclude the dye because the membrane is intact. These cells do not emit red fluorescence and are detected as PI-negative population during the analysis.
In the flow cytometer, the PI fluorescence is mostly collected in FL-2 or FL-3 channel depending on other fluorochromes used. The scatter plot or histogram clearly separates the live and dead cells on the basis of fluorescence intensity. The final data gives the proportion of each group in the sample.
This result is important because it indicates the true viability of the cell suspension before any further experiment. Dead cells if present in higher number may cause non-specific staining and can interfere in downstream applications. It is also used to understand the effect of different treatments as cells under stress show higher PI positivity.
Among the important applications, the viability result is used in oncology studies for checking drug response where the increase in PI-positive cells is associated with loss of viability. In addition, information related to DNA content and cell cycle distribution can also be obtained during the same analysis.
Advantages of Propidium Iodide (PI) Staining
- It is a quick method because PI stained cells are analysed directly in the flow cytometer and the proportion of live and dead cells is obtained rapidly.
- It is effective in excluding the non-viable cells since PI is membrane impermeant and the viable cells exclude the dye.
- It helps to reduce artifacts because the dead cells which take up fluorescent probes non-specifically are identified and removed from further analysis.
- It is compatible with multi-color staining as PI is excited at 488 nm and the emission is around 617 nm, so it can be used along with FITC or PE in the same sample.
- It provides additional information on the DNA content and cell cycle stage because PI binds with nucleic acids in a proportional manner.
- It is significant in clinical and research areas, especially in oncology, where the viability result is taken to check the drug response of treated cells.
Limitations of Propidium Iodide (PI) Staining
- PI cannot be used for intracellular staining because the membrane is permeabilized during such procedures and the dye will enter all cells, so the distinction between live and dead cells is lost.
- It is a suspected carcinogen, therefore proper safety precautions are required and the waste must be disposed safely.
- The PI stock solutions have limited stability as solutions exposed to room temperature for long time or showing dark red colour are discarded, and the working aliquots can be stored only for a short duration at 4°C.
- The staining procedure is sensitive to light because strong light can affect PI fluorescence, and the incubation must be done in the dark.
- The cells must not be washed after adding PI, and any washing at this stage can alter the staining result.
Uses of Propidium Iodide (PI) Staining
- It is used to quantify the viable and non-viable cells in a population because PI staining gives a clear live and dead cell separation in flow cytometry.
- It is used to evaluate the cellular response under different treatments as the change in PI positive cells indicate loss of membrane integrity.
- It helps to exclude the dead cells from analysis since these cells produce non-specific staining and can affect further experiments.
- It is used in oncology studies to check the effect of anticancer drugs where an increase in PI staining shows reduced cell viability.
- It provides information related to DNA content and cell cycle stage because PI binds with nucleic acids proportionally.
- It can be used along with other fluorochromes like FITC and PE, as PI is excited at 488 nm, allowing simultaneous staining of surface antigens with viability checking.
- https://www.rndsystems.com/resources/protocols/flow-cytometry-protocol-analysis-cell-viability-using-propidium-iodide
- https://flowcyt.rutgers.edu/wp-content/uploads/2017/10/Cell-Viability-using-PI.pdf