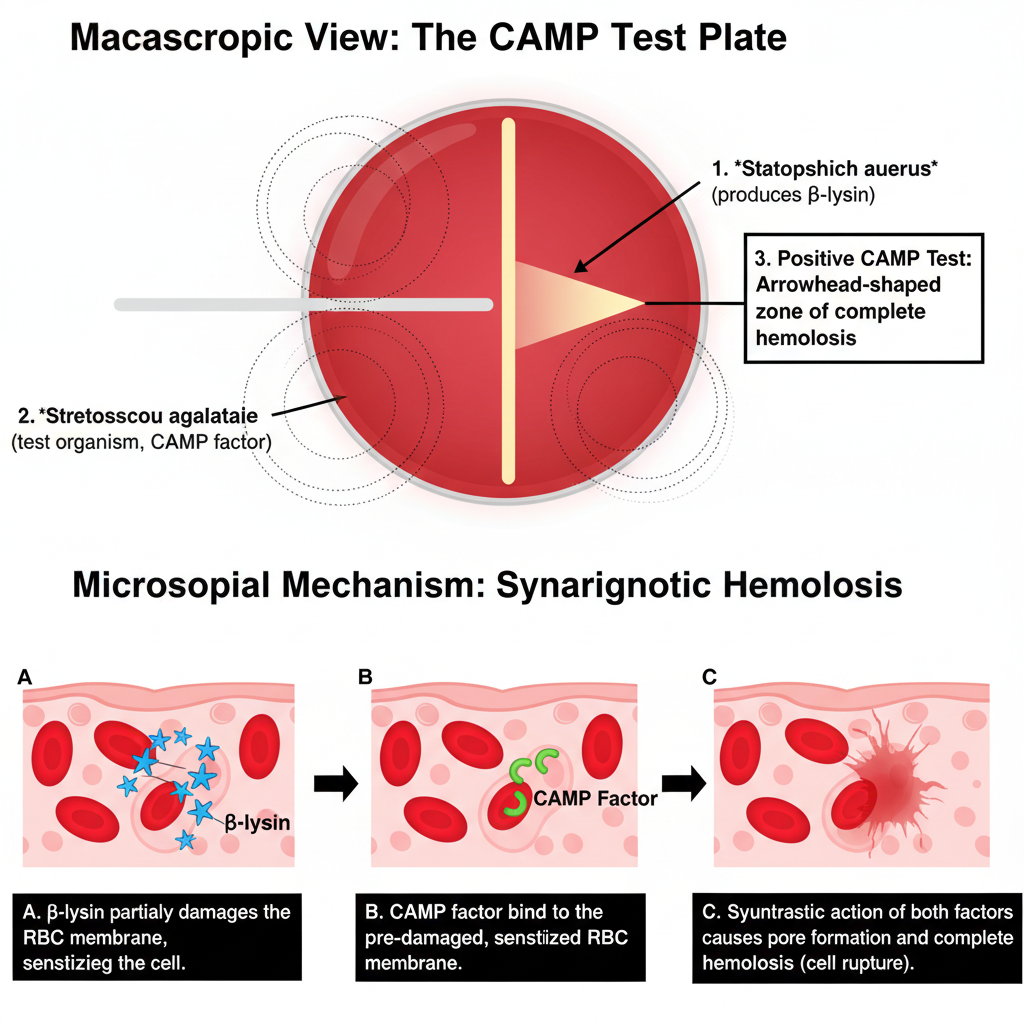
CAMP test (Christie-Atkins-Munch-Petersen test) is a biochemical test that is used for presumptive identification of Group B Streptococcus mainly Streptococcus agalactiae and it helps to differentiate it from other beta hemolytic streptococci.
It is based on synergistic interaction between extracellular products of two bacteria. Streptococcus agalactiae secretes a diffusible heat-stable protein called CAMP factor (Protein B) which acts synergistically with the beta hemolysin (sphingomyelinase C) produced by Staphylococcus aureus.
In this test, Staphylococcus aureus and the test streptococcus is streaked perpendicularly on sheep blood agar plate. After incubation, enhanced zone of complete hemolysis is produced at the junction area and it appears as a distinct arrowhead or flame shaped hemolysis.
CAMP test is also used to help in identification of Listeria monocytogenes as it can also show a similar positive CAMP reaction.
Objectives of CAMP test
- To presumptively identify Streptococcus agalactiae (Group B Streptococcus) and differentiate it from other beta hemolytic streptococci.
- To detect CAMP factor production by the test isolate (extracellular CAMP factor protein).
- To assist in identification of other CAMP positive organisms mainly Listeria monocytogenes.
Principle of CAMP Test
CAMP test works on the principle of synergistic action between extracellular products of two different bacteria which results in enhanced hemolysis on sheep (or bovine) blood agar.
In this principle, Staphylococcus aureus produces beta hemolysin (sphingomyelinase C) which acts on sphingomyelin present in RBC membrane and it is cleaved to form ceramide. This action does not completely lyse the RBC immediately but the RBC membrane becomes presensitized (metastable state).
Streptococcus agalactiae (Group B Streptococcus) produces a diffusible heat-stable CAMP factor. CAMP factor alone cannot lyse intact RBC but it binds to the ceramide rich area produced by beta hemolysin and the membrane integrity is disturbed and pores are formed leading to rapid osmotic lysis of RBC.
The synergistic hemolysis is observed as a distinct arrowhead shaped zone of enhanced complete hemolysis at the junction area where the extracellular products of the two organisms meet.
Requirement for CAMP test
- Culture media– 5% sheep blood agar plate (Trypticase soy agar with sheep blood) is required. Bovine blood can also be used but human horse rabbit or guinea pig blood agar do not give proper reaction. Agar depth should be around 1.5 mm because excess depth can affect the interpretation.
- Microorganisms– A known Staphylococcus aureus strain producing beta-lysin (sphingomyelinase C) is required (example ATCC 25923) for primary streak.
- Test organism– Suspected isolate (Group B Streptococcus or Listeria monocytogenes) to be streaked perpendicularly to S. aureus.
- Positive control– A known CAMP positive organism such as Streptococcus agalactiae (example ATCC 13813).
- Negative control– A known CAMP negative organism such as Streptococcus pyogenes (example ATCC 19615).
- Equipment and supplies– Sterile inoculating loop/ wooden applicator stick/ edge of needle for making straight line streaks.
- Incubator– 35°C to 37°C is used. Ambient air incubation is preferred because anaerobic condition or high CO2 (candle jar) can give false positive with some Group A streptococci.
- Alternative reagents (modified rapid test)– For disk or spot method, commercial beta-lysin disk or liquid CAMP spot test reagent (staphylococcal beta-lysin extract) is used instead of live S. aureus culture.
Procedure of CAMP test
Standard Plate Procedure
- The test is performed on a trypticase soy agar plate containing 5% sheep blood, because sheep erythrocytes have sphingomyelin required for beta-lysin activity.
- A straight line of beta-lysin producing Staphylococcus aureus is streaked down the center of the plate.
- The suspected organism is streaked perpendicular to the S. aureus streak and it is kept at a distance of about 1–2 mm without touching.
- More than one test organism can be streaked on same plate, but they should remain parallel to each other and perpendicular to the main streak.
- A known Group B Streptococcus strain is used as positive control and Streptococcus pyogenes or Enterococcus species is used as negative control.
- The plate is incubated at 35–37°C for 18–24 hours in ambient air.
- Positive result is indicated when a clear arrowhead-shaped enhanced hemolysis is seen at the junction area. Negative result shows no enhanced hemolysis and only normal hemolytic pattern is seen.

Disk Method (CAMP test) – Procedure
- A sheep blood agar plate is prepared for observing hemolysis because sheep RBC contains sphingomyelin needed for beta-lysin activity.
- A beta-lysin impregnated disk is placed on agar surface using sterile forceps.
- The test organism is streaked in a straight line at a distance of about 2–3 mm from the edge of the disk, keeping the streak parallel to the disk border.
- More than one organism may be tested on same plate but each streak should maintain required spacing from the disk.
- The plate is incubated at 35–37°C for 18–24 hours in ambient air.
- After incubation, a clear crescent-shaped or arc-shaped enhanced hemolysis adjacent to the disk is taken as positive reaction.

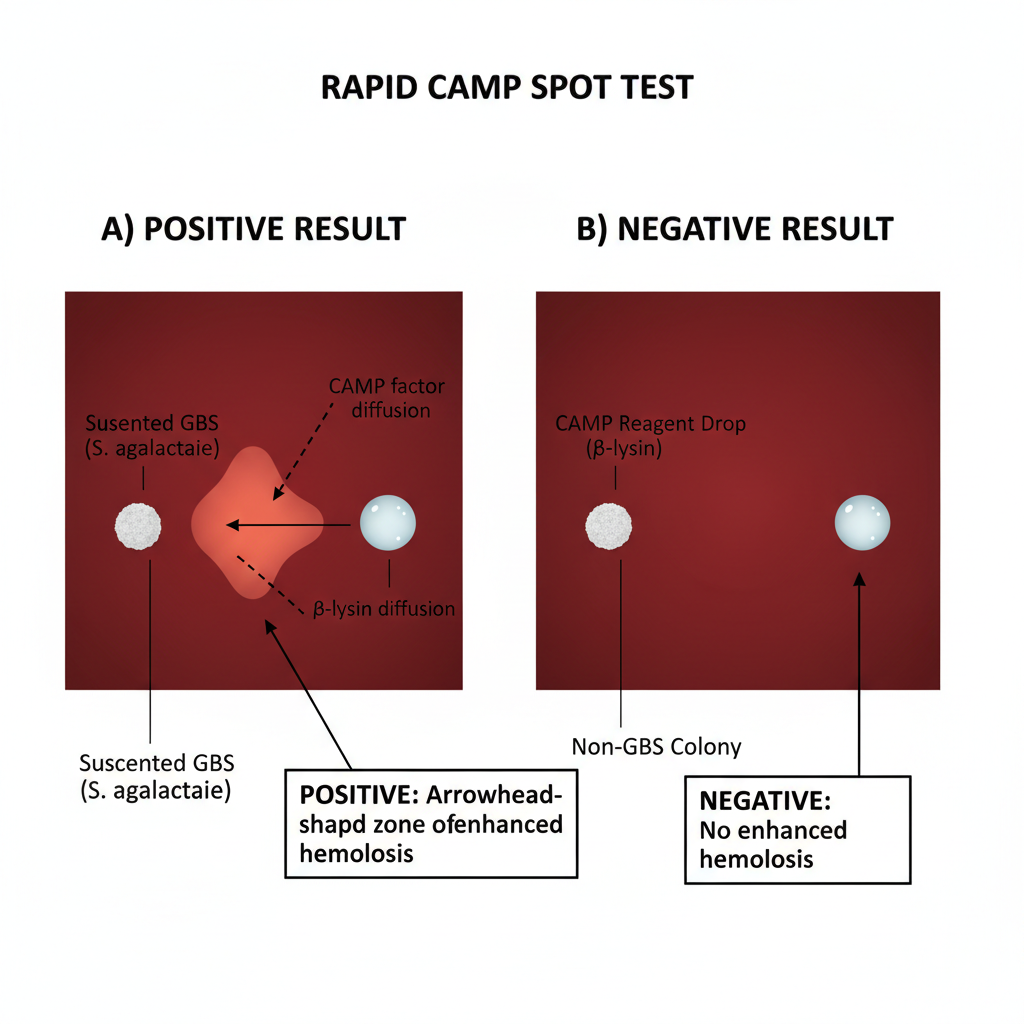
Rapid Spot Test
- A sheep blood agar plate having well isolated colonies of suspected organism is selected.
- A small drop of CAMP reagent containing extracted beta-lysin is placed close to the presumptive colony, keeping the drop near but not touching the colony.
- The plate is kept at room temperature or slightly warmed condition to allow diffusion of reagent towards the colony.
- In this step, CAMP factor diffuses from the colony and it interacts with beta-lysin in the reagent producing localized enhanced hemolysis.
- The plate is observed after about 20 minutes to 1 hour for a clear hemolytic zone around the reagent drop near the colony.
- Positive reaction is indicated by enhanced hemolysis next to suspected colony, and absence of such hemolysis is taken as negative reaction.
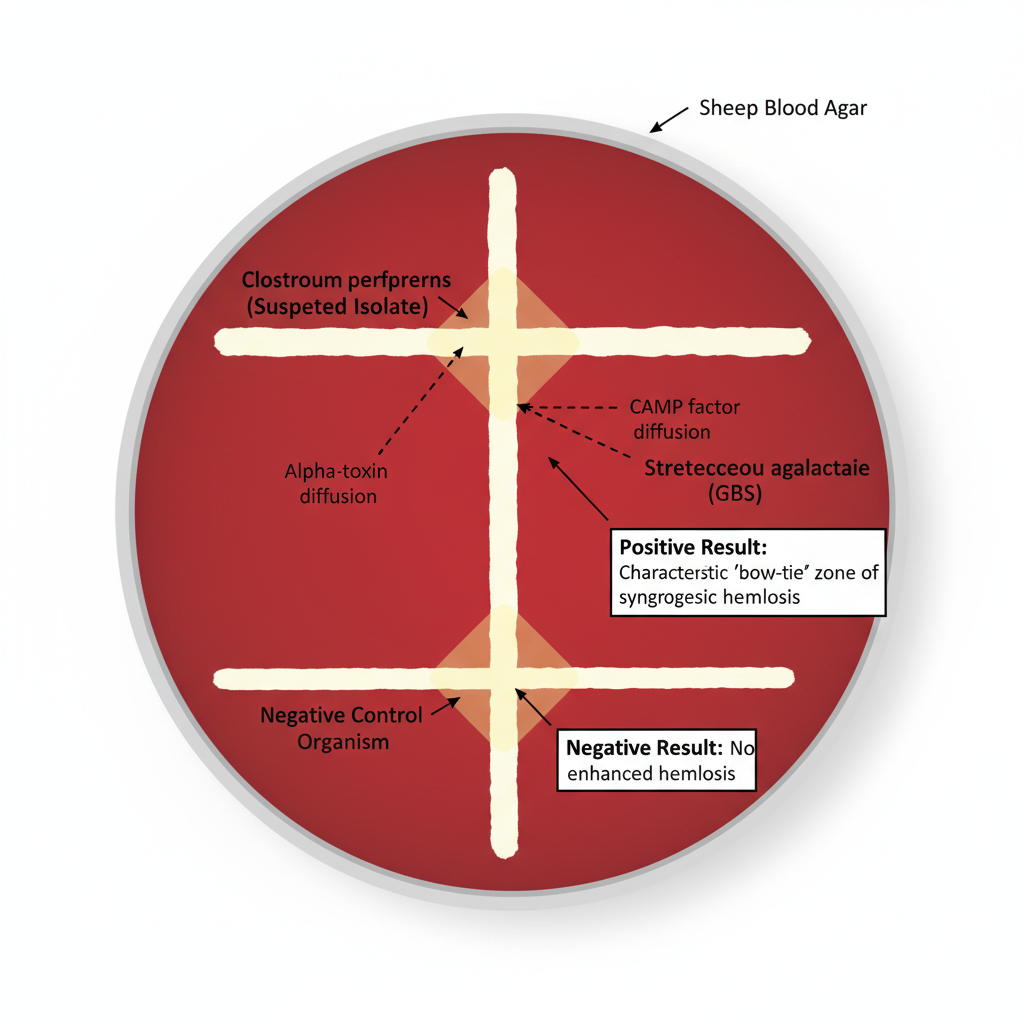
Reverse CAMP Test
- A sheep blood agar plate is selected as the medium because sheep erythrocytes contain sphingomyelin required for beta-lysin activity and synergistic lysis.
- A known CAMP-positive Streptococcus agalactiae (GBS) strain is streaked in a straight line down the center of the plate.
- The suspected Clostridium perfringens isolate is streaked perpendicular to the central GBS streak, keeping a small gap (1–2 mm) and the lines should not touch.
- In this step the alpha-toxin (phospholipase C) produced by C. perfringens diffuses toward the CAMP factor from the GBS strain and synergistic hemolysis condition is produced.
- The plate is incubated anaerobically at 35–37°C for 18–24 hours because C. perfringens requires anaerobic condition for proper toxin expression.
- After incubation, positive reaction is observed as a bow-tie or reverse-arrow shaped enhanced hemolysis at the junction area. Absence of this pattern is taken as negative reaction.
Result of CAMP test

Positive result
- A clear zone of enhanced complete (beta) hemolysis is seen at the junction area where the test organism and Staphylococcus aureus streak comes near.
- In standard plate method, the enhanced hemolysis forms a distinct arrowhead/flame/wedge shaped zone pointing towards the S. aureus streak.
- In Listeria monocytogenes, the positive pattern may appear different and it is seen as weaker rectangular or matchstick shaped hemolysis.
- In rapid methods like CAMP spot test or disk test, positive result is seen as arc/circle/crescent shaped clear zone of enhanced hemolysis.
- Positive CAMP test indicates presence of Streptococcus agalactiae (Group B Streptococcus) or other CAMP positive organism like Listeria monocytogenes.
Negative result
- No enhancement of hemolysis is seen where the streaks approach each other.
- The hemolytic zones of the two organisms do not interact and they remain separate.
- Negative result indicates the organism does not produce CAMP factor (example Streptococcus pyogenes Group A Streptococcus).
List of organisms which gives positive and negative result in CAMP test
Organisms giving Positive CAMP test
- Streptococcus agalactiae (Group B Streptococcus).
- Listeria monocytogenes (rectangular or matchstick type positive).
- Listeria seeligeri (weak positive).
- Actinomyces neuii.
- Rhodococcus equi.
- Turicella otitidis.
- Propionibacterium avidum/granulosum.
- Cutibacterium acnes (Propionibacterium acnes).
- Corynebacterium species (C. glucuronolyticum, C. coyleae, C. imitans and some strains of C. striatum group and C. afermentans group).
- Other Streptococcus species (S. porcinus, S. pseudoporcinus, S. iniae and some Group G streptococci).
- Pasteurella haemolytica.
- Aeromonas spp. and Vibrio spp.
- Listeria ivanovii (positive only in modified CAMP test using Rhodococcus equi instead of Staphylococcus aureus).
- Clostridium perfringens (positive in reverse CAMP test showing bow-tie pattern).
- Streptococcus pyogenes (Group A Streptococcus) can rarely show false positive if incubation is done in candle jar/anaerobic/elevated temperature.
Organisms giving Negative CAMP test
- Streptococcus pyogenes (Group A Streptococcus) usually negative in proper ambient air incubation (standard negative control).
- Other non Group B streptococci like Lancefield Group C and Group D.
- Listeria innocua.
- Clostridium septicum (negative in reverse CAMP test).
Note- Arcanobacterium haemolyticum shows CAMP inhibition reaction (it blocks hemolysis of Staphylococcus aureus) but it can show a positive crescent shaped reverse CAMP type reaction when tested with Streptococcus agalactiae.
Quality Control organisms of CAMP test
- Positive control– Streptococcus agalactiae (ATCC 13813) is used and it gives characteristic enhanced arrowhead zone of hemolysis.
- Negative control– Streptococcus pyogenes (ATCC 19615) is used and it shows only normal beta hemolysis without any enhanced arrowhead formation.
Uses of CAMP Test
- It is used for presumptive identification of Streptococcus agalactiae (Group B Streptococcus) and it helps to differentiate it from other beta hemolytic streptococci.
- It is used to identify Listeria monocytogenes as it also gives a positive CAMP reaction and it is affordable diagnostic test.
- It is used to detect CAMP factor production and to check whether a bacterial isolate can produce and secrete CAMP factor protein.
- It is used to assist in identification of some CAMP positive Gram positive rods like Rhodococcus equi Actinomyces neuii Propionibacterium avidum/granulosum and some Corynebacterium species.
- Modified CAMP test using Rhodococcus equi instead of Staphylococcus aureus is used to identify Listeria ivanovii.
- Reverse CAMP test (variation of CAMP principle) is used to identify Clostridium perfringens by bow-tie or reversed arrowhead hemolysis pattern.
- CAMP inhibition test is used to identify Arcanobacterium haemolyticum as it can inhibit the beta hemolysis of Staphylococcus aureus.
Limitations of CAMP Test
- Specific blood agar is required– The test must be performed on sheep or bovine blood agar. Human horse rabbit or guinea pig blood agar do not give proper reaction because lipid composition is not suitable.
- Agar depth and quality issue– If agar is too thick or too thin or already hemolyzed then reaction becomes feeble and it is difficult to interpret (about 1.5 mm depth is recommended).
- False positive result can occur– Some Group A streptococci (Streptococcus pyogenes) can show positive CAMP reaction especially when plate is incubated anaerobically or in CO2 (candle jar) or for extended time or at high temperature. Other organisms like Listeria monocytogenes Streptococcus porcinus and group G streptococci can also show similar enhanced hemolysis.
- False negative result (CAMP negative strains)– Some strains of Streptococcus agalactiae may not produce CAMP factor due to mutation/deletion in cfb gene so the characteristic hemolysis is not produced.
- Timing sensitivity– If colonies are tested before 18 hours, CAMP factor production may be less and false negative is seen. If incubation is prolonged beyond 48 hours blood medium can darken and results becomes difficult to read.
- Dependency on Staphylococcus aureus strain– The test depends on beta hemolysin producing S. aureus strain. If the strain used does not secrete beta hemolysin the test fails. Some sheep blood batches may contain beta antitoxin that inhibits the toxin so quality control is required.
- Not a standalone diagnostic test– CAMP test alone cannot confirm the pathogen. It should be combined with colony morphology gram staining catalase test and other biochemical tests (like PYR test to rule out Group A streptococcus).
Advantages of CAMP Test
- Cost effective and affordable– It is an inexpensive diagnostic test and it is useful for small laboratories where costly methods like PCR or MALDI-TOF is not available.
- Simple and easy to perform– It needs basic laboratory skills and simple materials like sheep blood agar and simple streaking method.
- Prompt and reliable– It gives rapid presumptive identification of Group B Streptococcus and Listeria monocytogenes. Result is observed in about 18 hours in standard method and faster result is possible in disk or spot test.
- High specificity– It helps to differentiate Streptococcus agalactiae from other beta hemolytic streptococci and false positive is less in most Streptococcus species.
- Clinically useful for Listeria– It helps in separating Listeria monocytogenes (important pathogen) from other less pathogenic Listeria species.
- Functional assessment– It gives functional idea of hemolytic synergy and virulence character because actual toxin interaction is seen rather than only genetic marker detection.
- Good confirmatory tool– It can be used as backup confirmatory test when automated systems or grouping kits gives ambiguous or inconclusive results.
Precautions of CAMP test
- Sheep or bovine blood agar should be used. Human horse rabbit or guinea pig blood agar do not give proper synergistic reaction.
- Incubation should be done in ambient air. Anaerobic condition or candle jar (CO2) can give false positive with Group A streptococci.
- Incubation time and temperature should be controlled. Prolonged incubation and elevated temperature can produce false positive. For spot test, colonies of about 18 to 48 hours is preferred otherwise false negative or unreadable result can occur due to darkened agar.
- Agar depth and quality should be checked. Agar should not be too thin and it should not be already hemolyzed because reaction becomes feeble. Around 1.5 mm depth is recommended (especially for spot test).
- CAMP test should not be used alone. It should be combined with colony morphology Gram staining catalase test and PYR test to differentiate Group B Streptococcus from Group A Streptococcus and also from Listeria.
- Hemolysis pattern should be observed carefully. The definite arrowhead hemolysis of Group B Streptococcus should be differentiated from non specific matchstick type hemolysis.
- False negative should be considered because some Streptococcus agalactiae isolates are naturally CAMP negative so further tests are needed even if result is negative.
- Quality control of sheep blood agar lot should be done because some blood batches may contain beta antitoxins which inhibits staphylococcal beta toxin and the test can fail.
References
- Bernheimer, A. W., Linder, R., & Avigad, L. S. (1979). Nature and mechanism of action of the CAMP protein of group B streptococci. Infection and Immunity, 23(3), 838–844. https://doi.org/10.1128/iai.23.3.838-844.1979
- Bioactiva. (n.d.). CAMP Spot Test Reagent – for Group B Streptococcus and Listeria monocytogenes, “Bow Tie” enhanced hemolysis.
- Boyanov, V. S., Alexandrova, A. S., & Gergova, R. T. (2025). Diagnostic challenges and genotypic characteristics of CAMP-negative phenotype GBS isolates. Journal of Pure and Applied Microbiology, 19(4), 2779–2787. https://doi.org/10.22207/JPAM.19.4.16
- Creti, R., Imperi, M., Stanziale, A., Giuliani, G., Fazii, P., & Savini, V. (2023). Group B streptococci (GBS) strains evading molecular diagnostics showed novel chromosomal deletions encompassing the CAMP-factor (cfb) encoding gene. European Journal of Clinical Microbiology & Infectious Diseases, 42(7), 913–916. https://doi.org/10.1007/s10096-023-04620-x
- Department of Medical Microbiology and Infectious Diseases, ErasmusMC. (n.d.). CAMP-test_Listeria monocytogenes & L. ivanovii. Microbe Canvas.
- Doran, K. S., & Nizet, V. (2004). Molecular pathogenesis of neonatal group B streptococcal infection: no longer in its infancy. Molecular Microbiology, 54(1), 23–31. https://doi.org/10.1111/j.1365-2958.2004.04266.x
- DrChika. (2023, January 9). CAMP test for identification of Group B streptococci. Microbiology Class.
- El-Huneidi, W., Mui, R., Zhang, T. H., & Palmer, M. (2007). Streptococcus agalactiae CAMP factor/protein B does not bind to human IgG. Medical Microbiology and Immunology, 196(2), 73–77. https://doi.org/10.1007/s00430-006-0028-8
- Gase, K., Ferretti, J. J., Primeaux, C., & McShan, W. M. (1999). Identification, cloning, and expression of the CAMP factor gene (cfa) of group A streptococci. Infection and Immunity, 67(9), 4725–4731. https://doi.org/10.1128/IAI.67.9.4725-4731.1999
- Hanson, A. (2006, October 9). CAMP test protocols. American Society for Microbiology.
- Harrens Lab. (n.d.). What is camp test microbiology?
- Jürgens, D., Sterzik, B., & Fehrenbach, F. J. (1987). Unspecific binding of group B streptococcal cocytolysin (CAMP factor) to immunoglobulins and its possible role in pathogenicity. Journal of Experimental Medicine, 165(3), 720–732. https://doi.org/10.1084/jem.165.3.720
- Kang, H., Park, G., Kim, H., & Chang, K. (2016). Haemolytic differential identification of Arcanobacterium haemolyticum isolated from a patient with diabetic foot ulcers. JMM Case Reports, 3(1), e005016. https://doi.org/10.1099/jmmcr.0.005016
- Krawczyk-Balska, A., & Bielecki, J. (2005). Listeria monocytogenes listeriolysin O and phosphatidylinositol-specific phospholipase C affect adherence to epithelial cells. Canadian Journal of Microbiology, 51(9), 745–751. https://doi.org/10.1139/w05-058
- Lang, S., Xue, J., Guo, Z., & Palmer, M. (2007). Streptococcus agalactiae CAMP factor binds to GPI-anchored proteins. Medical Microbiology and Immunology, 196(1), 1–10. https://doi.org/10.1007/s00430-006-0021-2
- Listeria monocytogenes virulence factors, including listeriolysin O, are secreted in biologically active extracellular vesicles. (n.d.). PubMed Central (PMC).
- Merck Manual Professional Edition. (n.d.). Table: Classification of Common Pathogenic Bacteria.
- Pokhrel, P. (2015, September 22). CAMP test- principle, purpose, procedure, result and limitation. Microbiology Notes.
- Rao, P. N. S. (2009, November). CAMP test. Microrao.
- Sapkota, A. (2022, January 27). CAMP test- principle, procedure, types, results, uses, limitations. Microbe Notes.
- Savini, V., Paparella, A., Serio, A., Marrollo, R., Carretto, E., & Fazii, P. (2014). Staphylococcus pseudintermedius for CAMP-test. International Journal of Clinical and Experimental Pathology, 7(4), 1733–1734.
- Streptococcus pyogenes streptolysin O as a cause of false-positive… (n.d.). PubMed Central (PMC).
- Structural basis for the unique molecular properties of broad-range phospholipase C from Listeria monocytogenes. (n.d.). PubMed Central (PMC).
- Structure determination of the CAMP factor of Streptococcus agalactiae with the aid of an MBP tag and insights into membrane-surface attachment. (n.d.). PubMed Central (PMC).
- The biochemical and clinical foundations of the Christie-Atkins-Munch-Petersen phenomenon in diagnostic microbiology. (n.d.).
- Wikipedia contributors. (2025, November 17). CAMP test. In Wikipedia, The Free Encyclopedia. https://en.wikipedia.org/w/index.php?title=CAMP_test&oldid=1322615888
- Wilkinson, H. W. (1977). CAMP-disk test for presumptive identification of group B streptococci. Journal of Clinical Microbiology, 6(1), 42–45. https://doi.org/10.1128/jcm.6.1.42-45.1977
- Yeh, E., Pinsky, B. A., Banaei, N., & Baron, E. J. (2009). Hair sheep blood, citrated or defibrinated, fulfills all requirements of blood agar for diagnostic microbiology laboratory tests. PLoS ONE, 4(7), e6141. https://doi.org/10.1371/journal.pone.0006141
- Zhou, J., Zhang, L., Zhang, Y., et al. (2023). Analysis of molecular characteristics of CAMP-negative Streptococcus agalactiae strains. Frontiers in Microbiology, 14, 1189093.