Benedict’s test is a chemical analytical test. It is used for detection of reducing sugars in given sample. The reducing sugars mainly detected are glucose fructose and lactose. It is a simple test and the result is seen by colour change.
In this test Benedict’s reagent is used. It is a bright blue solution and it contains copper sulphate (CuSO4) sodium carbonate (Na2CO3) and sodium citrate. When sample containing reducing sugar is mixed with Benedict’s reagent and heated then reducing sugar acts as reducing agent in alkaline medium. The blue cupric ions (Cu2+) is reduced to cuprous ions (Cu+). Due to this reaction an insoluble precipitate of cuprous oxide (Cu2O) is formed and colour change is obtained.
The colour change depends on concentration of reducing sugar. Blue colour changes to green yellow orange or brick red. Brick red precipitate indicate high concentration of sugar. It is widely used in laboratory and in food science for checking simple sugars. It is also used in clinical test for detection of glucose in urine as presumptive test for diabetes mellitus.
Benedict’s Test Definition
Benedict’s Test is a qualitative chemical assay used to detect the presence of reducing sugars in a solution, characterized by the formation of a brick-red precipitate upon reaction with Benedict’s reagent.
Objectives of Benedict’s Test
Objectives of Benedict’s test are–
- Detection – It is used for detection of reducing sugars in given sample solution.
- Medical diagnosis – It is used as presumptive diagnostic test for diabetes mellitus by detecting glucose in urine sample.
- Estimation – It is used for estimating approximate concentration of reducing sugar present in sample based on colour change.
- Differentiation – It is used for differentiating and identifying unknown extracted carbohydrates in biochemical analysis.
Principle of Benedict’s Test
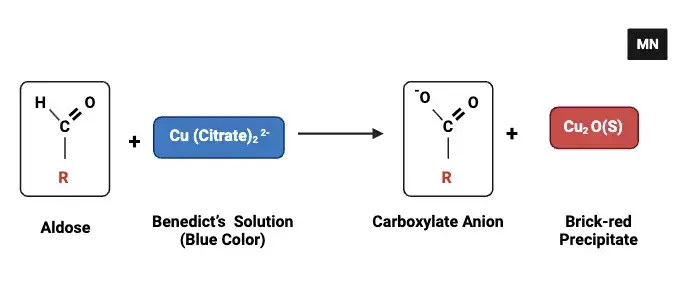
Principle of Benedict’s test is based on reaction between reducing sugar and copper ions in warm alkaline medium. The reducing sugars contain free aldehyde group or ketone group. When it is heated with Benedict’s reagent in presence of sodium carbonate then enediols is formed and it acts as strong reducing agent. These enediols reduces the blue cupric ions (Cu2+) of copper sulphate to cuprous ions (Cu+).
In Benedict’s reagent sodium citrate is present. It acts as chelating agent and it keeps cupric ions in dissolved form and prevents premature precipitation. After reduction the cuprous ions forms insoluble cuprous oxide (Cu2O) precipitate. Due to formation of this precipitate the colour of solution changes from blue to green yellow orange or brick red. The intensity of colour is proportional to concentration of reducing sugar present in sample.
Materials Required of Benedict’s Test
Materials required of Benedict’s test are–
- Benedict’s reagent – It is the reagent used for test. It contains copper sulphate (CuSO4) sodium carbonate (Na2CO3) and sodium citrate.
- Sample solution – The sample to be tested like carbohydrate solution food extract or urine sample.
- Test tubes – It is used for taking reagent and sample mixture.
- Test tube holder/stand – It is used for holding test tube during heating and for keeping it upright for observation.
- Pipette or dropper – It is used for measuring and transferring reagent and sample.
- Heat source – Boiling water bath hot plate or Bunsen burner is used for heating and for starting reaction.
- Glass rod – It is used for mixing the solution gently.
- Water – It is used for dissolving solid sample or for preparing hot water bath.
Benedict’s Reagent Preparation
Benedict’s reagent composition (for 1 litre) are–
- Copper(II) sulphate pentahydrate (CuSO4·5H2O) – 17.3 g. It provides cupric ions (Cu2+) which is reduced during test.
- Sodium carbonate (Na2CO3) anhydrous – 100 g. It provides alkaline medium for reaction. (Alternatively sodium carbonate decahydrate 270 g can be used).
- Sodium citrate (Na3C6H5O7) – 173 g. It acts as complexing/chelating agent and it keeps copper ions in dissolved form and prevents premature precipitation.
- Distilled water – It is used as solvent and final volume is made upto 1000 ml.
Preparation of Benedict’s reagent are–
- Sodium carbonate and sodium citrate is weighed and dissolved together in about 800 ml distilled water. Gentle warming can be done for complete dissolution.
- In separate beaker 17.3 g copper(II) sulphate pentahydrate is dissolved in about 100 ml distilled water.
- Copper sulphate solution is added slowly into carbonate–citrate solution with constant stirring.
- Distilled water is added to make final volume exactly 1 litre (1000 ml).
- The solution is mixed properly and stored in clean reagent bottle. The reagent is clear deep blue solution.
Procedure of Benedict’s Test
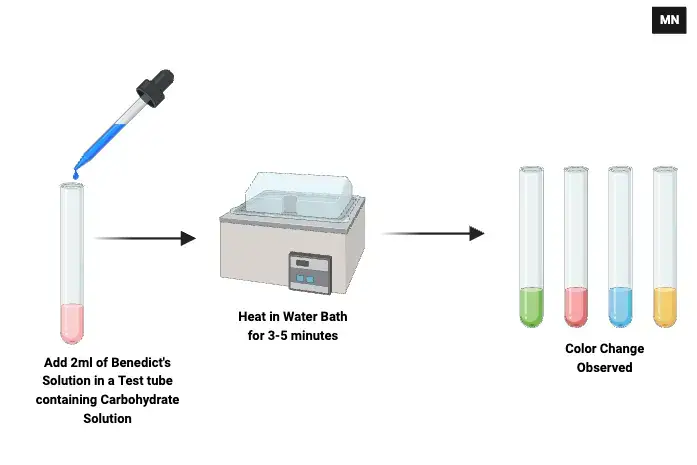
Procedure of Benedict’s test are–
- Take 1 ml of sample solution (carbohydrate solution or urine) in a clean test tube.
- Add 2 ml of Benedict’s reagent into the test tube.
- Heat the mixture in boiling water bath for 3–5 minutes. Direct heating can be done but slow heating in water bath is preferred to avoid rapid boiling.
- Remove the test tube and allow it to cool. Observe for colour change and precipitate formation.
- If solution remain clear blue then it is negative test (no reducing sugar). If colour changes to green yellow orange or brick red then it is positive test and colour with precipitate indicate approximate concentration of reducing sugar.
Observation and Results of Benedict’s Test
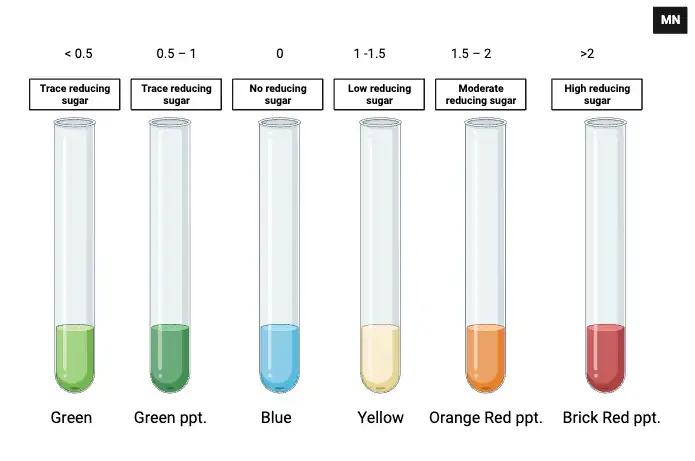
Observation and results of Benedict’s test are–
- Blue (no change) – Solution remains clear blue. It indicate negative test and 0% reducing sugar is present.
- Green solution/green precipitate – It indicate trace amount of reducing sugar. About 0.1% to 0.5% sugar is present.
- Yellow precipitate – It indicate low amount of reducing sugar. About 0.5% to 1.0% sugar is present.
- Orange or orange red precipitate – It indicate moderate amount of reducing sugar. About 1.0% to 2.0% sugar is present.
- Brick red precipitate – It indicate strong positive test. High amount of reducing sugar is present and it is more than 2.0%.
These visible result is obtained because reducing sugar reduces blue cupric ions (Cu2+) to cuprous ions (Cu+). Due to this insoluble cuprous oxide (Cu2O) precipitate is formed and colour changes according to concentration. Benedict’s test is semi quantitative and sugar level is estimated by colour gradient and density of precipitate.
Advantages of Benedict’s Test
Advantages of Benedict’s test are–
- It is simple and rapid test. Basic materials is required and result is obtained quickly by visible colour change.
- Benedict’s reagent is stable. Sodium citrate is present so copper ions is kept in solution and reagent can be stored as single ready to use solution for long time.
- It is safer to handle. The reagent is less corrosive because sodium carbonate is used instead of sodium hydroxide and the chemicals are relatively non toxic.
- It is cost effective. The chemicals is inexpensive and it is suitable for routine laboratory work and large number of samples.
- It gives qualitative and semi quantitative result. Presence of reducing sugar is detected and approximate concentration is estimated by colour change from green to brick red.
- It is highly sensitive. Gradual and distinct colour change is obtained so interpretation becomes easy.
Limitation of Benedict’s Test
Limitation of Benedict’s test are–
- It is not specific. It detects reducing substances but exact sugar cannot be identified like glucose and galactose. Other test is required for identification of specific carbohydrate.
- It is not fully quantitative. It gives only semi quantitative estimation based on colour change so exact numerical value is not obtained.
- Interpretation is subjective. Result depends on visual colour reading and it can vary due to observer difference lighting condition and pigments present in sample.
- False positive may occur. Non carbohydrate reducing agents can give colour change like high dose vitamin C (ascorbic acid) and drugs like penicillin streptomycin isoniazid salicylates (aspirin) and levodopa.
- Inhibitory interference is seen. High concentration of urine compounds like urate (uric acid) and creatinine can retard reaction and low glucose level may be masked.
- Non reducing sugars is not detected. Sucrose or trehalose do not react unless it is hydrolyzed first into monosaccharides by acid.
Uses of Benedict’s Test
Uses of Benedict’s test are–
- It is used for detection of reducing sugars in unknown sample. The sugars detected are glucose fructose and lactose.
- It is used in medical diagnosis. It is rapid presumptive test for diabetes mellitus by detecting glucose in urine sample.
- It is used in food science and quality control. It is used for checking simple sugars and monitoring sugar content in beverages and food products like honey.
- It is used in biochemical research. It is used for qualitative analysis differentiation and identification of unknown carbohydrate extracts.
- It is used for educational purpose. It is used in classroom and school laboratory to demonstrate redox reaction and chemical property of carbohydrates.
Precautions of Benedict’s Test
Precautions of Benedict’s test are–
- Measurement is done properly. Sample and reagent volume should be taken accurately.
- Proper PPE is used. Safety goggles gloves and lab coat/apron is worn because Benedict’s reagent contains toxic component and alkaline solution is mildly corrosive to skin and eyes.
- Hot test tubes is handled carefully. Test tube holder insulated gloves or tongs is used while heating and removing from water bath.
- Heating is done slowly and carefully. Water bath heating is preferred and rapid boiling/overheating is avoided because splattering can occur and result may be affected.
- Test tube mouth should not be directed towards self or others during heating.
- Droppers is used for taking reagent from bottle. Direct pouring should be avoided.
- Negative result should be confirmed. The mixture is heated at least three times before concluding negative test.
References
- Bender, C., Vestergaard, P., & Cichosz, S. L. (2025). The history, evolution and future of continuous glucose monitoring (CGM). Diabetology, 6(3), 17. https://doi.org/10.3390/diabetology6030017
- BYJU’S. (n.d.). Benedict’s test.
- Chemistry Learner. (n.d.). Barfoed’s test: Principle, procedure, reaction, and result.
- Cunningham, K. (2019). 2.6: Properties of monosaccharides. In CHEM 121: Concepts for a molecular view of biology II. Chemistry LibreTexts.
- Dahal, P. (2024, December 25). Benedict’s test: Principle, procedure & practical uses. Microbe Notes.
- Flinn Scientific. (n.d.). Benedict’s quantitative solution.
- GeeksforGeeks. (2025, July 23). Benedict’s test.
- Gpatindia. (2025, March 31). Carbohydrate identification chemical tests, notes, lecture and MCQ with answer.
- Hernández-López, A., Sánchez Félix, D. A., Zuñiga Sierra, Z., García Bravo, I., Dinkova, T. D., & Avila-Alejandre, A. X. (2020). Quantification of reducing sugars based on the qualitative technique of Benedict. ACS Omega, 5(50), 32403–32410. https://doi.org/10.1021/acsomega.0c04467
- HiMedia Laboratories. (2022). R003 Benedict’s quantitative reagent.
- Jensen, W. B. (2014). Stanley Rossiter Benedict. Museum Notes, 1-4.
- Simoni, R. D., Hill, R. L., & Vaughan, M. (2002). Benedict’s solution, a reagent for measuring reducing sugars: the clinical chemistry of Stanley R. Benedict. Journal of Biological Chemistry, 277(16), e5-e6. https://doi.org/10.1016/S0021-9258(19)61050-1
- Singla, R. (n.d.). Benedict’s test: Detecting reducing sugars in chemistry and biology. Vedantu.
- Study Mind. (n.d.). Tests for carbohydrates (A-level biology).
- Sweeney, M. (2024). A review: Biosensor progression in glucose monitoring for patients with diabetes. Advances in Bioscience and Biotechnology, 15(8), 503-510. https://doi.org/10.4236/abb.2024.158031
- Testbook. (n.d.). Fehling test – Reaction, reagent, mechanism, and difference between Fehling’s and Benedict’s test.
- Wikipedia contributors. (2026, March 2). Benedict’s reagent. In Wikipedia, The Free Encyclopedia.