Membrane filtration method is a laboratory method which is used for estimation of microbial load (bioburden) in liquid sample like drinking water environmental water and pharmaceutical solution. It is a quantitative method and the bacteria are concentrated on a membrane filter and then counted as colony forming unit (CFU).
Principle of this method is based on physical trapping of microorganism by microporous membrane. A measured volume of sample is passed through membrane filter (generally 0.45 µm pore size) with the help of vacuum or pressure difference. The pore size is smaller than bacteria so the organisms are retained on surface of membrane and they are uniformly distributed.
After filtration the membrane is transferred carefully on selective and differential culture medium (solid agar plate or absorbent pad soaked with medium). Then it is incubated at suitable temperature for specific time (about 24 hours). The trapped bacteria uses nutrient and they multiply to form visible colonies on membrane surface.
After incubation colonies are observed and they are identified by colour or metallic sheen and counted. The number of colonies is taken as CFU and it gives bacterial density of original sample. This method is rapid accurate and it is widely used for water testing and sterility monitoring.
Principle of Membrane Filter (MF) Method
Principle of membrane filter (MF) method is based on physical separation and cultivation of microorganism from liquid sample. In this method a measured volume of water sample is passed through a microporous membrane filter by using pressure difference (vacuum pump or aspirator). The standard pore size of membrane is about 0.45 µm and it is smaller than common indicator bacteria so membrane acts as mechanical barrier. Water is passed through but bacterial cells are trapped and concentrated uniformly on surface of membrane.
After filtration the membrane is transferred aseptically on selective and differential culture medium (agar plate or absorbent pad saturated with nutrient broth) with bacteria side up. The membrane acts as nutrient permeable interface so nutrients diffuse from medium to trapped cells. During incubation selective agents suppress non target organisms and differential indicators allow target bacteria to metabolize and form characteristic colonies.
After incubation colonies are identified by characteristic appearance and they are counted manually. The number of colony forming unit (CFU) is used to calculate bacterial density in original sample.
Requirements for Membrane Filtration Method
- Filtration apparatus– Filter holding assembly is required (funnel and porous base or receptacle). It is made of borosilicate glass stainless steel autoclavable plastic or porcelain.
- Vacuum source– Vacuum pump or water aspirator or hand pump is used to create pressure difference for filtration.
- Membrane filter– Sterile microporous membrane filter is required (generally 47 mm diameter and 0.45 µm pore size). Grid marked filter and mixed cellulose ester (MCE) type is commonly used.
- Forceps– Smooth flat round tip forceps is used for handling membrane. It is sterilized by dipping in alcohol and flaming before use.
- Petri dish– Sterile petri dishes are required (about 60×15 mm or 50×12 mm) with tight lid to maintain moisture.
- Absorbent pad– Sterile sulfite free absorbent pad is used (it absorbs about 2 ml broth) when agar plate is not used.
- Culture media– Selective and differential media is required (m-Endo for total coliform mFC for fecal coliform Chromogenic coliform agar etc).
- Incubator– Incubator or water bath is required to maintain temperature (35 ± 0.5°C for total coliform and 44.5 ± 0.2°C for fecal coliform).
- Magnification and light– Low power dissecting microscope (10×–15×) and cool white light source is used for observing metallic sheen and counting colonies.
- Sterile dilution and rinse water– Buffered dilution water (PBS or peptone water) is used for dilution and for rinsing funnel wall so all bacteria are washed on membrane.
- Measuring and sterilization items– Sterile pipette and measuring cylinder are required. Autoclave hot air oven or UV chamber is used for sterilization of apparatus and glassware.



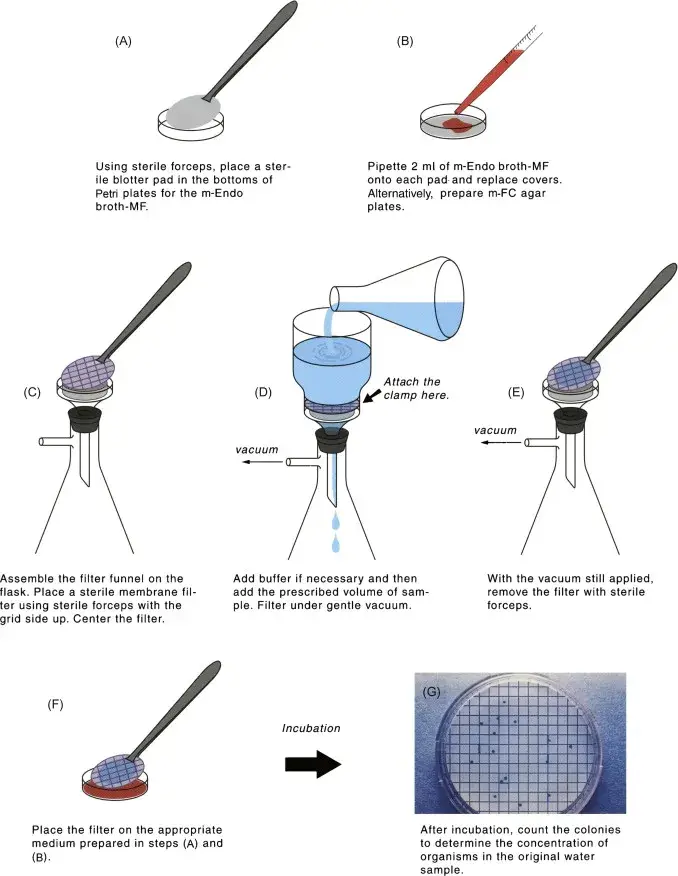
Procedure

- Preparation– Work bench is disinfected and hands are washed properly. Forceps is sterilized by dipping in alcohol and passing through flame and it is cooled before use.
- Apparatus setup– Sterile filtration apparatus is assembled. By sterile forceps membrane filter (0.45 µm) is placed on centre of vacuum support with grid side up. Sterile funnel is fixed tightly over receptacle.
- Sample addition– Water sample is mixed well by shaking or inverting about 25 times so bacteria is distributed evenly. Specific volume of sample is poured in funnel (generally 100 ml for drinking water). If sample volume is less than 20 ml then 10–20 ml sterile dilution water is added first before applying vacuum.
- Filtration and rinsing– Vacuum is applied and sample is drawn completely through membrane. Funnel wall is rinsed 2–3 times with sterile dilution water (about 20–30 ml each time) so all bacteria are washed on filter. Vacuum is released when filter becomes dry to avoid damage.
- Membrane transfer– Funnel is removed and membrane is lifted by sterile forceps. Membrane is transferred immediately on selective culture medium in petri dish (agar plate or absorbent pad saturated with broth). It is placed with gentle rolling so proper contact is made and air bubble is avoided.
- Incubation– Petri dish lid is closed and dish is inverted. It is incubated at required temperature and time (35 ± 0.5°C for 22–24 hours for total coliform or 44.5°C for fecal coliform).
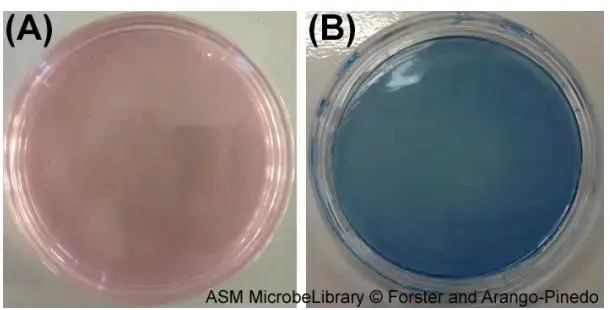
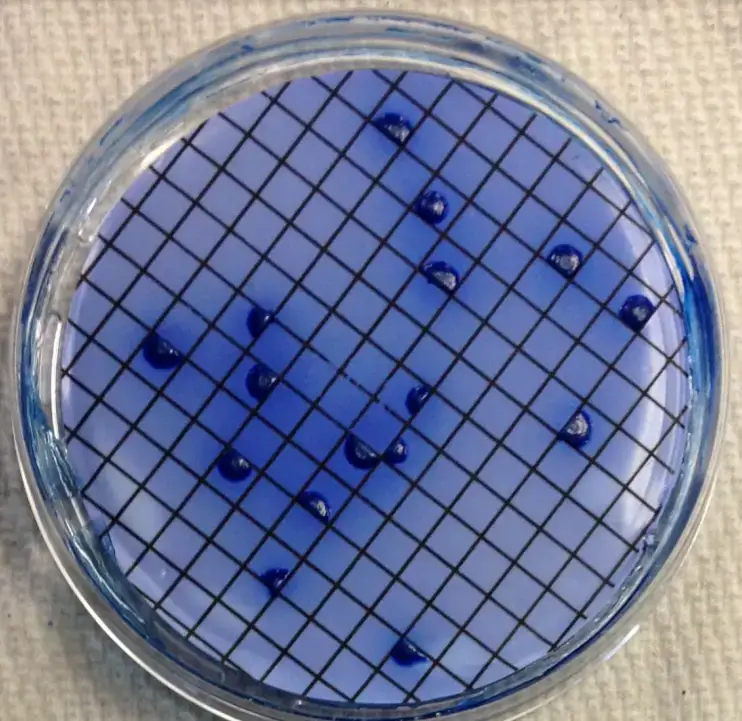
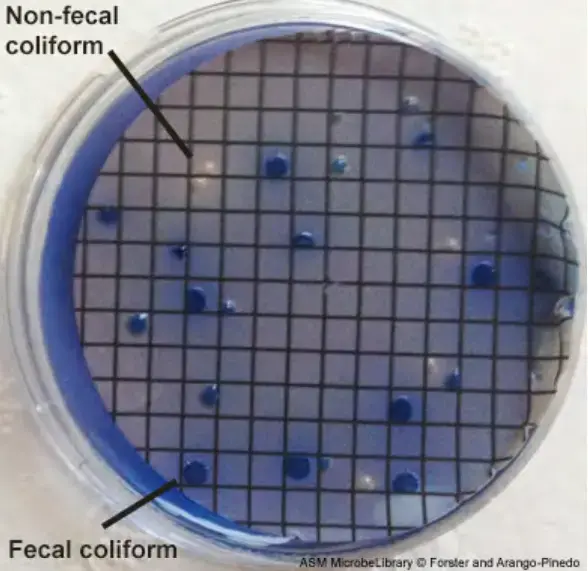
- Enumeration and calculation– After incubation membrane is examined by illuminated magnifier or low power microscope (10×–15×). Characteristic colonies are identified (red colony with greenish gold metallic sheen for total coliform) and colonies are counted. The count is expressed as colony forming unit (CFU) per 100 ml of original water sample.

Calculations
Calculating the cell density of the original sample:
- The original density is approximated based on the amount of filtered sample (or the volume of dilution and the dilution factor) and the number of colonies enumerated on the membrane. As counts are provided per 100 ml of sample rather than per ml, the per-ml numbers must be multiplied by 100.
- The best range for calculations is between 20 and 80 total coliforms (with no more than 200 total colonies) each plate. For calculations, counts between 20 and 60 faecal coliforms per plate are optimum.
To calculate density (CFU per 100 ml):
Density = number of colonies on membrane x 100/volume (ml) of undiluted sample filtered
Or if the sample was diluted and a volume of the dilution was filtered:
Original Density = number of colonies on membrane x 100/(volume (ml) filtered x dilution)
Uses of Membrane Filter (MF) Technique
- Drinking and bottled water– It is used for monitoring microbiological safety of municipal drinking water private well groundwater spring and bottled water and for regulatory compliance.
- Recreational water– It is used to assess sanitary condition of bathing beach swimming pool and natural recreational water for prevention of water borne disease.
- Wastewater and sewage– It is used to evaluate purification and wastewater treatment process by testing raw sewage chlorinated effluent and treated sewage sludge.
- Environmental water testing– It is used for lakes rivers streams estuaries coastal water and storm water runoff to detect microbial load and fecal contamination.
- Food and beverage processing– It is used to indicate sanitary condition in food processing and it is used for liquid foods citrus juice and beverages (low particulate).
- Shellfish harvest monitoring– It is used to test harvest water and shellfish meat (bivalve molluscan) for sanitary and consumption standard.
- Agricultural irrigation water– It is used to assess microbiological safety of irrigation water (creek water roof harvested rainwater secondary treated wastewater farm pond etc).
- Emergency and field testing– It is used in disaster relief remote area and military or civilian camps because it gives quantitative result with simple setup.
- Detection of indicator organism– It is used for estimation of total coliform E. coli intestinal enterococci Pseudomonas aeruginosa and Clostridium perfringens etc (regulatory indicator organisms).
Advantages of Membrane Filter (MF) Technique
- Rapid and quantitative– It gives direct numerical result in short time (generally within 24 hours) and bacteria is counted accurately as CFU.
- High sensitivity– Large volume of water can be processed (up to 1 liter or more) so detection limit is low and trace contamination can be detected.
- Reliable count– Colony count obtained by MF method gives higher precision and less variability compared to MPN method so statistical reliability is more.
- Simple and cost effective– Procedure is simple and sophisticated equipment is not required. It is low cost method for routine testing.
- Less space and material– Large volume sample is tested on single membrane in one petri dish so it requires less glassware and less incubator space than multiple tube method.
- Viable colonies obtained– Discrete viable colonies are formed on membrane so it can be inspected and isolated for further confirmation (species test virulence or antibiotic resistance).
- Background flora observation– By visual inspection overall level of non target bacteria can be seen so it gives idea about background flora in sample.
Limitations of Membrane Filter (MF) Technique
- Clogging problem– If sample has high suspended solid algae or food particles then membrane pores are clogged quickly. Due to this large volume cannot be filtered and smaller volume is taken so statistical error is increased and sensitivity is reduced.
- Time consuming– Incubation time is long (about 18–96 hours) and if confirmation is needed then more time is required. So it is not very rapid for public health decision like beach advisory.
- Background bacteria interference– If non target bacteria (heterotrophs) are high then overgrowth occurs on membrane. It suppresses target organism and colony features (metallic sheen) is obscured so bacterial density may be underestimated.
- Toxic substance effect– Heavy metals like copper zinc and other toxic chemical may be concentrated on membrane and it inhibits growth so false low coliform count is obtained (neutralizer like EDTA is required).
- Injured cell recovery– It is less effective than MPN broth method for recovery of injured or stressed bacteria (chlorine damaged cell or marine stress).
- False positive colonies– Some non coliform bacteria may produce byproduct and it can react with indicator dye and colonies may mimic target colony (false metallic sheen on m-Endo).
- Manual counting error– Colony counting is laborious subjective and time consuming. Technician variability subtle colour difference and confluent growth reduces accuracy.
References
- American Public Health Association, American Water Works Association, & Water Environment Federation. (1999). 9222 B. Standard total coliform membrane filter procedure. In Standard methods for the examination of water and wastewater.
- Best, S., Rediske, R., & Lane, M. (2018). A comparison study of Colilert and qPCR methods at Pere Marquette Beach, Muskegon County, MI. ScholarWorks@GVSU.
- Buckalew, D. W., Hartman, L. J., Grimsley, G. A., & Martin, A. E. (2006). A long-term study comparing membrane filtration with Colilert defined substrates in detecting faecal coliforms and Escherichia coli in natural waters. Journal of Environmental Management, 80(3), 191–197. https://doi.org/10.1016/j.jenvman.2005.08.024
- Cao, L., Zeng, L., Wang, Y., Cao, J., Han, Z., Chen, Y., Wang, Y., Zhong, G., & Qiao, S. (2024). U2-Net and ResNet50-based automatic pipeline for bacterial colony counting. Microorganisms, 12(1), 201. https://doi.org/10.3390/microorganisms12010201
- Chemie Brunschwig / Laboratorios Conda. (n.d.). Microbiological analysis.
- Comprehensive analysis of membrane filtration methodologies in the bacteriological examination of water. (n.d.).
- Eckner, K. F. (1998). Comparison of membrane filtration and multiple-tube fermentation by the Colilert and Enterolert methods for detection of waterborne coliform bacteria, Escherichia coli, and enterococci used in drinking and bathing water quality monitoring in southern Sweden. Applied and Environmental Microbiology, 64(8), 3079–3083.
- Fecal coliform and E. coli analysis in wastewater and water by Quanti-Tray, Method 9223 B. (n.d.).
- Feng, P., Weagant, S. D., Grant, M. A., & Burkhardt, W. (2020). Bacteriological analytical manual (BAM) chapter 4: Enumeration of Escherichia coli and the coliform bacteria. U.S. Food and Drug Administration.
- Florida Spectrum Environmental. (n.d.). Microbiological fact sheet SM9222D fecal coliform membrane filter procedure.
- Forster, B., & Arango Pinedo, C. (2015, June 23). Bacteriological examination of waters: Membrane filtration protocol. American Society for Microbiology.
- Francis, M. R., Sarkar, R., Roy, S., Jaffar, S., Mohan, V. R., Kang, G., & Balraj, V. (2016). Effectiveness of membrane filtration to improve drinking water: A quasi-experimental study from rural southern India. American Journal of Tropical Medicine and Hygiene, 95(5), 1192–1200. https://doi.org/10.4269/ajtmh.15-0675
- Hach Company. (n.d.). Coliforms—Total, fecal and E. coli.
- Hach Company. (n.d.). Membrane filtration guidelines.
- Illinois General Assembly. (n.d.). Section 465.360 Methodology. Administrative Code, Title 77, Part 465.
- International Organization for Standardization. (1984). Water quality – Detection and enumeration of faecal streptococci – Part 2: Method by membrane filtration (ISO 7899-2:1984).
- International Organization for Standardization. (2000). Water quality – Detection and enumeration of intestinal enterococci – Part 2: Membrane filtration method (ISO 7899-2:2000).
- International Organization for Standardization. (2013). Water quality – Enumeration of Clostridium perfringens – Method using membrane filtration (ISO 14189:2013).
- International Organization for Standardization. (2023). Water quality – Requirements for the performance testing of membrane filters used for direct enumeration of microorganisms by culture methods (ISO 7704:2023).
- Kemper, M. A., Veenman, C., Blaak, H., & Schets, F. M. (2023). A membrane filtration method for the enumeration of Escherichia coli in bathing water and other waters with high levels of background bacteria. Journal of Water & Health, 21(8), 995–1003. https://doi.org/10.2166/wh.2023.004
- Mahmoud, R., Rapti, I., & Pil, D. (n.d.). Enhancing water testing standards for a safer tomorrow. Thermo Fisher Scientific.
- Marotz, J., Lübbert, C., & Eisenbeiss, W. (2001). Effective object recognition for automated counting of colonies in Petri dishes (automated colony counting). Computer Methods and Programs in Biomedicine, 66(2-3), 183–198. https://doi.org/10.1016/S0169-2607(00)00128-0
- Maruf, M. H., Islam, M. H., Mahmud, H., & Sunny, S. K. (2025). Image processing-based automated detection and counting of coliform colonies on Petri dishes for water quality analysis. International Conference on Civil Engineering, Research and Innovations.
- Mayer, M. J. (2014, December 9). Updated ISO guideline recommends chromogenic coliform agar for water quality analysis. Thermo Fisher Scientific.
- McNair, J. N., Rediske, R. R., Hart, J. J., Jamison, M. N., & Briggs, S. (2025). Performance of Colilert-18 and qPCR for monitoring E. coli contamination at freshwater beaches in Michigan. Environments, 12(1), 21. https://doi.org/10.3390/environments12010021
- MilliporeSigma / Sigma-Aldrich. (2018). Chromocult® Coliform Agar acc. ISO 9308-1.
- My Life Science Career. (2026, March 10). Membrane filtration method.
- NEMI. (n.d.). Standard methods: 9222D: Membrane filtration test for fecal coliforms.
- National Health and Medical Research Council. (2011). Australian drinking water guidelines 6: Intestinal enterococci.
- Neogen. (n.d.). Automation in colony counting | Modern microbial enumeration methods.
- Noble, R. T., Blackwood, A. D., Griffith, J. F., McGee, C. D., & Weisberg, S. B. (2010). Comparison of rapid quantitative PCR-based and conventional culture-based methods for enumeration of Enterococcus spp. and Escherichia coli in recreational waters. Applied and Environmental Microbiology, 76(22), 7437–7443. https://doi.org/10.1128/AEM.00651-10
- Rapidmicrobiology. (2024, December 10). The revised EN ISO 7704:2023 is advancing water quality analysis, is your lab keeping up?
- Rapidmicrobiology. (n.d.). Water – Microbiological analysis test method guide.
- Rodrigues, P. M., Luís, J., & Tavaria, F. K. (2022). Image analysis semi-automatic system for colony-forming-unit counting. Bioengineering, 9(7), 271. https://doi.org/10.3390/bioengineering9070271
- Sartorius. (n.d.). Enumeration of Clostridium perfringens — Microbiological performance testing of membrane filters in accordance with EN ISO 7704:2023.
- Schiff, L. J., Morrison, S. M., & Mayeux, J. V. (1970). Synergistic false-positive coliform reaction on M-Endo MF medium. Applied Microbiology, 20(5), 778–781.
- Tiwari, A., Hokajärvi, A.-M., Santo Domingo, J. W., Kauppinen, A., Elk, M., Ryu, H., Jayaprakash, B., & Pitkänen, T. (2018). Categorical performance characteristics of method ISO 7899-2 and indicator value of intestinal enterococci for bathing water quality monitoring. Journal of Water and Health, 16(5), 711–723. https://doi.org/10.2166/wh.2018.293
- Turnbull, A. (2024, March 15). 1.9: Bacteriological analysis of water. Biology LibreTexts.
- U.S. Environmental Protection Agency. (1986). Method 9132: Total coliform: Membrane-filter technique.
- Veolia. (n.d.). What is membrane filtration? How it works & benefits.