Bacitracin Susceptibility Test is a screening test used in microbiology to differentiate Group A beta hemolytic streptococci from other beta hemolytic streptococci. It is mainly used for presumptive identification of Streptococcus pyogenes (Group A streptococcus).
It is also used to differentiate Micrococcus species from Staphylococcus species. Micrococcus is usually susceptible to bacitracin and Staphylococcus is usually resistant.
In this test, a paper disc containing a very low concentration of bacitracin (0.04 units) is used. The disc is placed on blood agar plate which is inoculated with the test organism. If the organism is susceptible, a clear zone of inhibition is seen around the disc. If the organism is resistant, no zone is formed and growth occurs up to the edge of the disc.
Objectives of Bacitracin Susceptibility Test
- Objectives of Bacitracin Susceptibility Test
- To do presumptive identification of Group A beta hemolytic streptococci (Streptococcus pyogenes).
- To differentiate Group A streptococci from other beta hemolytic streptococci.
- To differentiate Micrococcus species from Staphylococcus species, because Micrococcus is susceptible to bacitracin and Staphylococcus species are resistant.
- To observe the susceptibility pattern of different organisms against bacitracin by zone of inhibition around the disc.
Principle of Bacitracin Susceptibility Test
Bacitracin Susceptibility Test is based on the selective ability of bacitracin antibiotic to inhibit the bacterial cell wall synthesis in highly sensitive organisms. Bacitracin is a polypeptide antibiotic that blocks the transport of NAM and NAG sugars by inhibiting bactoprenol (lipid carrier) across the bacterial cell membrane. Due to this, peptidoglycan formation is disrupted and cell wall synthesis is inhibited.
In this test, a filter paper disc impregnated with a very low concentration of bacitracin (0.04 units) is placed on the agar plate which is heavily inoculated with the suspected organism. The bacitracin diffuses into the agar medium around the disc. If the organism is susceptible (most commonly Group A beta-hemolytic streptococci, Streptococcus pyogenes), growth is inhibited and a clear zone of inhibition is formed around the disc.
If the organism is resistant (Group B streptococci or Staphylococcus species), such trace amount of bacitracin does not affect the growth. Therefore, there will be no zone and the organisms grow up to the edge of the disc.
Requirements
- Pure culture of suspected beta hemolytic organism.
- Blood agar plate (5% sheep blood) or Mueller Hinton agar.
- Bacitracin disc (0.04 unit or 0.05 unit).
- Sterile saline/broth for making suspension (0.5 McFarland).
- Sterile swab. Sterile loop.
- Forceps (sterile / heated) for placing disc.
- Incubator 35°C to 37°C (CO2 5% to 10% can be used).
- Quality control strains- Streptococcus pyogenes ATCC 19615 (positive). Streptococcus agalactiae ATCC 13813 / ATCC 27956 (negative).
Procedure of Bacitracin test
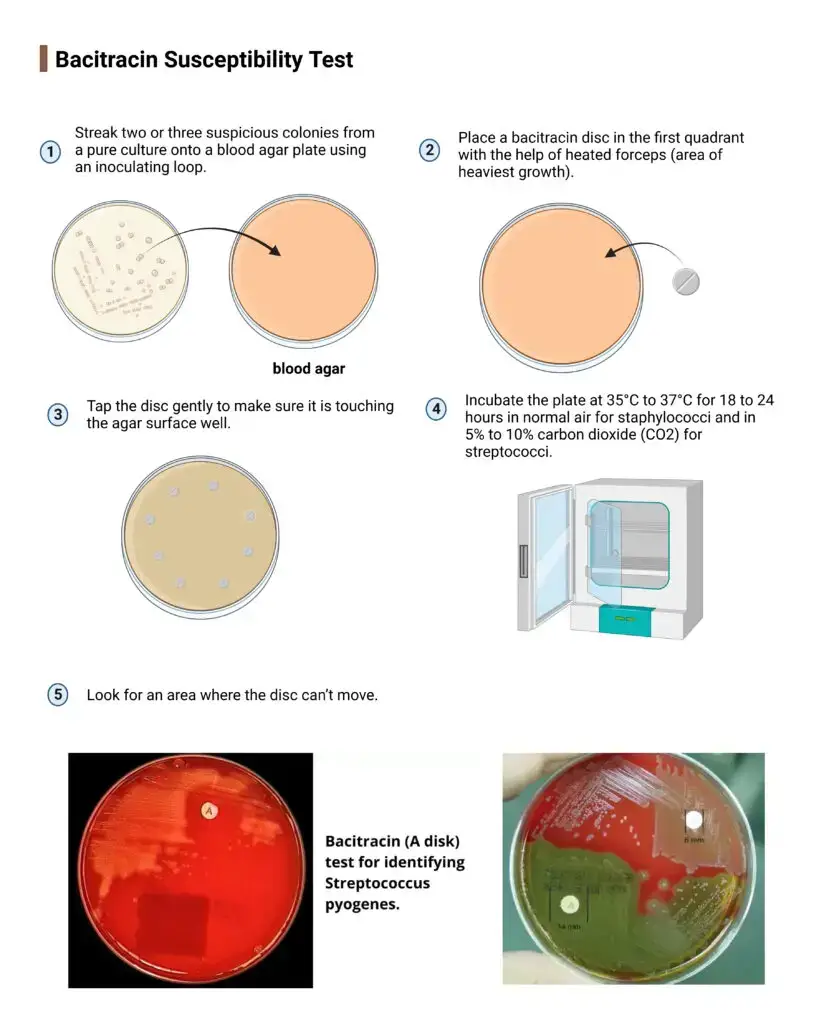
- Select 2 to 4 well isolated colonies from a pure culture (18 to 24 hours old).
- If needed, prepare suspension in sterile saline/broth and adjust to 0.5 McFarland.
- Take a blood agar plate (5% sheep blood agar) and label it.
- Dip a sterile swab in the suspension and remove excess.
- Streak the organism on the plate in multiple directions to make heavy confluent growth (lawn culture).
- Allow the plate to stand for 5 to 10 minutes so that the surface gets dried.
- Using sterile/heated forceps place bacitracin disc (0.04 unit) on the center of the inoculated area.
- Press/tap the disc gently so that it makes full contact with agar surface.
- Invert the plate and incubate at 35°C to 37°C for 18 to 24 hours.
- For streptococci, incubate in 5% to 10% CO2 if available.
- After incubation, observe the zone of inhibition around the disc.
- Measure the zone if present and report as susceptible. If no zone, report as resistant.

Result of Bacitracin Susceptibility Test
1. Positive (Susceptible) Result
Observation- A clear zone of inhibition is formed around the bacitracin disc and bacterial growth is prevented around the disc. It is taken as any visible zone or a zone more than 10 mm or zone ≥14 mm or ≥15 mm (based on the lab standard used).
Interpretation- It indicates presumptive positive for Group A beta hemolytic Streptococcus (Streptococcus pyogenes). In other gram positive cocci differentiation, it indicates Micrococcus species rather than Staphylococcus.
2. Negative (Resistant) Result
Observation- No zone of inhibition is formed. The organism grows up to the edge of the bacitracin disc. In some guidelines, zone less than 14 mm is also taken as resistant.
Interpretation– It indicates non Group A beta hemolytic Streptococci like Group B Streptococci (Streptococcus agalactiae). In other gram positive cocci differentiation, it indicates Staphylococcus species.
3. Indeterminate (Probable Susceptibility) Result
Observation– A small zone of inhibition is seen between 6 mm to 10 mm.
Interpretation– The result is not confirm. The test should be repeated or it should be confirmed by other methods like PYR test or serological grouping.
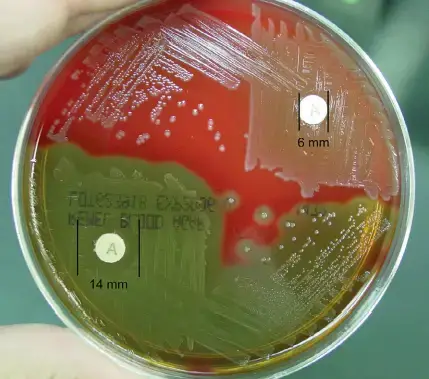
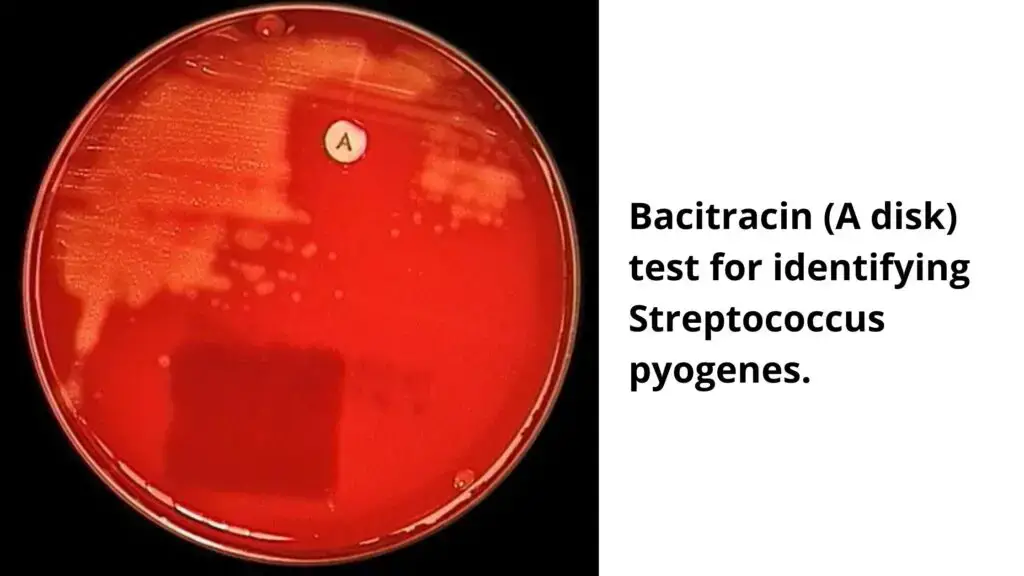
Organisms gives positive and negative result in Bacitracin Susceptibility Test (0.04 unit disc)
1. Positive (Susceptible) organisms
- Group A beta hemolytic Streptococci- mainly Streptococcus pyogenes.
- Micrococcus species- highly susceptible.
- Alpha hemolytic Streptococci- many strains including Streptococcus pneumoniae may show susceptibility.
- Some Group C and Group G Streptococci- up to 10% can be susceptible. Streptococcus equi (many are sensitive) and Streptococcus zooepidemicus (many are sensitive) can give positive.
- Some Group B Streptococci- about 5% to 6% strains can show susceptibility.
2. Negative (Resistant) organisms
- Group B beta hemolytic Streptococci- mainly Streptococcus agalactiae.
- Staphylococcus species- coagulase negative staphylococci and Staphylococcus aureus are usually resistant.
- Most non Group A beta hemolytic Streptococci- like Streptococcus anginosus (Group G) and Streptococcus equisimilis (Group C) are resistant.
- Haemophilus influenzae– no effect, it gives resistant type result (no zone).
Quality Control
1. Positive controls (Susceptible)
- Streptococcus pyogenes ATCC 19615 (Group A Streptococcus)- This is the primary positive control strain. It must show a clear zone of inhibition around the bacitracin disc.
- Staphylococcus epidermidis ATCC 12228- It is used as an additional sensitive control organism. It will show a zone of inhibition.
2. Negative controls (Resistant)
- Streptococcus agalactiae (Group B Streptococcus)- Strains like ATCC 13813, ATCC 27956 and ATCC 12386 are used as negative controls. It shows no zone of inhibition and growth is seen up to the edge of the disc.
- Haemophilus influenzae ATCC 10211- It is used as an additional resistant control organism. It will give no zone of inhibition.
Uses of Bacitracin Susceptibility Test
- It is used for presumptive identification of Group A beta hemolytic Streptococci. It is used as a primary screening test to identify Streptococcus pyogenes from clinical samples like throat swab.
- It is used for differentiation of beta hemolytic Streptococci. It helps to differentiate Group A Streptococci (bacitracin susceptible) from non Group A beta hemolytic Streptococci (bacitracin resistant).
- It is used to differentiate Micrococcus and Staphylococcus species. Micrococcus is highly susceptible and Staphylococcus is resistant.
- It is used for sub classification of other gram positive cocci. It helps in categorization of some bacteria like penicillin susceptible strains like Rothia.
- It is used to distinguish some specific bacterial species when combined with other tests. It helps in differentiating Aerococcus viridans and Streptococcus milleri from enterococci and Streptococcus mitis.
- It is used in general antibiotic susceptibility testing. It is used to check resistance or susceptibility pattern of different microorganisms to bacitracin.
Limitations of Bacitracin Susceptibility Test
- Cross sensitivity- This test is not fully specific. More than 10% of Group C and Group G streptococci and about 5% to 6% of Group B strains may also show susceptibility. It can give false positive as Group A Streptococci.
- It is only for beta hemolytic strains- The test is valid only for beta hemolytic Streptococci. Alpha hemolytic strains (like Streptococcus pneumoniae) can give misleading result because they are naturally susceptible to low concentration of bacitracin.
- Emerging resistance- Some Group A Streptococci clones are developing bacitracin resistance. So resistant result does not fully rule out Streptococcus pyogenes.
- Inoculum density error- If inoculum is too light, false zone may be seen. If inoculum is too heavy, the antibiotic concentration may be overcome and susceptible organism can look resistant. Mixed culture will make the result unreliable.
- Media quality dependence- Old and dried blood agar plate can reduce diffusion of bacitracin. Zone size becomes smaller and false negative (resistant) result may be obtained.
- Environmental and pH sensitivity- Some strains of Streptococcus pyogenes may show poor growth or poor hemolysis if incubated in ambient air instead of CO2 enriched condition. Agar pH should be maintained around 7.2 to 7.4, if not maintained bacitracin activity is affected and zone measurement will change.
- Disk concentration mix up- If 10 unit susceptibility disc is used instead of 0.04 unit differentiation disc, almost all beta hemolytic Streptococci will be inhibited. The test will not differentiate properly.
- Presumptive nature- It gives only presumptive identification. It should be confirmed by other tests like PYR test or serological grouping.
Advantages of Bacitracin Susceptibility Test
- Cost effective- It is low cost and gives good cost benefit. It is used as an affordable alternative to serological antisera, automated identification systems and molecular assays. It is useful in resource limited labs.
- Time and labor saving- It is used as a preliminary screening test. It saves time, materials and labor when compared to other complex diagnostic methods.
- Simple and convenient- The disc diffusion method is simple. Steps are easy and it is convenient for routine diagnostic work in clinical laboratory.
- Rapid clinical insight- It gives quick presumptive result. It helps in early therapeutic decision and timely antibiotic treatment.
- Effective differentiation- It differentiates Group A beta hemolytic Streptococci from other streptococcal groups. It is also used to differentiate susceptible Micrococcus from resistant Staphylococcus species.
Precautions of Bacitracin Susceptibility Test
- It is for in vitro diagnostic use only. The test should be done by trained and qualified laboratory personnel only.
- Aseptic technique should be followed. Standard biohazard precautions should be maintained and PPE like gloves and eye protection should be used while handling cultures.
- All contaminated materials and biohazard waste should be decontaminated properly. Sterilization is done by autoclaving or incineration before disposal.
- Bacitracin disc storage- Discs should be stored as per instruction (usually 2°C to 8°C or frozen at -20°C). It should be protected from light and moisture. Expired discs should not be used.
- Media freshness- Freshly prepared media should be used. Old or dried blood agar plate will reduce diffusion of antibiotic and can give false negative (resistant) result.
- Media pH- Agar pH should be maintained between 7.2 to 7.4. Wrong pH can reduce bacitracin activity and zone size will be affected.
- Correct disc concentration should be used- Differential disc of bacitracin (0.04 or 0.05 unit) should be used. High concentration disc like 10 unit should be avoided because it inhibits most beta hemolytic streptococci and result becomes misleading.
- Test is done only for appropriate isolates- It should be used only for pure culture of β hemolytic streptococci. Alpha hemolytic streptococci (like Streptococcus pneumoniae) should not be tested because it is naturally susceptible and gives error.
- Inoculum should be standard- Heavy inoculum should be taken to get confluent growth (around 0.5 McFarland). Light inoculum can give false large zone (false susceptible). Very heavy inoculum can give false resistant result.
- Confirmatory test should be used- Due to cross sensitivity in some Group B, C and G streptococci, this test should not be used alone. Presumptive positive should be confirmed by serological grouping or biochemical test like PYR test.
References
- Abbas, N. H. (n.d.). Streptococci (Lab. 3).
- Bacitracin-resistant clone of Streptococcus pyogenes isolated from pharyngitis patients in Belgium. (n.d.). PubMed Central (PMC).
- Bio-Rad. (2003, November). BACITRACIN 53842 presumptive diagnosis of group A streptococci.
- Chauhan, A. (2025, November 12). Bacitracin sensitivity test: Uses and interpretation guide. Flabs.
- Clinical and biochemical foundations of the bacitracin susceptibility test: A comprehensive diagnostic analysis. (n.d.).
- Dalynn Biologicals. (2014, October). Bacitracin disks (Catalogue No. DB10).
- Differentiation of Staphylococcus and Micrococcus spp. with the Taxo A bacitracin disk. (n.d.). PubMed Central (PMC).
- Hardy Diagnostics. (2020). HardyDisk™ bacitracin differentiation disks – for group A Strep identification.
- HiMedia Laboratories Pvt. Ltd. (2023, January). Bacitracin (50 discs/vl) – Technical data.
- Jelinková, J., & Rotta, J. (1967). Appendix 3: The bacitracin test for recognition of group A streptococci. International Journal of Systematic and Evolutionary Microbiology, 17(3), 297. https://doi.org/10.1099/00207713-17-3-297
- Kaplan, N. M., Khader, Y. S., & Ghabashineh, D. M. (2022). Laboratory diagnosis, antimicrobial susceptibility and genuine clinical spectrum of Streptococcus anginosus group; Our experience at a university hospital. Medical Archives, 76(4), 252–258. https://doi.org/10.5455/medarh.2022.76.252-258
- Mast Group Ltd. (n.d.). MASTDISCS® ID bacitracin discs.
- MilliporeSigma. (2018). 08382 Bacitracin disks. Merck KGaA.
- Pokhrel, P. (2015, September 30). Bacitracin test- Principle, procedure, result with limitation. Microbiology Notes.
- Sapkota, A. (2022, January 27). Bacitracin susceptibility test- Principle, procedure, results, uses. Microbe Notes.
- Spellerberg, B., & Brandt, C. (2016). Laboratory diagnosis of Streptococcus pyogenes (group A streptococci). In J. J. Ferretti, D. L. Stevens, & V. A. Fischetti (Eds.), Streptococcus pyogenes: Basic biology to clinical manifestations. University of Oklahoma Health Sciences Center.
- SXT and Taxo A disks for presumptive identification of group A and B streptococci in throat cultures. (n.d.). PubMed Central (PMC).
- Tille, P. M. (2025). Laboratory diagnosis of Staphylococcus, Micrococcus, and similar organisms. In Bailey & Scott’s Diagnostic Microbiology (13th ed., pp. 235-242).
- Variations in bacitracin susceptibility observed in Staphylococcus and Micrococcus species. (n.d.). PubMed Central (PMC).
- VUMIE. (2022, June 5). Bacitracin susceptibility test. Virtual Microbiology Lab Simulator Software.
- Williams, G. S. (2003). Group C and G streptococci infections: Emerging challenges. Clinical Laboratory Science, 16(4), 209-213.