IMViC test is a group of four biochemical tests which are used in microbiology laboratory to identify and differentiate Gram-negative bacteria especially coliform group and Enterobacteriaceae family. These tests are commonly performed after Gram staining and the reactions are stable and used as important identification tests.
IMViC is an acronym in which I stands for Indole test, M stands for Methyl red test, V stands for Voges-Proskauer test and C stands for Citrate utilization test. The small “i” is included to represent the word “in” as in Citrate.
Each test checks different metabolic ability of bacteria. Indole test is used to detect the ability of organism to produce indole from tryptophan breakdown. Methyl red test is used to detect production of stable mixed acids from glucose metabolism.
Voges-Proskauer test is used to determine the production of neutral end product acetoin. Citrate test is used to determine the ability of organism to utilize citrate as the sole carbon source.
The pattern of positive and negative results of these four tests is used to differentiate and identify different enteric bacteria and pathogens.
Principle of IMViC Test
IMViC test works on the principle of four biochemical reactions which are used to identify and differentiate Gram-negative enteric bacteria (coliforms). These tests are Indole, Methyl red, Voges-Proskauer and Citrate test and each test detects a specific metabolic pathway or enzyme activity.
Indole test is based on the ability of bacteria to produce tryptophanase enzyme which breaks tryptophan into indole, pyruvic acid and ammonia. After incubation Kovac’s reagent or Ehrlich’s reagent is added and a cherry red/pink ring is formed which indicates positive indole test.
Methyl red (MR) test works on the principle of mixed-acid fermentation of glucose. In this process stable organic acids are produced which lowers the pH upto 4.4 or below and methyl red indicator turns bright red showing positive MR test.
Voges-Proskauer (VP) test works on the principle of butanediol fermentation pathway of glucose in which a neutral intermediate acetoin (acetylmethylcarbinol) is produced. After adding Barritt’s reagents (alpha-naphthol and potassium hydroxide) acetoin is oxidized to diacetyl and a pink-red colour complex is formed which indicates positive VP test.
Citrate test works on the principle of citrate utilization as the sole carbon and energy source by enzyme citrase. During citrate and ammonium salt utilization alkaline byproducts such as sodium carbonate and ammonium hydroxide is produced which raises pH above 7.6 and bromothymol blue indicator changes from forest green to deep Prussian blue showing positive citrate test.
Objectives of IMViC test
- To identify coliform bacteria which are Gram-negative rods and these are aerobic or facultative anaerobic organisms.
- To differentiate different genera of Enterobacteriaceae family such as Escherichia, Salmonella, Shigella, Klebsiella and Proteus.
- To differentiate closely related bacterial species such as Klebsiella pneumoniae from Klebsiella oxytoca.
- To detect fecal contamination in water system, food and environmental samples and to help in differentiating human and animal origin contamination.
- To help in diagnosis of infections such as urinary tract infection, gastrointestinal infection, enteric fever, foodborne and waterborne infections by identifying the causative pathogen.
- To support public health and research by studying microbial diversity, infection control in hospital and community and to provide a practical diagnostic tool for pathology and public health laboratory.
Requirements for IMViC Test
General requirement
- Pure test bacterial culture (fresh culture suspension)
- Indole test
- Culture media– Tryptophan broth, peptone/tryptone water, SIM (Sulfide Indole Motility) medium or MIU medium.
- Reagents– Kovac’s reagent (commonly used) or Ehrlich’s reagent (preferred for anaerobic bacteria).
- Methyl red (MR) test
- Culture media– MR-VP broth or sterile glucose phosphate broth.
- Reagents– Methyl red pH indicator.
- Voges-Proskauer (VP) test
- Culture media– MR-VP broth or sterile glucose phosphate broth (same broth is used for MR and VP then it is separated).
- Reagents– Barritt’s reagent A (5% alpha-naphthol in absolute ethanol) and Barritt’s reagent B (40% potassium hydroxide/KOH solution).
- Citrate utilization test
- Culture media– Simmons citrate agar (slant) or Koser’s citrate broth.
- Reagents– Bromothymol blue (generally present in the medium as pH indicator).
IMViC test Procedure
- Indole test– Organism is inoculated in tryptophan broth (tryptone water/peptone water/SIM). Tube is incubated at 37°C for 24–48 hours. Kovac’s reagent is added (6–8 drops) (Ehrlich’s reagent can be used for anaerobes). Cherry-red/pink ring at top indicates positive result and yellow ring/no change indicates negative result.
- Methyl red (MR) test– Organism is inoculated in MR-VP broth (glucose phosphate broth). Tube is incubated at 37°C for 24 hours. Methyl red indicator is added (5–8 drops) and mixed. Bright red colour indicates positive MR test and yellow/orange colour indicates negative result.
- Voges-Proskauer (VP) test– Organism is inoculated in MR-VP broth. Tube is incubated at 37°C for 24–48 hours. Barritt’s reagent A (5% alpha-naphthol) is added and mixed. Barritt’s reagent B (40% KOH) is added and tube is shaken for aeration (oxygen is required). Tube is kept open and undisturbed for 30–45 minutes. Pink-red colour at top indicates positive VP test and brown/no change indicates negative result.
- Citrate utilization test– Simmons citrate agar slant is inoculated by streaking on surface. Tube is incubated at 37°C for 24–48 hours (some organism may require upto 4 days). Deep Prussian blue colour with growth indicates positive citrate test and green colour with no growth indicates negative result.
Indole Test
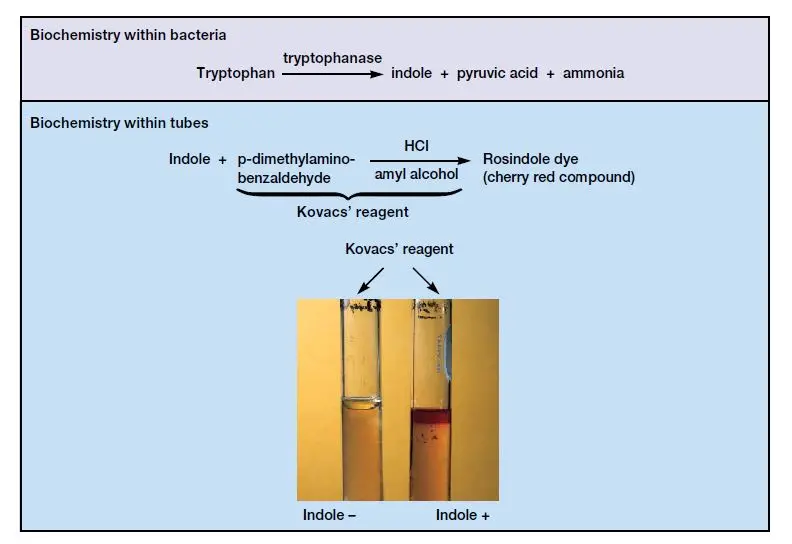

Principle of Indole test
Indole test works on the principle of determining the ability of bacteria to produce enzyme tryptophanase. This enzyme catalyzes the breakdown of amino acid tryptophan into indole, pyruvic acid and ammonia. When Kovac’s reagent or Ehrlich’s reagent is added it reacts with indole and a cherry-red or pink quinoid compound is formed.
Procedure of Indole test
- Standard tube method
- Sterile test tube containing tryptophan broth/peptone water/tryptone water is inoculated with test bacteria under aseptic condition.
- The tube is incubated at 37°C for 24 to 48 hours.
- About 0.5 mL (or 6 to 8 drops) of Kovac’s reagent is added directly to the broth culture.
- The tube is shaken gently and colour change at the surface is observed immediately.
- Rapid spot method
- Few drops of indole spot reagent (DMACA) is placed on filter paper to saturate it.
- A portion of isolated colony (18–24 hours old) is picked using inoculating loop or wooden applicator stick and rubbed on the saturated filter paper.
- The paper is examined immediately for colour development within 1 to 3 minutes.
Results of Indole test
- Tube test positive– Bright pink or cherry-red ring is formed in the oily reagent layer at the top of the liquid.
- Tube test negative– No colour change is seen and yellow or unaltered ring remains at the top.
- Spot test positive– Bacterial smear on the filter paper turns blue to blue-green colour.
- Spot test negative– Bacterial smear remains colourless or it turns light pink.
Indole Test Positive (+) and Negative (–) Bacteria
Positive (+)
- Escherichia coli
- Klebsiella oxytoca
- Proteus vulgaris
- Morganella morganii
- Citrobacter koseri
- Vibrio cholerae
- Providencia species
Negative (–)
- Klebsiella pneumoniae
- Enterobacter aerogenes
- Proteus mirabilis
- Citrobacter freundii
- Salmonella species
- Shigella species
- Pseudomonas aeruginosa
- Serratia marcescens
2. Methyl red test

Principle of Methyl red test
Methyl red (MR) test works on the principle of detecting mixed-acid fermentation of glucose by bacteria. In this process glucose is fermented and stable organic acids are produced such as lactic acid, acetic acid and formic acid. These acids lowers the pH of the culture medium to 4.4 or below.
When methyl red indicator is added it shows colour change based on acidity of medium. It turns red when pH is below 4.4 and it remains yellow when pH is above 6.0.
Procedure of Methyl Red Test
- Sterile glucose phosphate broth (MR-VP broth) is taken and it is inoculated with pure test culture suspension.
- The inoculated broth is incubated at 37°C for 24 hours.
- After incubation 5 to 6 drops of methyl red indicator is added to the tube.
- The tube is shaken well and immediate colour change of medium is observed.
Results of Methyl red (MR) test
- Positive result– Medium turns bright red colour. This indicates stable acids are produced and pH is dropped to 4.4 or below. Examples– Escherichia coli, Proteus vulgaris and Salmonella spp.
- Negative result– Medium turns yellow or orange colour. This indicates sufficient stable acids are not produced and pH is not lowered (butanediol pathway is used). Examples– Klebsiella spp. and Enterobacter aerogenes.
Methyl Red (MR) Test Positive (+) and Negative (–) Bacteria
Positive (+)
- Escherichia coli
- Proteus vulgaris
- Proteus mirabilis
- Salmonella species
- Shigella species
- Citrobacter freundii
- Yersinia species
- Staphylococcus aureus
Negative (–)
- Klebsiella pneumoniae
- Klebsiella oxytoca
- Enterobacter aerogenes
- Serratia marcescens
- Hafnia species
3. Voges-Proskauer (VP ) Test

Principle of Voges Proskauer test
Voges Proskauer (VP) test works on the principle of detecting glucose metabolism through butanediol pathway. In this pathway glucose is metabolized and a neutral intermediate compound acetoin (acetylmethylcarbinol) is produced.
When the culture is exposed to oxygen and potassium hydroxide (KOH) the acetoin present is oxidized into diacetyl. In presence of α-naphthol this diacetyl reacts with guanidine component of peptone in the broth and a pink or red coloured complex is formed which indicates positive VP test.
Procedure of Voges-Proskauer (VP) test
- Sterile glucose phosphate broth (MR-VP broth) is inoculated with test bacterial culture.
- The broth is incubated at 37°C for 24 to 48 hours.
- Barritt’s reagent A (5% α-naphthol) is added (0.6 mL or about 12 drops) and mixed well.
- Barritt’s reagent B (40% KOH) is added (0.2 mL or about 4 drops) and the tube is shaken vigorously for aeration because oxygen is required.
- The tube is kept open to air and left undisturbed for 30 to 45 minutes (cap is not replaced).
Results of Voges-Proskauer (VP) test
- Positive result– Pinkish-red/red colour develops at the top of medium within 5 to 30 minutes. This indicates acetoin is produced. Examples– Klebsiella spp., Enterobacter spp. and Staphylococcus aureus.
- Negative result– Medium remains yellow or turns brownish colour. This indicates acetoin is not produced. Examples– Escherichia spp., Proteus vulgaris and Citrobacter freundii.
Voges–Proskauer (VP) Test Positive (+) and Negative (–) Bacteria
Positive (+)
- Klebsiella pneumoniae
- Klebsiella oxytoca
- Enterobacter aerogenes
- Serratia marcescens
- Proteus mirabilis
- Hafnia species
- Staphylococcus aureus
Negative (–)
- Escherichia coli
- Proteus vulgaris
- Citrobacter freundii
- Salmonella species
- Shigella species
4. Citrate utilization test

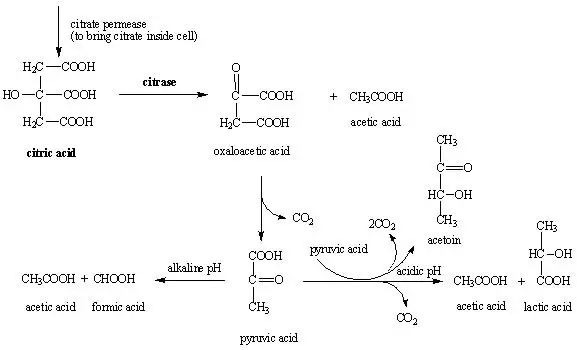
Principle of Citrate utilization test
Citrate utilization test works on the principle of determining the ability of microorganism to use citrate as the sole carbon and energy source. Organisms which can utilize citrate produces an enzyme citrase which breaks citrate into oxaloacetic acid and acetic acid. Oxaloacetic acid is then decarboxylated and pyruvate and carbon dioxide (CO2) is produced.
The CO2 released combines with water and sodium (from sodium citrate) to form alkaline sodium carbonate. At the same time ammonium salts present in the medium is used as sole nitrogen source and alkaline ammonia/ammonium hydroxide is produced. Due to these alkaline byproducts pH of the medium increases above 7.6.
At high pH bromothymol blue indicator changes from deep forest green to Prussian blue (royal blue) colour and this indicates positive citrate utilization.
Procedure of Citrate utilization test
- Simmons citrate agar slant (or Koser’s citrate broth) is prepared as the test medium.
- The medium is inoculated with test bacterial suspension. In case of citrate agar slant inoculum is streaked directly up the surface of slant.
- The tube is incubated at 37°C for 24 hours.
Results of Citrate utilization test
- Positive result (Simmons citrate agar)– Medium changes from green to deep Prussian blue/royal blue with visible bacterial growth on the slant.
- Positive result (Koser’s citrate broth)– Broth shows turbidity (cloudiness) indicating growth.
- Negative result– No bacterial growth is seen and the agar medium remains green.
Citrate Utilization Test Positive (+) and Negative (–) Bacteria
Positive (+)
- Klebsiella pneumoniae
- Klebsiella oxytoca
- Enterobacter aerogenes
- Citrobacter freundii
- Salmonella Typhimurium
- Serratia marcescens
- Proteus mirabilis
- Pseudomonas aeruginosa
- Providencia species
Negative (–)
- Escherichia coli
- Shigella species
- Salmonella Typhi
- Salmonella Paratyphi A
- Morganella morganii
- Yersinia enterocolitica
IMVIC test results of Common bacteria
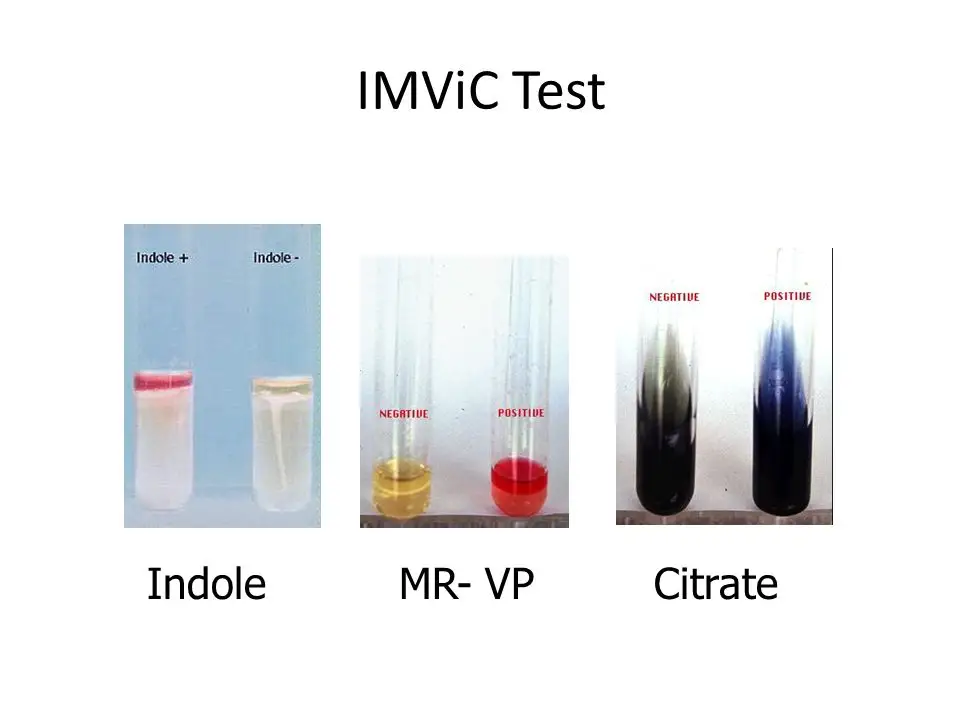
- Escherichia coli– Indole (+), MR (+), VP (–), Citrate (–).
- Klebsiella pneumoniae– Indole (–), MR (–), VP (+), Citrate (+).
- Klebsiella oxytoca– Indole (+), MR (–), VP (+), Citrate (+).
- Enterobacter spp. (E. aerogenes/E. cloacae)– Indole (–), MR (–), VP (+), Citrate (+).
- Proteus mirabilis– Indole (–), MR (+), VP (–), Citrate (+).
- Proteus vulgaris– Indole (+), MR (+), VP (–), Citrate (–).
- Salmonella typhi– Indole (–), MR (+), VP (–), Citrate (–).
- Salmonella typhimurium– Indole (–), MR (+), VP (–), Citrate (+).
- Shigella spp. (S. dysenteriae/S. flexneri)– Indole (negative/variable), MR (+), VP (–), Citrate (–).
- Citrobacter freundii– Indole (–), MR (+), VP (–), Citrate (+).
- Citrobacter koseri– Indole (+), MR (+), VP (–), Citrate (+).
- Morganella morganii– Indole (+), MR (+), VP (–), Citrate (–).
- Pseudomonas aeruginosa– Indole (–), MR (–), VP (–), Citrate (+).
- Staphylococcus aureus– Indole (–), MR (+), VP (+), Citrate (negative/variable).

IMViC Test – Positive and Negative Result Bacteria
| Test | Positive (+) Bacteria | Negative (–) Bacteria |
|---|---|---|
| Indole Test | Escherichia coli Proteus vulgaris Klebsiella oxytoca Morganella morganii Citrobacter koseri | Klebsiella pneumoniae Enterobacter aerogenes Proteus mirabilis Citrobacter freundii Salmonella species |
| Methyl Red (MR) Test | Escherichia coli Proteus vulgaris Proteus mirabilis Salmonella species Shigella species Citrobacter freundii | Klebsiella pneumoniae Klebsiella oxytoca Enterobacter aerogenes Serratia marcescens |
| Voges Proskauer (VP) Test | Klebsiella pneumoniae Klebsiella oxytoca Enterobacter aerogenes Serratia marcescens Proteus mirabilis | Escherichia coli Proteus vulgaris Citrobacter freundii Salmonella species Shigella species |
| Citrate Utilization Test | Klebsiella pneumoniae Klebsiella oxytoca Enterobacter aerogenes Citrobacter freundii Salmonella Typhimurium Proteus mirabilis | Escherichia coli Shigella species Salmonella Typhi Morganella morganii |
Uses of IMViC test
- It is used to identify coliform bacteria and to differentiate different genera of Enterobacteriaceae such as Escherichia, Salmonella, Shigella, Klebsiella and Proteus.
- It is used to differentiate closely related bacterial species such as Klebsiella pneumoniae and Klebsiella oxytoca.
- It is used in clinical diagnosis to identify causative pathogens of gastrointestinal infections, urinary tract infections, enteric fever and waterborne illnesses.
- It is used in water quality assessment to detect coliforms and fecal contamination and it also helps to differentiate contamination of human and animal origin.
- It is used in food and beverage industry to detect spoilage organisms and VP test is also used in production of fermented beverages such as beer and wine.
- It is used in environmental monitoring to identify bacteria in natural environment and to monitor soil and water quality for bacterial contamination.
- It is used in research and laboratory quality control to study microbial diversity and bacterial metabolic pathways (glucose fermentation) and to ensure culture media quality and to support identification of antimicrobial resistance patterns.
- It is used in public health and infection control to identify source of contamination and specific pathogens and it supports environmental remediation and infection control in hospitals and communities.
Limitations of IMVIC Test
- IMViC test alone is not sufficient to identify bacteria upto species level so further biochemical tests are required for complete identification.
- Different bacterial genera can sometimes show same IMViC pattern so overlapping results can occur and clear identification becomes difficult.
- The test requires incubation period (about 1 to 4 days) so results are not immediate.
- Results are sensitive to procedural errors. Misuse of reagents or improper incubation can lead to false positive or false negative result.
- In indole test presence of glucose in media can reduce indole production due to acid end product formation and it can give false negative. Media containing dyes (MacConkey/EMB) can also interfere with colour interpretation.
- Rapid spot indole method is less sensitive than standard tube method and Kovac’s reagent is not suitable for anaerobic bacteria.
- Indole can diffuse into surrounding media and nearby indole negative colonies may appear indole positive giving false interpretation.
Advantages of IMViC Test
- It gives stable and reliable biochemical reactions and it is considered as important identification test after Gram stain.
- It is effective in differentiation of Enterobacteriaceae and it helps to separate closely related genera and species such as Escherichia coli from Klebsiella and Enterobacter and also Klebsiella pneumoniae from Klebsiella oxytoca.
- It is used as a diagnostic tool in clinical laboratory to identify enteric pathogens causing urinary tract infection, gastrointestinal infection, enteric fever and foodborne illnesses.
- It is used in water quality testing to differentiate fecal coliforms and non-fecal coliforms and to determine contamination source (human or animal).
- It is used in industrial and food quality control and VP test is used to detect spoilage organisms and to monitor bacteria in fermented beverages such as beer and wine.
- It is used for media quality assurance and tests like MR test helps to check contamination and acceptable pH range of culture media.
- It is used in metabolic and ecological research to study microbial diversity, bacterial physiology and to track metabolic pathways such as glucose fermentation.
Precautions of IMViC test
- Pure culture should be used. Mixed culture can give irregular or false observation.
- For indole test media containing glucose should not be used because acid end products can suppress indole production. Mueller Hinton agar should also be avoided because tryptophan can be destroyed during preparation.
- Colonies should not be picked from dye containing media such as MacConkey agar or EMB agar for indole test because dye carryover can interfere with final colour interpretation.
- For spot indole test isolated colonies should be selected and colonies should be separated by about 5 mm to avoid false positive due to indole diffusion from nearby colony.
- Kovac’s reagent should not be used for anaerobic bacteria. Ehrlich’s reagent or DMACA spot test should be used for anaerobes.
- When SIM agar deep is used for indole portion straight inoculating needle is used and it should be stabbed completely to the bottom of tube.
- In VP test reagents should be added in correct order and correct amount because VP reaction is sensitive.
- Aeration is required in VP test so tube cap should be kept off and the tube should be mixed for about 1 minute and kept open to air.
- After adding VP reagents and aeration the tube should be kept undisturbed for 30 to 45 minutes and it should not be shaken during waiting period.
- VP result should be read within 30 minutes after colour development and final result should not be recorded after 1 hour.
References
- Abdullhamzah, A. R., Numea, O. S., Khalil, A. M., Mohammed, H. N., Esmaein, M. A., Amir, S. H. A., & Almeer, A. K. (2023). Providencia Rettgeri Common Causes of UTI. International Journal of Medical Science and Clinical Research Studies, 3(4), 727–730. https://doi.org/10.47191/ijmscrs/v3-i4-28,
- Al-Gburi, N. M. (2020). Isolation and Molecular Identification and Antimicrobial Susceptibility of Providencia spp. from Raw Cow’s Milk in Baghdad, Iraq. Veterinary Medicine International, 2020, 8874747. https://doi.org/10.1155/2020/8874747,
- Al Montasir, A., & Al Mustaque, A. (2013). Purple Urine Bag Syndrome. Journal of Family Medicine and Primary Care, 2(1), 104–105. https://doi.org/10.4103/2249-4863.109970
- BacDive. (2025). Providencia heimbachae (CCUG 16446, NCTC 12003, DSM 3591). https://doi.org/10.13145/bacdive5075.20251217.10,
- BacDive. (2025). Providencia stuartii (DSM 4539, ATCC 29914, CCUG 14805). https://doi.org/10.13145/bacdive137499.20251217.10,
- ClinicalTrials.gov. (n.d.). Microbiological Characterization and Antibiotic Resistance Profiling of Providencia Spp,: An Emerging Pathogen Isolated From Urinary Tract Infections (NCT07204769). Retrieved from ClinicalTrials.gov,
- Dong, X., Jia, H., Yu, Y., Xiang, Y., & Zhang, Y. (2024). Genomic revisitation and reclassification of the genus Providencia. mSphere, 9(3), e00731-23. https://doi.org/10.1128/msphere.00731-23,
- Flabs. (n.d.). IMViC Test for Enteric Bacteria: Principle, Procedure, Results.
- Hawkey, P. M., McCormick, A., & Simpson, R. A. (1986). Selective and differential medium for the primary isolation of members of the Proteeae. Journal of Clinical Microbiology, 23(3), 600–603. https://doi.org/10.1128/jcm.23.3.600-603.1986,
- Indole Test- Principle, Reagents, Procedure, Result Interpretation and Limitations. (n.d.).
- Kurmasheva, N., Vorobiev, V., Sharipova, M., Efremova, T., & Mardanova, A. (2018). The Potential Virulence Factors of Providencia stuartii: Motility, Adherence, and Invasion. BioMed Research International, 2018, 3589135. https://doi.org/10.1155/2018/3589135,
- Lopes, J., Tetreau, G., Pounot, K., El Khatib, M., & Colletier, J.-P. (2022). Socialization of Providencia stuartii Enables Resistance to Environmental Insults. Microorganisms, 10(5), 901. https://doi.org/10.3390/microorganisms10050901,
- Maiti, T. K., Singh, V. K., & Pandey, P. (2013). Providencia rettgeri: An Unusual Cause of Central Nervous System Infections. American Journal of the Medical Sciences, 346(2), 158–159. https://doi.org/10.1097/MAJ.0b013e318294f998
- Microbe Notes. (n.d.). Biochemical Test of Providencia stuartii.
- Rajakumar, R., Khan, N., & Mahadik, A. (2020). Purple urine bag syndrome: an unusual clinical presentation. International Journal of Research in Medical Sciences, 8(5), 1891–1893. https://doi.org/10.18203/2320-6012.ijrms20201944
- Reynolds, J. (2024, February 6). 37: IMViC tests. Biology LibreTexts.,
- Taraganar Abubacker, N. R. (2015). Purple Urine Bag Syndrome. Journal of Clinical and Diagnostic Research, 9(8), OD01-OD02. https://doi.org/10.7860/JCDR/2015/14736.6304,
- Transferable urease activity in Providencia stuartii. (1981). Journal of Clinical Microbiology, 13(3), 561–565. https://doi.org/10.1128/jcm.13.3.561-565.1981
- Trivedi, M. K., Branton, A., Trivedi, D., Nayak, G., Mondal, S. C., & Jana, S. (2015). Antibiogram, Biochemical Reactions and Biotyping of Biofield Treated Providencia rettgeri. American Journal of Health Research, 3(6), 344–351. https://doi.org/10.11648/j.ajhr.20150306.15,
- Wikipedia contributors. (n.d.). Providencia rettgeri. Wikipedia.
- Wikipedia contributors. (n.d.). Providencia stuartii. Wikipedia.
سلام،ممنون بابت مطالب فوق العاده عالی،خیلی کمک کننده بود برای من