What is Triple Sugar Iron Test (TSIA Test)?
Triple Sugar Iron (TSI) agar is a differential solid culture medium. It is mainly used in clinical microbiology to identify and distinguish different Gram negative enteric bacteria such as Enterobacteriaceae family.
The medium is prepared in a test tube and it is solidified at an angle. Two growth environments is formed. The upper oxygen rich surface is called slant and the lower oxygen deprived portion is called butt.
It is formulated with three fermentable sugars. These are glucose (0.1%) lactose (1%) and sucrose (1%). A pH indicator phenol red is also present.
When the bacteria ferment these sugars acid is produced and pH is lowered. Due to this phenol red changes its colour from red orange to yellow.
If the organism cannot ferment the complex sugars after glucose is depleted it is switched to protein (peptone) breakdown. The alkaline byproducts is released and the slant colour is reverted back to red or deep pink.
Iron salts and sodium thiosulfate is present in this medium. It is used for testing hydrogen sulfide (H2S) gas production. This is indicated by formation of black precipitate in the bottom of the tube.
The medium can also detect fermentation gas like carbon dioxide and molecular hydrogen. This is shown by bubbles cracks or upward displacement of agar.
By observing colour change blackening and gas disruption the metabolic profile is determined and the species is presumptively identified.
Principle of Triple Sugar Iron Agar test (TSIA Test)
The principle of Triple Sugar Iron agar test (TSIA test) is based on carbohydrate fermentation and hydrogen sulfide (H2S) production. It is mainly used for differentiation of Gram negative enteric bacilli by their fermentation pattern.
TSI medium contains three sugars in specific ratio. These are lactose (1%) sucrose (1%) and glucose (0.1%). Peptones are present in the medium and phenol red is used as pH indicator.
When the organism ferment the sugars acidic end products is produced and the pH is lowered. Due to this phenol red changes the colour from red to yellow.
Glucose is present in very small amount so it is rapidly exhausted by glucose fermenters. After depletion of glucose the organism is switched to oxidative metabolism of peptones in oxygen rich slant. Alkaline byproducts is formed and the slant colour is reverted back to red while butt remains acidic and yellow due to anaerobic condition.
If the organism can ferment lactose or sucrose also acid is continuously produced. Due to this both slant and butt remains yellow.
If no sugars are fermented the organism break down only proteins. Both slant and butt becomes alkaline and it remains red.
Gas production (CO2 and H2) may also occur during fermentation. It is indicated by bubbles cracks or displacement of agar.
Sodium thiosulfate and iron salts is present for detection of H2S production. If sulfur compound is reduced H2S gas is produced and it reacts with iron to form black precipitate in butt of tube.
Composition of Triple Sugar Iron Agar
- Peptones and extracts (20.0–26.0 g/L) – It is meat extract beef extract yeast extract and peptones (casein or animal tissue) which provides nitrogen vitamins amino acids and minerals.
- Lactose (10.0 g/L) – It is a fermentable disaccharide (1%).
- Sucrose (10.0 g/L) – It is a fermentable disaccharide (1%).
- Glucose or dextrose (1.0 g/L) – It is a fermentable monosaccharide (0.1%) and it is depleted quickly.
- Sodium chloride (5.0 g/L) – It is added to maintain osmotic equilibrium and bacterial turgor.
- Sodium thiosulfate (0.2–0.3 g/L) – It is inorganic sulfur substrate which is reduced to hydrogen sulfide (H2S) by some bacteria.
- Iron salts (0.2–0.5 g/L) – It is ferrous sulfate or ferric ammonium citrate or ferrous ammonium sulfate which reacts with H2S to form black precipitate.
- Phenol red (0.024–0.025 g/L) – It is pH indicator which turns yellow in acidic condition and deep red in alkaline condition.
- Agar (9.0–13.0 g/L) – It is solidifying agent and it forms slant and butt in tube.
- Note – Exact concentration may vary slightly depending on manufacturer formulation.
Preparation of Triple Sugar Iron Agar
- The dehydrated Triple Sugar Iron (TSI) agar powder is weighed as per manufacturer instruction (usually 61.6–64.62 g). It is suspended in 1 litre distilled or demineralized water.
- The mixture is stirred continuously and it is heated to rolling boil so that the ingredients and agar is completely dissolved.
- The dissolved medium is dispensed into test tubes (about 5–7 mL per tube).
- The tubes are covered loosely with caps or cotton plugs.
- Sterilization is done by autoclaving at 121°C (15 lbs pressure) for 15 minutes. Some formulations may recommend 115°C for 15–30 minutes.
- After autoclaving the hot tubes is removed carefully and it is kept in slant position (about 30°) for cooling and solidification.
- The medium is allowed to set in sloped position so that deep butt (about 2.5–5 cm) and an upper aerobic slant is formed.
Methods or test procedure using Triple Sugar Iron Agar
- The prepared TSI agar tubes are kept at room temperature before use.
- A sterile straight inoculating needle is used. Inoculating loop is not used for this test.
- A single well isolated colony is picked from a fresh pure culture (18–24 hours).
- The medium is inoculated by stabbing the needle through the centre of agar. It is stabbed deep into butt up to 3–5 mm above bottom of tube.
- While withdrawing the needle the slant surface is streaked in zig zag (fishtail) manner.
- The cap is replaced and it is kept loose so that air exchange occurs for oxidative reaction on slant.
- The tubes are incubated in aerobic condition at 35°C–37°C for 18–24 hours.
- After incubation the tubes are examined for colour change (fermentation) bubbles or cracks (gas production) and black precipitate (H2S production).
- The carbohydrate fermentation result is read within 18–24 hours only to avoid false result. If only H2S production is checked incubation may be extended for another 24–48 hours.
Result interpretation on TSI agar
Carbohydrate fermentation (colour change)
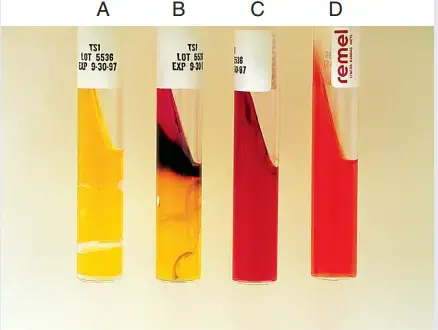
- Red slant or Yellow butt (K/A) – It indicates the organism ferment glucose only. After glucose is exhausted the organism is switched to protein (peptone) breakdown on oxygen rich slant and alkaline byproducts is produced so slant is reverted back to red while butt remains yellow.
- Yellow slant or Yellow butt (A/A) – It indicates the organism ferment glucose and lactose and or sucrose. The acid is produced continuously due to high concentration of lactose or sucrose so whole tube remains yellow.
- Red slant or Red butt (K/K) – It indicates no sugar fermentation. The organism rely on protein breakdown only and alkaline byproducts is produced so both slant and butt remains red.
- Red slant or Orange red butt (K/NC) – It indicates no sugar fermentation and butt remains unchanged (original uninoculated colour).
Gas production
- Bubbles cracks or lifted agar – It indicates fermentation gas (CO2 and H2) is produced. In vigorous fermenters the agar may be displaced and it may be pushed up in tube.
Hydrogen sulfide (H2S) production
- Black precipitate in butt – It indicates H2S gas is produced and it reacts with iron salts to form dense black precipitate in butt.
- Important rule for black butt – H2S production requires acidic condition. So even if black precipitate masks the butt colour the butt is always interpreted as acidic (yellow).
List of organisms (positive and negative result in TSI agar)
Note – In TSI agar there is no single positive or negative result. The organisms is interpreted for sugar fermentation (slant and butt) gas production and H2S production.
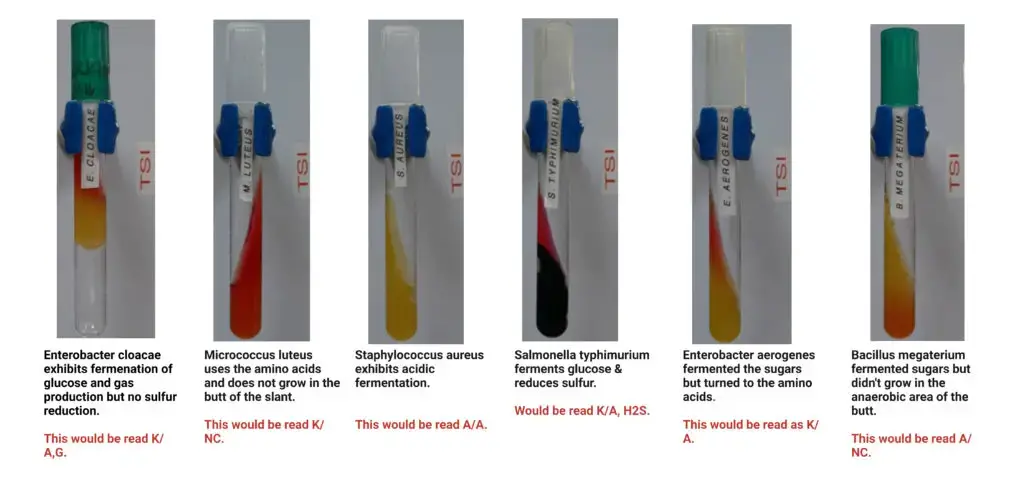
A/A (Yellow slant or Yellow butt) – Positive for glucose lactose and or sucrose fermentation
- Escherichia coli – Gas positive. H2S negative.
- Klebsiella pneumoniae – Gas positive. H2S negative.
- Enterobacter aerogenes – Gas positive. H2S negative.
- Enterobacter cloacae – Gas positive. H2S negative.
- Proteus vulgaris – Gas positive. H2S positive.
K/A (Red slant or Yellow butt) – Positive for glucose fermentation only
- Salmonella Typhimurium (and S. Enteritidis) – Gas positive. H2S positive.
- Salmonella Typhi – Gas negative. H2S positive (often black ring is seen).
- Shigella species (S. dysenteriae S. flexneri S. sonnei etc.) – Gas negative. H2S negative.
- Proteus mirabilis – Gas positive. H2S positive.
- Morganella morganii – Gas positive. H2S negative.
- Serratia marcescens – Gas negative. H2S negative.
- Yersinia enterocolitica – Gas negative. H2S negative.
K/K or K/NC (Red slant or Red butt or Orange red butt) – Negative for all carbohydrate fermentation
- Pseudomonas aeruginosa – Gas negative. H2S negative.
- Alcaligenes faecalis – Gas negative. H2S negative.
Variable reaction
- Citrobacter freundii – It may show A/A or K/A. Gas is positive and H2S is positive.

Precautions of Triple Sugar Iron Agar test (TSIA Test)
- A straight sterile inoculating needle is used always. Inoculating loop is not used because it can crack or tear agar and it may give false gas production.
- The butt is stabbed properly and deeply. If stabbing is not done in butt the organism does not reach anaerobic zone so fermentation and H2S production is not detected.
- The caps are kept loose during incubation. If cap is tightened air exchange is prevented and gas is trapped so alkaline reversion on slant does not occur and false yellow slant may be seen.
- The incubation time is maintained strictly (18–24 hours). Early reading (12 hours) may give false acid slant because glucose is not exhausted. Late reading (48–72 hours) may give false negative acid due to sugar exhaustion and protein breakdown and blackening may mask colour. If reading is delayed tubes are kept in refrigerator at 4°C after 24 hours.
- Pure culture is used. A single well isolated colony is taken. Mixed culture gives composite inaccurate result.
- The medium integrity is checked before use. Cracked bubbled or gap containing tubes are not used because it will look like gas production.
- Black butt is interpreted correctly. H2S production requires acidic condition so black butt is recorded as acidic (yellow) even if yellow colour is not visible.
- It is kept in mind that TSI is less sensitive for H2S. Sucrose may suppress H2S production so weak or false negative result may occur.
- It is also noted that TSI is a presumptive screening test. It does not confirm whether lactose sucrose or both is fermented in A/A reaction so further tests are required.
- Biohazard precautions are followed. Aseptic technique is used and waste is sterilized properly because pathogenic enteric bacteria may be handled.
Uses of Triple Sugar Iron Agar
- It is used for differentiating and presumptive identification of Gram negative enteric bacilli mainly Enterobacteriaceae from other Gram negative rods.
- It is used for determination of carbohydrate fermentation pattern (glucose lactose sucrose) and also for detection of H2S production and other fermentation gas.
- It is used for distinguishing pathogenic enteric bacteria such as Salmonella and Shigella (usually lactose and sucrose non fermenters) from commensal bacteria which ferment these sugars.
- It is used to help in differentiation of Salmonella and Proteus when it is done along with urease broth or urease agar test.
- It is used in food microbiology for testing and confirmation of Salmonella and other pathogens from food meat milk and dairy products.
- It is used as primary screening test and phenotypic cross check for genomic data. It is also cost effective in resource limited laboratory or field settings.
Limitations of Triple Sugar Iron Agar
- It is a presumptive identification test only. The confirmatory identification of species is done by further biochemical immunological molecular or mass spectrometry tests.
- It cannot distinguish specific sugar fermentation. If lactose sucrose or both is fermented the result is same (A/A) so it is not confirmed which sugar is fermented.
- H2S production may be suppressed. The presence of sucrose (1%) can suppress enzyme pathway for H2S production so some H2S producing organism may appear negative or weak positive.
- It has low sensitivity for H2S detection. A negative result in TSI does not rule out H2S production. More sensitive methods like SIM agar or lead acetate paper is used.
- Strict time is required. The result is read within 18–24 hours. Early reading (12 hours) may give false A/A and late reading (48 hours) may give false K/A due to protein breakdown after sugar exhaustion.
- Black precipitate may mask butt colour. Heavy H2S production can hide yellow butt so acid production is difficult to see but acidic butt is assumed if H2S is present.
- Proper inoculation is required. If stabbing is not done deep in butt anaerobic condition is not produced so fermentation and H2S detection may be affected.
- Cap tightening error may occur. If cap is tightened oxygen exchange is prevented and alkaline reversion on slant does not occur so false yellow slant may be seen.
- Pure culture is required. Mixed culture gives composite result so it is not suitable for direct mixed samples.
FAQ
What is the primary purpose of the TSIA Test?
The TSIA Test is used to differentiate Gram-negative enteric bacilli based on their carbohydrate fermentation and hydrogen sulfide production patterns.
Why are there three sugars in the TSIA medium?
The medium contains glucose, lactose, and sucrose to test the bacterium’s ability to ferment these specific carbohydrates, which helps in differentiation.
What does a red slant and yellow butt indicate in a TSIA test?
This indicates that only glucose has been fermented by the bacterium, leading to an alkaline slant and an acidic butt.
How is hydrogen sulfide (H2S) production detected in the TSIA test?
H2S production results in a blackening of the medium due to the reaction of H2S with ferric ions, forming an insoluble black precipitate.
Is gas production visible in the TSIA test?
Yes, gas production is evident through the formation of cracks, bubbles, or displacements in the agar.
Why is it crucial to read the test results within 18 to 24 hours?
Reading within this timeframe ensures accurate interpretation of sugar fermentation. Results read too early or too late can lead to incorrect conclusions.
Can the TSIA test be used for all bacteria?
No, the TSIA test is primarily designed for differentiating members of the Enterobacteriaceae family and other related Gram-negative rods.
What does a red slant and red butt indicate?
This suggests that the bacterium did not ferment any of the three sugars in the medium.
Is the TSIA test definitive for bacterial identification?
No, the TSIA test provides presumptive results. Further confirmatory tests are often required for a definitive identification.
How is the TSIA medium prepared?
The TSIA medium ingredients are mixed in appropriate proportions, dissolved in water, autoclaved for sterilization, and then dispensed in test tubes at an angle to create a slant with a deep butt.
References
- Biokar Diagnostics. (2018). TSI agar (ISO 6579-1): Identification of Enterobacteriaceae [Technical data sheet].
- Chamberlain, N. (n.d.). Archaea. Microbiology and Immunology On-line.
- Chauhan, A. (2024, December 18). Triple sugar iron test for Enterobacteriaceae diagnosis. Flabs.
- Comprehensive biochemical and diagnostic analysis of triple sugar iron (TSI) agar in clinical microbiology. (n.d.).
- Cooper, C. R., Jr. (2018). Triple sugar iron (TSI) test. Youngstown State University.
- Dahal, P. (2023, October 5). TSIA test: Principle, media, procedure, results, uses. Microbe Notes.
- Dalynn Biologicals. (2014, October). Triple sugar iron agar (TSI).
- Enterobacteriaceae. (2016, July 10). Veterian Key.
- Hardy Diagnostics. (2020). Triple sugar iron (TSI) agar [Instructions for use].
- HiMedia Laboratories. (2024, April). Triple sugar iron agar [Technical data].
- Kammel, M., Erdmann, C., & Sawers, R. G. (2024). The formate-hydrogen axis and its impact on the physiology of enterobacterial fermentation. Advances in Microbial Physiology, 84, 51-82.
- Lehman, D. (2005, September 1). Triple sugar iron (TSI) agar. American Society for Microbiology.
- Lehman, D. (2005, September 30). Triple sugar iron agar protocols. American Society for Microbiology.
- Melideo, S. L. (2015). Towards understanding the pathway for hydrogen sulfide metabolism [Doctoral dissertation, Drexel University].
- Miller, E. (2024). Triple sugar iron medium. In Microbiology laboratory manual. Open Oregon Educational Resources.
- Remel. (2008, August 14). Triple sugar iron (TSI) agar [Instructions for use]. ThermoFisher.
- Sigma-Aldrich. (n.d.). 44940 Triple sugar iron agar (TSI agar). Merck KGaA.
- TSI agar test: Results and interpretation. (n.d.). Scribd.
- TSI agar. (n.d.).
- Vivijs, B., Haberbeck, L. U., Mbong, V. B. M., Bernaerts, K., Geeraerd, A. H., Aertsen, A., & Michiels, C. W. (2015). Formate hydrogen lyase mediates stationary-phase deacidification and increases survival during sugar fermentation in acetoin-producing enterobacteria. Frontiers in Microbiology, 6, 150.
- Watson, R. (n.d.). Summary of biochemical tests. University of Wyoming.
- Wu, C. H. K. (2013). The role of hydrogen sulphide in lung diseases. ResearchGate.