Rickettsia is a small, Gram-negative, obligately intracellular bacterium. It is the organism which can grow and multiply only inside the living host cell.
It is highly specialized bacteria showing extreme genomic reduction. Due to this, many metabolic pathways are absent and the bacteria is unable to synthesize its own energy. It depends on the host cell cytoplasm for Adenosine Triphosphate (ATP). This condition is referred to as energy parasitism.
These organisms cannot be cultivated on ordinary artificial media. It is grown in tissue culture, embryonated eggs, or experimental animals.
Some of the important features are–
- It is Gram-negative and pleomorphic coccobacilli.
- It is obligate intracellular parasite.
- It cannot survive outside living cell.
- It is transmitted by blood sucking arthropods.
- Humans are accidental or dead-end host.
Transmission occurs through the bite, saliva, or feces of infected ticks, fleas, lice, mites and chiggers. In arthropods, it is maintained as a symbiotic or parasitic association.
After entering into bloodstream, Rickettsia infects the endothelial cells of small and medium sized blood vessels. The organism multiplies within these cells and causes damage.
This process results in inflammation of blood vessels, which is referred to as rickettsial vasculitis.
The infection is characterized by acute fever, severe headache and maculopapular or petechial rash. In many cases, a dark necrotic lesion called eschar is formed at the site of bite.
If treatment is not given with doxycycline, severe complications is produced. These includes central nervous system involvement, interstitial pneumonia, multiorgan failure and may lead to death.
Characteristics of Rickettsia
- It is a gram-negative, obligate intracellular bacteria. They are small and highly pleomorphic, occurring as cocci, bacilli or thread like forms. It can survive, grow and replicate only inside living eukaryotic cells.
- It cannot be grown in artificial nutrient broth or agar media. These bacteria require living tissue culture, cell culture or embryonated egg for laboratory cultivation because of their intracellular nature.
- In mammalian host, it preferentially infects microvascular endothelial cells lining small and medium sized blood vessels. Some species infect macrophages. After entering the host cell, it escapes from phagosome before fusion with lysosome and replicates freely in cytoplasm and sometimes in nucleus.
- It possess highly reduced genome (about 1.1–1.5 Mbp) due to reductive evolution. Genes for many metabolic pathways such as anaerobic glycolysis and synthesis of amino acids, nucleotides and lipids are absent. Because of this, the organism depend heavily on host cell nutrients.
- It shows energy parasitism. Although tricarboxylic acid (TCA) cycle is present and oxidative phosphorylation can occur, these bacteria can exchange its ADP with host ATP through specialized transport system. Thus host ATP is utilized for survival.
- It possess a typical gram-negative cell wall containing lipopolysaccharide (LPS) and peptidoglycan. A polysaccharide rich slime layer or microcapsule is present outside the wall which helps in resisting phagocytosis.
- Major outer membrane proteins (OmpA and OmpB) are present on surface. These proteins act as surface antigens and are important for adhesion and invasion of host cells.
- Many species shows actin-based motility. They polymerize host actin forming comet tails which propel the bacteria within cytoplasm and help in spreading from one cell to another without entering extracellular environment.
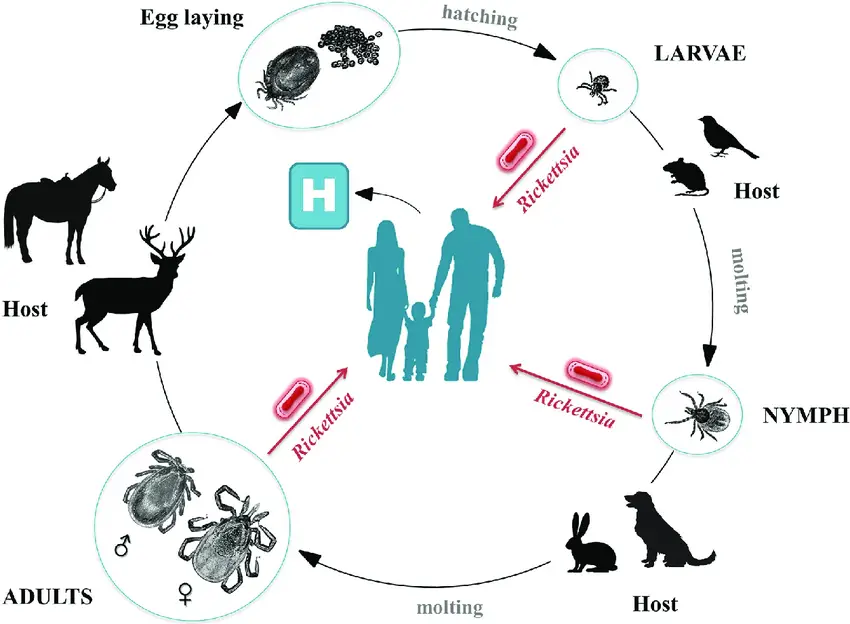
- It is transmitted by hematophagous arthropods such as ticks, fleas, lice and mites. These arthropods serve as vector as well as reservoir. Transmission may occur by transovarial and transstadial methods.
- Replication inside endothelial cells causes vascular damage and vasculitis. Increased vascular permeability is produced. In humans, it presents as acute febrile illness with fever, headache and rash. Sometimes an eschar is formed at the site of bite.
- Based on phylogenetic classification, the genus is divided into four groups – Spotted Fever Group (SFG), Typhus Group (TG), Transitional Group (TRG) and Ancestral Group (AG).
Ecology and Epidemiology of Rickettsia
General Ecology and Transmission
- It is obligate intracellular, gram-negative bacteria. The organism survive and multiply only within cytoplasm of vertebrate and invertebrate host cells.
- It is maintained in nature through hematophagous arthropods such as ticks, fleas, lice and mites. These vectors form the natural ecological cycle of the organism.
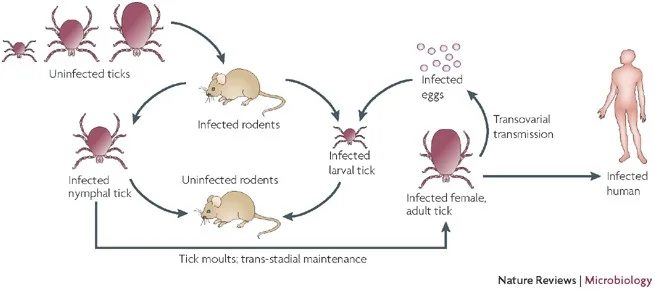
- In arthropod vectors, vertical transmission occurs. The bacteria is passed from infected female to eggs (transovarial transmission). It also survive through different life stages of vector such as larva, nymph and adult which is referred to as transstadial transmission.
- Horizontal transmission also occurs. Uninfected vectors acquire infection while feeding on blood of infected vertebrate host. Co-feeding transmission may also take place when infected and uninfected ticks feed in close proximity on same host.
- The interference phenomenon is observed in some tick populations. Non-pathogenic endosymbionts such as R. peacockii may block acquisition or transovarial transmission of highly pathogenic species like R. rickettsii.
- Humans are accidental or dead-end host in most infections. They usually do not contribute to maintenance of natural cycle except in epidemic typhus. Transmission to humans occurs through bite of infected tick or mite by salivary secretion or by rubbing infected flea or louse feces into skin abrasions or mucous membrane.
Major Epidemiological Groups
Spotted Fever Group (SFG)
- It is mainly transmitted by ticks and is the most diverse group.
- Rickettsia rickettsii – It is the causative agent of Rocky Mountain spotted fever. It is found in Americas and transmitted by ticks such as Dermacentor variabilis, Dermacentor andersoni and Rhipicephalus sanguineus. Reservoir hosts are rodents and dogs.
- Rickettsia conorii – It causes Mediterranean spotted fever. It is distributed in Europe, Africa and Asia. It is transmitted mainly by Rhipicephalus sanguineus and dogs act as reservoir.
- Rickettsia africae – It causes African tick bite fever. It is prevalent in sub-Saharan Africa and Caribbean region. It is transmitted by Amblyomma ticks and commonly seen in travellers.
- Rickettsia japonica – It causes Japanese spotted fever. It is transmitted by ticks such as Haemaphysalis longicornis and is found in Japan, South Korea and China.
Typhus Group (TG)
- It is transmitted by fleas and lice.
- Rickettsia prowazekii – It causes epidemic typhus. It is transmitted by human body louse (Pediculus humanus corporis). The bacteria kills its louse vector. Epidemics occur during war, famine and natural disaster where overcrowding and poor hygiene is present. Humans are primary reservoir.
- Rickettsia typhi – It causes murine or endemic typhus. It has worldwide distribution especially in tropical and subtropical port cities. It is transmitted by rat flea (Xenopsylla cheopis). Rats are main reservoir though opossums and domestic cats are involved in some regions.
Transitional Group (TRG)
- It shows features of both SFG and TG.
- Rickettsia akari – It causes rickettsialpox. It is maintained by house mice and transmitted by mouse mite (Liponyssoides sanguineus). It is commonly seen in urban areas.
- Rickettsia felis – It causes flea-borne spotted fever. It is transmitted by cat flea (Ctenocephalides felis). It is maintained in domestic cats, dogs and opossums.
Environmental and Demographic Factors
- Climate change and deforestation influence the distribution of vectors. Changes in temperature and rainfall expand the habitat range of ticks and fleas exposing new population to infection.
- Seasonality is observed in most tick-borne and flea-borne rickettsiosis. Incidence is higher during spring and summer months when vector activity is increased and human outdoor activity is more.
- Louse-borne epidemic typhus frequently occurs in cold months. Sharing of heavy clothing and blankets promote louse infestation.
- Certain infections such as tick-borne lymphadenopathy caused by R. slovaca shows peak incidence during cooler months when its vector Dermacentor marginatus is active.
Pathogenesis of Rickettsioses
- It is transmitted to human through bite of infected tick, mite or flea. In some infections, contamination of skin abrasion with infected arthropod feces introduce the organism into body.
- After entry into skin, the bacteria infect CD68+ mononuclear phagocytes, macrophages and dendritic cells present in dermis. This is the initial site of multiplication.
- From the site of inoculation, it spreads through lymphatic vessels to regional lymph nodes. Later it enters bloodstream and dissemination occurs to systemic and pulmonary circulation.
- It shows marked tropism for microvascular endothelial cells lining small and medium sized blood vessels. Infection of endothelial cells is the central event in pathogenesis.
- Entry into host cell is an active process. Surface cell antigens such as OmpA and OmpB bind to specific host receptors like Ku70, α2β1 integrin and FGFR1. This binding induces endocytosis mediated by clathrin and caveolin.
- After internalization, it escapes rapidly from phagosomal vacuole before fusion with lysosome. Membranolytic enzymes such as phospholipase D, phospholipase A2 and hemolysins are secreted which disrupt the vacuolar membrane.
- In cytoplasm, multiplication occurs by binary fission. Host ATP and metabolites are utilized because the organism depend on host cell for energy.
- In Spotted Fever Group, actin based motility is present. Bacterial proteins such as RickA and Sca2 stimulate polymerization of host actin forming actin tails. This help in direct spread from one cell to adjacent cell without entering extracellular space.
- In Typhus Group, actin based motility is absent. Massive intracellular multiplication occurs and host cell rupture releases the bacteria into bloodstream.
- Infection of endothelial cells activates NF-κB pathway and oxidative stress responses. Endothelial cells shift to pro-inflammatory and pro-coagulant state and cytokines such as IL-1α and IL-6 are produced.
- Perivascular infiltration of CD4 and CD8 T lymphocytes and macrophages occurs. The characteristic lesion formed is referred to as rickettsial vasculitis.
- Endothelial injury leads to disruption of tight junction proteins such as ZO-1 and adherens junction proteins like VE-cadherin. Increased vascular permeability is produced.
- Loss of vascular integrity results in plasma leakage and interstitial edema. Clinically it presents as maculopapular or petechial rash, hypotension and in severe cases multi organ failure and pulmonary edema.
Transmission of Rickettsia
Transmission of Rickettsia involves interaction between the bacteria, arthropod vectors and vertebrate hosts. Humans generally act as accidental or dead-end host (except Rickettsia prowazekii). The organisms are maintained in nature through different biological mechanisms in vectors.
Tick-Borne Transmission (Spotted Fever Group)
- Bite and salivary secretion – Ticks such as Dermacentor, Amblyomma and Rhipicephalus species transmit spotted fever group rickettsiae through infected saliva during blood meal. Transmission may occur within 30 minutes of tick attachment in case of Rickettsia rickettsii.
- Transovarial transmission – The infected female tick passes the bacteria to its eggs. This is referred to as transovarial transmission and helps in maintaining infection in tick population.
- Transstadial transmission – The organism survives during molting from larva to nymph to adult stage. This persistence in different stages is called transstadial transmission.
- Horizontal acquisition – Uninfected ticks acquire infection by feeding on rickettsemic vertebrate host.
- Co-feeding transmission – Bacteria can be transferred from infected tick to uninfected tick feeding closely on same host, even without systemic infection in host.
Louse-Borne Transmission (Typhus Group)
- Fecal contamination – Rickettsia prowazekii is transmitted by human body louse. The bacteria multiply in gut of louse and are excreted in feces. Infection occurs when contaminated feces are scratched into bite wound or skin abrasion.
- Inhalation – The organism can survive in dried louse feces for months. Infection may occur by inhalation of aerosolized infective fecal particles.
- Vector mortality – Body lice usually die within few weeks due to infection. There is no transovarial transmission in lice, therefore vertebrate reservoir is required for maintenance.
Flea-Borne Transmission (Typhus and Transitional Group)
- Fecal transmission – Rickettsia typhi and Rickettsia felis are transmitted by rat and cat fleas. Infection occurs when infected flea feces are rubbed into bite site or mucous membrane.
- Vertical maintenance – Unlike lice, fleas are not killed by infection. They maintain bacteria by transovarial transmission across generations.
Mite-Borne Transmission (Transitional and Scrub Typhus Group)
- Bite transmission – Mites transmit infections like rickettsialpox (Rickettsia akari) and scrub typhus (Orientia tsutsugamushi) through bite of infected mite.
- Transovarial and transstadial transmission – Mites act as reservoir and maintain infection in their population through both transovarial and transstadial transmission.
- Larval stage transmission – In scrub typhus, only larval stage (chigger) feeds on vertebrate host. Acquisition and transmission occurs during this stage only.
Atypical and Rare Transmission
- Blood transfusion and organ transplantation – Rare transmission has been reported through infected blood products and solid organ transplantation.
- Sylvatic transmission – In North America, Rickettsia prowazekii is maintained in flying squirrels. Humans may acquire infection by close contact with squirrels or their nests, possibly through ectoparasites or inhalation of dried feces.
Clinical Syndrome of Rickettsioses
Rickettsioses are acute febrile illnesses caused by infection of vascular endothelial cells by Rickettsia species. The disease is characterized by fever, headache and rash due to development of vasculitis.
Early Clinical Features
- The classical triad is fever, severe headache and rash. The complete triad may not be present in early stage of infection.
- It usually presents as an acute undifferentiated febrile illness. The condition resemble viral fever in beginning.
- Common general symptoms are chills, malaise, myalgia and arthralgia. Loss of appetite is frequently present.
- Gastrointestinal symptoms such as nausea, vomiting, abdominal pain and diarrhea may occur in early phase.
Cutaneous Manifestations
- Rash is the major clinical feature. It begins as macular or maculopapular eruption. With progression of vascular injury, it may become petechial or hemorrhagic.
- In Rocky Mountain spotted fever, rash appears after 2–5 days of fever. It starts from wrists and ankles and spreads to trunk. Palms and soles are commonly involved.
- In Typhus group infections, rash begins on trunk and spreads centrifugally. Face, palms and soles are usually spared.
- In some rickettsial infections, papulovesicular or pustular lesions are seen.
- Eschar (tache noire) is a dark necrotic crusted lesion present at the site of arthropod bite. It is commonly seen in Mediterranean spotted fever, African tick bite fever and scrub typhus. It is rarely seen in Rocky Mountain spotted fever.
Systemic Complications
- Infection of endothelial cells results in rickettsial vasculitis. Increased vascular permeability and plasma leakage occurs. Severe cases may progress to multi organ dysfunction.
- Neurological manifestations are meningoencephalitis, confusion, delirium, seizures and coma. Ataxia and hearing loss may develop.
- Respiratory involvement includes cough, dyspnea, interstitial pneumonia and non-cardiogenic pulmonary edema. Acute respiratory distress syndrome may occur.
- Renal involvement leads to acute kidney injury. Oliguria and prerenal azotemia may be present.
- Hepatic involvement is manifested by hepatitis, hepatomegaly and jaundice. Serum transaminases are elevated.
- Cardiovascular complications include hypotension, shock, myocarditis and cardiac failure.
- Hematological abnormalities are thrombocytopenia and disseminated intravascular coagulation. Severe vascular occlusion may produce gangrene of extremities.
Other Manifestations
- Regional lymphadenopathy may be present especially draining the site of eschar. Generalized lymphadenopathy is common in scrub typhus.
- Conjunctival injection and intraocular inflammation may occur.
Diagnosis Methods of Rickettsioses
Serological Tests
- Serological tests are the main method for diagnosis. These tests detect antibodies against rickettsial antigens. Antibodies usually appear after first week of illness, therefore it is mainly used for retrospective confirmation.
- Indirect Immunofluorescence Assay (IFA) is considered the gold standard test. It detects IgG and IgM antibodies. A definitive diagnosis is made by demonstrating four fold or greater rise in IgG antibody titre between acute and convalescent serum samples taken 2–4 weeks apart.
- Enzyme Linked Immunosorbent Assay (ELISA) is widely used alternative test. It is suitable for low resource settings and allows batch testing. It usually provides qualitative results such as reactive or non reactive.
- Weil–Felix test is an agglutination test based on cross reactivity between rickettsial antibodies and Proteus antigens. It has poor sensitivity and specificity but still used in some resource limited areas.
- Western blot and cross adsorption assays are specialized tests. These are performed in reference laboratories to differentiate specific Rickettsia species because cross reactivity is common in standard serological tests.
Molecular Methods
- Molecular methods are useful in acute phase before development of antibody response. These methods detect nucleic acid of organism.
- Polymerase Chain Reaction (PCR) is used to amplify specific genes such as gltA, ompA, ompB and 17-kDa antigen gene. It is highly sensitive when performed on tissue samples like skin biopsy of rash or swab of eschar. Sensitivity is low in whole blood because the organism mainly infect endothelial cells.
- Metagenomic next generation sequencing is an advanced genomic method. It can identify Rickettsia species directly from clinical specimen even without prior suspicion of pathogen.
- Isothermal amplification methods such as LAMP and RPA are newer techniques. These are simple, rapid and suitable for field diagnosis in resource limited settings.
Direct Detection in Tissue
- Immunohistochemistry and immunofluorescence techniques are used for direct visualization of rickettsial antigens in tissue samples. Formalin fixed skin biopsy from rash or eschar is commonly used. These methods provide acute diagnosis but require specialized laboratory.
Culture and Isolation
- Rickettsia are obligate intracellular bacteria and cannot be grown on ordinary nutrient media.
- Isolation requires living host cells such as tissue culture or embryonated chicken eggs. This method is technically difficult, hazardous and restricted to Biosafety Level 3 laboratories.
Clinical and Epidemiological Diagnosis
- In many cases, laboratory confirmation is delayed or negative in early stage. Therefore diagnosis is based on clinical suspicion.
- History of travel, outdoor exposure and contact with ticks, fleas, lice or mites is important. Presence of fever, headache, rash or eschar supports diagnosis.
- Empirical treatment with doxycycline should be started based on clinical suspicion without waiting for laboratory confirmation.
Treatment of Rickettsioses
Treatment of rickettsioses is mainly based on early administration of appropriate antibiotics. It is very important that therapy is started immediately on clinical suspicion, because delay in treatment increases the risk of severe complications and mortality. Treatment should not be delayed while waiting for laboratory confirmation.
The management of rickettsioses includes the following–
First-Line Therapy
- Doxycycline – It is the drug of choice for all rickettsial infections including Rocky Mountain spotted fever, typhus and other spotted fevers. It is considered as definitive first-line treatment in all age groups.
- Dose (Adults) – 100 mg is given twice daily.
- Dose (Children) – 2.2 mg/kg body weight is given twice daily in children below 45 kg body weight.
- Duration – Treatment is given for minimum 5–7 days. It should be continued for at least 3 days after fever subsides and clinical improvement is observed.
- Use in children and pregnancy – Doxycycline is recommended even in children under 8 years of age as short course (5–10 days) does not produce dental staining or enamel hypoplasia. In pregnant women with severe rickettsioses such as Rocky Mountain spotted fever, the life saving benefits is more compared to the potential risks.
Alternative Drugs (Used with Caution)
- Chloramphenicol – It was used previously as alternative drug, especially in pregnant women or in patients with severe tetracycline allergy. Its use is associated with higher case fatality rate in Rocky Mountain spotted fever. It may cause bone marrow suppression and aplastic anemia.
- Macrolides (Azithromycin, Clarithromycin) – These drugs may be used in mild infections such as Mediterranean spotted fever or African tick bite fever. It is sometimes considered in children and pregnant patients. It is not recommended in severe rickettsioses like epidemic typhus.
- Rifampin – It has limited clinical use. It may be effective in certain infections such as African tick bite fever or anaplasmosis in patients who are intolerant to doxycycline.
Contraindicated and Ineffective Drugs
- Sulfa-containing drugs (Trimethoprim-sulfamethoxazole) – These drugs are strictly contraindicated. It is not only ineffective but also may worsen the disease condition and increase the risk of fatal outcome.
- Fluoroquinolones (Ciprofloxacin, Levofloxacin) – These drugs are generally not recommended as it is associated with increased severity and poor clinical outcome.
- Penicillins and Cephalosporins – These broad spectrum antibiotics lack activity against Rickettsia species.
General Clinical Management
- Immediate empiric treatment – Therapy must be initiated immediately based on clinical suspicion such as fever, headache, rash or history of tick or flea exposure. Treatment should never be withheld while awaiting serological reports.
- Supportive care – Severely ill patients may require hospitalization. Intravenous fluids and electrolyte balance is maintained. Fluid administration should be carefully monitored because increased vascular permeability may lead to pulmonary edema and cerebral edema.
- No prophylaxis – Post exposure antibiotic prophylaxis is not recommended in asymptomatic individuals following tick bite. Patients should be monitored for development of symptoms up to two weeks.
Thus, early recognition and prompt treatment with doxycycline is the most important step in reducing morbidity and mortality in rickettsioses.
Prevention and Control of Rickettsioses
Prevention and control of rickettsioses is mainly based on avoidance of arthropod vectors and maintenance of proper hygiene. It is essential because these infections are transmitted by ticks, mites, fleas and lice. There is no commercial vaccine available for general public. Therefore preventive measures is the main method of control.
Personal Protection and Vector Avoidance
- Avoid infested habitats – It is advised to avoid dense vegetation, tall grasses, bushes and endemic regions where vectors are highly active. This reduces the chance of contact with infected arthropods.
- Protective clothing – Long-sleeved shirts, long pants, closed shoes and long socks are worn to minimize exposed skin. These are simple mechanical barriers against vector bites.
- Use of insect repellents – Repellents containing DEET, picaridin, IR3535 or Oil of Lemon Eucalyptus (OLE) is applied directly on exposed skin. These agents repel ticks and other vectors.
- Treatment of clothing – Clothing and outdoor equipment is treated with permethrin. Pre-treated garments can also be used and remain effective even after repeated washing.
Post-Exposure Hygiene and Checking
- Body inspection – After outdoor exposure, the body should be examined carefully. Areas such as under arms, behind knees, around ears, scalp and umbilical region must be checked.
- Immediate removal of ticks – If a tick is found attached, it is removed immediately. Early removal decreases the transmission of rickettsial pathogen.
- Bathing after exposure – Showering within two hours of returning from outdoor areas helps in removing unattached ticks. It reduces the risk of infection.
- Laundering of clothes – Clothes possibly infested should be washed in hot water (at least 50°C/122°F for lice) and dried at high heat for minimum 10 minutes. Alternatively, clothes can be kept in sealed bag for one week.
- Maintenance of hygiene – Regular bathing and washing of clothes is important in preventing body lice infestation which transmit epidemic typhus.
Environmental and Animal Management
- Rodent control – Rat and mouse harborages around houses should be eliminated. These rodents carry fleas and mites which transmit murine typhus and rickettsialpox.
- Avoidance of wild animals – Contact with wild and stray animals should be avoided. Flying squirrels act as natural reservoir of epidemic typhus and should not be allowed to nest in houses.
- Treatment of pets and livestock – Domestic animals especially dogs should be treated with acaricides and tick-preventive collars. This reduces tick population in surrounding areas.
- Community vector control – During outbreaks, insecticides are used in mass level. Permethrin dusting of clothing is also carried out, especially in louse-borne typhus epidemics.
Medical Measures
- Absence of vaccine – At present, no approved vaccines are available for prevention of spotted fever and typhus group rickettsioses.
- Avoid prophylactic antibiotics – Post-exposure antibiotic prophylaxis is generally not recommended in asymptomatic individuals. It does not prevent disease and may lead to antibiotic resistance.
- Exception – In scrub typhus caused by Orientia tsutsugamushi, weekly doxycycline has been used as a prophylactic measure during exposure.
Thus, prevention of rickettsioses mainly depends on personal protection, environmental management and vector control. Early detection and removal of vectors is important in reducing the transmission of disease.
FAQ
What is Rickettsia infection?
Rickettsia infection refers to an infection caused by bacteria belonging to the genus Rickettsia. These bacteria are primarily transmitted to humans through arthropod vectors, such as ticks, fleas, lice, and mites.
How is Rickettsia infection diagnosed?
Diagnosis of Rickettsia infection typically involves a combination of clinical recognition, epidemiological context, and laboratory testing. Serological tests, PCR assays, and immunohistochemical analyses are commonly used to confirm the presence of the bacteria.
What is the treatment for Rickettsia infection?
The primary treatment for Rickettsia infection involves the administration of antibiotics, such as doxycycline, tetracycline, chloramphenicol, or fluoroquinolones. Prompt initiation of antibiotic therapy is important to prevent complications and improve outcomes.
Can Rickettsia infection be prevented?
Yes, Rickettsia infection can be prevented. Measures such as using protective clothing, applying insect repellents, and avoiding direct contact with vectors (ticks, fleas, lice, and mites) can reduce the risk of transmission. Additionally, environmental control measures and public health education play a role in prevention.
Are there vaccines available for Rickettsia infection?
Currently, there are no vaccines available for Rickettsia infection. Prevention mainly relies on avoiding contact with infected vectors and implementing personal protective measures.
What are the common symptoms of Rickettsia infection?
Common symptoms of Rickettsia infection include fever, headache, malaise (general feeling of unwellness), and a widespread rash. The specific symptoms may vary depending on the type of Rickettsia species involved.
How are Rickettsia infections transmitted?
Rickettsia infections are primarily transmitted through the bite of infected arthropod vectors. This can occur during the feeding process when the vectors, such as ticks or fleas, transmit the bacteria to humans. Transmission can also occur when infectious fluids or feces from the vectors contaminate the skin through scratching or crushing of the vector.
Are Rickettsia infections contagious from person to person?
No, Rickettsia infections are not considered contagious from person to person. The primary mode of transmission is through arthropod vectors, and human-to-human transmission is rare.
Can pets transmit Rickettsia infections?
Pets, such as dogs and cats, can become infected with certain Rickettsia species, but their role in transmitting the bacteria to humans is generally minimal. However, it is still important to protect pets from arthropod vectors to prevent their own infections.
Are Rickettsia infections common worldwide?
Rickettsia infections are found globally, but their prevalence varies depending on geographical factors, climate, and the presence of suitable arthropod vectors. Different species of Rickettsia may be more common in specific regions, leading to variations in the incidence of infection.
References
- Akram, S. M., Ladd, M., & King, K. C. (2023). Rickettsia prowazekii. In StatPearls. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK448173/
- Azad, A. F., & Beard, C. B. (1998). Rickettsial pathogens and their arthropod vectors. Emerging Infectious Diseases, 4(2), 179–186. https://doi.org/10.3201/eid0402.980205
- Bechah, Y., Paddock, C. D., Capo, C., Mege, J.-L., & Raoult, D. (2010). Adipose tissue serves as a reservoir for recrudescent Rickettsia prowazekii infection in a mouse model. PLoS ONE, 5(1), e8547. https://doi.org/10.1371/journal.pone.0008547
- Blanton, L. S. (2019). The rickettsioses: A practical update. Infectious Disease Clinics of North America, 33(1), 213–229. https://doi.org/10.1016/j.idc.2018.10.010
- Blanton, L. S., Muir, S. E., Mendell, N. L., & Walker, D. H. (2025). Rickettsia parkeri rickettsiosis resembling Sweet syndrome: A differential diagnosis for critical discussion. Infectious Disease Reports, 17(3), 45. https://doi.org/10.3390/idr17030045
- Cacciaguerra, G., Pavone, P., Caltabiano, G. M., La Cognata, D., Liotta, E., Marino, S., Spampinato, S., Micali, C., Marino, A., Ceccarelli, M., & Nunnari, G. (2024). Rickettsial infection: An overview of clinical, epidemiological, diagnostic features. Case report with neurological complication. Academic Journal of Pediatrics & Neonatology, 13(4), 555925. https://doi.org/10.19080/AJPN.2024.13.555925
- Chan, Y. G. Y., Cardwell, M. M., Hermanas, T. M., Uchiyama, T., & Martinez, J. J. (2009). Rickettsial outer-membrane protein B (rOmpB) mediates bacterial invasion through Ku70 in an actin, c-Cbl, clathrin and caveolin 2-dependent manner. Cellular Microbiology, 11(4), 629–644. https://doi.org/10.1111/j.1462-5822.2008.01279.x
- Dalmanoğlu, E., Tüz, M. A., Tüz, H. İ., & Ecer, D. T. (2025). Rickettsiosis cases presenting with rash: A case series from an endemic region in Turkey. Revista do Instituto de Medicina Tropical de São Paulo, 67, e41. https://doi.org/10.1590/S1678-9946202567041
- Denison, A. M., Amin, B. D., Nicholson, W. L., & Paddock, C. D. (2014). Detection of Rickettsia rickettsii, Rickettsia parkeri, and Rickettsia akari in skin biopsy specimens using a multiplex real-time polymerase chain reaction assay. Clinical Infectious Diseases, 59(5), 635–642. https://doi.org/10.1093/cid/ciu358
- Dr.Oracle Medical Advisory Board & Editors. (2025). Cross-reactivity in Rickettsia serology and your patient’s unusual serologic pattern. Dr.Oracle.
- El Karkouri, K., Pontarotti, P., Raoult, D., & Fournier, P.-E. (2016). Origin and evolution of rickettsial plasmids. PLoS ONE, 11(2), e0147492. https://doi.org/10.1371/journal.pone.0147492
- Eremeeva, M. E., & Dasch, G. A. (2015). Challenges posed by tick-borne rickettsiae: Eco-epidemiology and public health implications. Frontiers in Public Health, 3, 55. https://doi.org/10.3389/fpubh.2015.00055
- Ganbold, T. G. T., Acharya, S. A. S., Sheet, S. S., Munkhtur, E. M., & Hong, S.-T. (2025). Advancements in vaccine development and therapeutic approaches for severe fever with thrombocytopenia syndrome. Frontiers in Cellular and Infection Microbiology, 15, 1650652. https://doi.org/10.3389/fcimb.2025.1650652
- Helminiak, L., Mishra, S., & Kim, H. K. (2022). Pathogenicity and virulence of Rickettsia. Virulence, 13(1), 1752–1771. https://doi.org/10.1080/21505594.2022.2132047
- Hussain, M. H., Yu, Y., Wang, L., & Qureshi, J. A. (2025). Editorial: Recent advances in vector-borne diseases and climate change. Frontiers in Microbiology, 16, 1746230. https://doi.org/10.3389/fmicb.2025.1746230
- Illinois Department of Public Health. (n.d.). Spotted fever rickettsioses information for clinicians. Retrieved March 13, 2026.
- Kwaghe, V. G., Erameh, C., Courtney, L. P., Samuels, J. O., Quiner, C. A., Kim, J. H., Edeawe, O. I., Vongdip, N. G., Ephraim, A. Z., Ejike, O. J., Odia, I., Asman, K., Chebu, P., Agbukor, J., Matthew, O. D., Orok, V., Owolagba, F., Okhiria, B., Ogbaini-Emovon, E., Odion, W. M., Obagho, B. A., Fayomade, R., & Oga, E. A. (2025). Epidemiology and clinical characteristics of human rickettsial infections detected in Nigeria: Discovery of a significant contributor to acute febrile illness. Clinical Infectious Diseases, 81(Suppl 4), S160–S167. https://doi.org/10.1093/cid/ciaf502
- Laukaitis, H. J., & Macaluso, K. R. (2021). Unpacking the intricacies of Rickettsia-vector interactions. Trends in Parasitology, 37(8), 734–746. https://doi.org/10.1016/j.pt.2021.05.008
- Levin, M. L., Schumacher, L. B., & Snellgrove, A. (2018). Effects of Rickettsia amblyommatis infection on the vector competence of Amblyomma americanum ticks for Rickettsia rickettsii. Vector-Borne and Zoonotic Diseases, 18(11), 579–587. https://doi.org/10.1089/vbz.2018.2284
- Londoño, A. F., Scorpio, D. G., & Dumler, J. S. (2023). Innate immunity in rickettsial infections. Frontiers in Cellular and Infection Microbiology, 13, 1187267. https://doi.org/10.3389/fcimb.2023.1187267
- McCormick, D. W., & Nicholson, W. L. (2025). Rickettsial diseases. In CDC Yellow Book: Health information for international travel (2026 ed.). Centers for Disease Control and Prevention.
- MDPI. (2025). Vaccines, Volume 13, Issue 9 (September 2025). Vaccines, 13(9).
- Miranda R., J., Mattar V., S., & Gonzalez T., M. (2017). Rickettsiosis. Revista MVZ Córdoba, 22.
- National Emerging Special Pathogens Training & Education Center (NETEC). (2024, March 25). Climate change and infectious diseases.
- Oakley, A. (2017). Rickettsial diseases. DermNet.
- Pawar, B., Loganathan, S., Belliappa, K. M., Ranganathan, L. B., Thekdi, K. P., & Hiware, S. D. (2026). A comprehensive review of vaccine development: From traditional platforms to messenger RNA (mRNA) technologies. Cureus, 18(1), e100608. https://doi.org/10.7759/cureus.100608
- Perlman, S. J., Hunter, M. S., & Zchori-Fein, E. (2006). The emerging diversity of Rickettsia. Proceedings of the Royal Society B: Biological Sciences, 273(1598), 2097–2106. https://doi.org/10.1098/rspb.2006.3541
- Phuklia, W., Padith, K., Phommasone, K., Chansamouth, V., Mayxay, M., Vongphachanh, S., Newton, P. N., Robinson, M. T., & Ashley, E. A. (2026). Targeting surface cell antigen 2 increases sensitivity of Rickettsia typhi detection. PLoS Neglected Tropical Diseases, 20(2), e0014004. https://doi.org/10.1371/journal.pntd.0014004
- Pipová, N., Peňazziová, K., Baňas, M., Majláth, I., & Majláthová, V. (2023). The behavior of Rickettsia-positive Dermacentor reticulatus ticks under laboratory conditions. Life, 13(3), 612. https://doi.org/10.3390/life13030612
- Rydkina, E., Sahni, A., Baggs, R. B., Silverman, D. J., & Sahni, S. K. (2006). Infection of human endothelial cells with spotted fever group rickettsiae stimulates cyclooxygenase 2 expression and release of vasoactive prostaglandins. Infection and Immunity, 74(9), 5067–5074. https://doi.org/10.1128/IAI.00182-06
- Sahni, A., Fang, R., Sahni, S. K., & Walker, D. H. (2019). Pathogenesis of rickettsial diseases: Pathogenic and immune mechanisms of an endotheliotropic infection. Annual Review of Pathology: Mechanisms of Disease, 14, 127–152. https://doi.org/10.1146/annurev-pathmechdis-012418-012800
- Sahni, A., Narra, H. P., Walker, D. H., & Sahni, S. K. (2016). Endothelial activation and injury: The mechanisms of rickettsial vasculitis. In K. Stokes & F. Gavins (Eds.), Vascular Responses to Pathogens (pp. 111–122). Elsevier Inc. https://doi.org/10.1016/B978-0-12-801078-5.00009-1
- Sahni, S. K., Narra, H. P., Sahni, A., & Walker, D. H. (2013). Recent molecular insights into rickettsial pathogenesis and immunity. Future Microbiology, 8(10), 1265–1288. https://doi.org/10.2217/fmb.13.102
- Snowden, J., Ladd, M., & King, K. C. (2023). Rickettsial infection. In StatPearls. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK431127/
- Spickler, A. R. (2017). Typhus fever – Rickettsia prowazekii. The Center for Food Security and Public Health.
- Stewart, A. G., & Stewart, A. G. A. (2021). An update on the laboratory diagnosis of Rickettsia spp. infection. Pathogens, 10(10), 1319. https://doi.org/10.3390/pathogens10101319
- Uchiyama, T. (2012). Tropism and pathogenicity of rickettsiae. Frontiers in Microbiology, 3, 230. https://doi.org/10.3389/fmicb.2012.00230
- Velatooru, L. R., Arroyave, E., Rippee-Brooks, M. D., Burch, M., Yang, E., Zhu, B., Walker, D. H., Zhang, Y., & Fang, R. (2024). Rickettsia disrupts and reduces endothelial tight junction protein zonula occludens-1 in association with inflammasome activation. Infection and Immunity, 93(1), e00468-24. https://doi.org/10.1128/iai.00468-24
- Walker, D. H. (1996). Rickettsiae. In S. Baron (Ed.), Medical Microbiology (4th ed.). University of Texas Medical Branch at Galveston. https://www.ncbi.nlm.nih.gov/books/NBK7624/
- Walker, D. H. (2016). Rickettsia and related genera. In Basicmedical Key.
- Wang, X., Shang, M., Wang, Z., Ji, H., Wang, Z., & Liu, Q. (2025). Projecting the potential distribution of Rickettsia japonica in China and Asian adjacent regions under climate change using the Maxent model. Frontiers in Public Health, 13, 1478736. https://doi.org/10.3389/fpubh.2025.1478736
- Weinert, L. A., Werren, J. H., Aebi, A., Stone, G. N., & Jiggins, F. M. (2009). Evolution and diversity of Rickettsia bacteria. BMC Biology, 7, 6. https://doi.org/10.1186/1741-7007-7-6
- Wikipedia contributors. (2026, February 7). Rickettsia. Wikipedia, The Free Encyclopedia. https://en.wikipedia.org/wiki/Rickettsia
- Wikipedia contributors. (2026, January 12). Rickettsia rickettsii. Wikipedia, The Free Encyclopedia. https://en.wikipedia.org/wiki/Rickettsia_rickettsii