PYR test (Pyrrolidonyl Arylamidase test) is a rapid colorimetric biochemical test used in clinical microbiology. It is done for detection of enzyme pyrrolidonyl arylamidase (pyrrolidonyl aminopeptidase) in bacteria. In this test the bacteria is exposed to a synthetic substrate L-pyrrolidonyl-β-naphthylamide. If the enzyme is present the substrate is hydrolyzed and β-naphthylamine is released which is colourless. After that a chromogenic developer (p-dimethylaminocinnamaldehyde) is added. The developer reacts with β-naphthylamine and bright pink or cherry-red colour is produced. This colour formation is taken as positive result. This test is used mainly for presumptive identification of Group A streptococci (Streptococcus pyogenes) and Enterococcus species because these organism gives strong positive reaction. It is also used for differentiating some coagulase-negative staphylococci like Staphylococcus haemolyticus and Staphylococcus lugdunensis. It is used for distinguishing Escherichia coli from other Gram negative bacteria.
Principle of PYR Test
The principle of PYR test is based on detection of enzyme L-pyrrolidonyl arylamidase (pyrrolidonyl peptidase or pyrrolidonyl aminopeptidase) produced by bacteria. In this test the organism is exposed to a synthetic substrate L-pyrrolidonyl-β-naphthylamide (PYR). If the enzyme is present it acts as peptidase and the PYR substrate is hydrolyzed (cleaved) to release L-pyroglutamic acid (L-pyrrolidone) and a colourless compound β-naphthylamine. This intermediate is not seen directly so a chromogenic developer (PYR reagent) containing p-dimethylaminocinnamaldehyde is added. The reagent reacts with free β-naphthylamine and a Schiff base is formed due to this bright pink to cherry-red colour is produced which indicates positive test. If the enzyme is absent the substrate is not broken down and after adding reagent there is no colour change or a yellow/orange colour is seen which indicates negative result.
Purpose of PYR Test
- It is used for detection of enzyme L-pyrrolidonyl arylamidase (pyrrolidonyl aminopeptidase) in bacteria.
- It is used for presumptive identification of Group A beta hemolytic streptococci like Streptococcus pyogenes because it gives PYR positive.
- It is used for rapid differentiation of Enterococcus species (PYR positive) from non enterococcal Group D streptococci like Streptococcus bovis and Streptococcus equinus (PYR negative).
- It is used for differentiation among coagulase negative staphylococci and identification of Staphylococcus lugdunensis Staphylococcus haemolyticus and Staphylococcus schleiferi.
- It is used for distinguishing Staphylococcus aureus (generally PYR negative) from Staphylococcus intermedius group (PYR positive).
- It is used for helping in identification of Escherichia coli (PYR negative) by separating it from other indole positive lactose positive Gram negative rods.
- It is used for differentiating within Enterobacteriaceae like Citrobacter species (PYR positive) from Salmonella species (PYR negative).
Requirements for PYR Test
- PYR broth or PYR agar is required for tube method.
- PYR disk or PYR strip is required for rapid disk method (it is impregnated with substrate L-pyrrolidonyl-β-naphthylamide).
- A non selective culture media is required for obtaining pure colonies like Blood agar or Brain heart infusion agar (BHI agar).
- PYR reagent (colour developer) is required which contains p-dimethylaminocinnamaldehyde (DMACA) in hydrochloric acid solution.
- Sterile distilled water or demineralized water is required for moistening the PYR disk (do not flood).
- Sterile inoculating loop swab or wooden applicator stick is required for picking and transferring colonies.
- Loop sterilizer incinerator or Bunsen burner is required for sterilization.
- Incubator is required for maintaining temperature (35–37°C) for growth and broth method.
- Sterile forceps is required for placing PYR disk.
- Clean glass slide or empty Petri dish is required for holding the PYR disk during testing.
- Micropipette (5–10 µl) and test tubes are required for dispensing water and performing tube method.
- Positive control organism is required like Streptococcus pyogenes (ATCC 19615) or Enterococcus faecalis (ATCC 29212).
- Negative control organism is required like Escherichia coli (ATCC 25922) or Streptococcus agalactiae (ATCC 12386).
Procedure of PYR Test
Rapid disk method
- A PYR disk is placed in a empty sterile Petri dish or on a clean glass slide by using sterile forceps.
- The disk is moistened slightly with sterile distilled water or deionized water (do not flood the disk).
- Several well isolated colonies are picked from a pure 18–24 hours old culture by using sterile inoculating loop or wooden applicator stick.
- A heavy visible inoculum (paste) is rubbed on a small area of the moistened PYR disk.
- The disk is incubated at room temperature for 1–2 minutes (it can be extended upto 10 minutes for slow growing organism).
- After incubation 1–2 drops of PYR reagent (N N-dimethylaminocinnamaldehyde) is added directly on the inoculated area.
- The colour change is observed within 1–2 minutes.
Tube broth method
- A tube of PYR broth is inoculated by using 3–5 colonies from a pure 18–24 hours old culture.
- The tube is incubated aerobically at 35–37°C for about 4 hours.
- After incubation 1–2 drops of PYR reagent is added into the tube.
- The development of red colour is observed within 1–2 minutes.
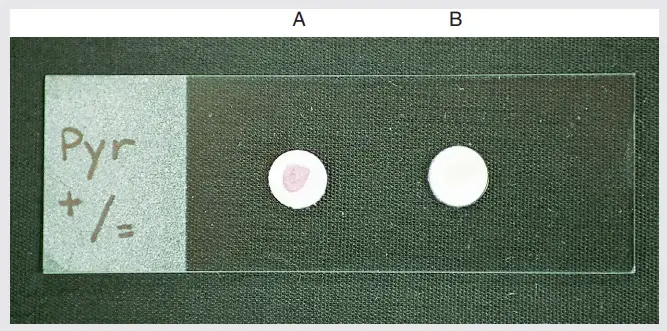
Results of PYR Test
Positive result–
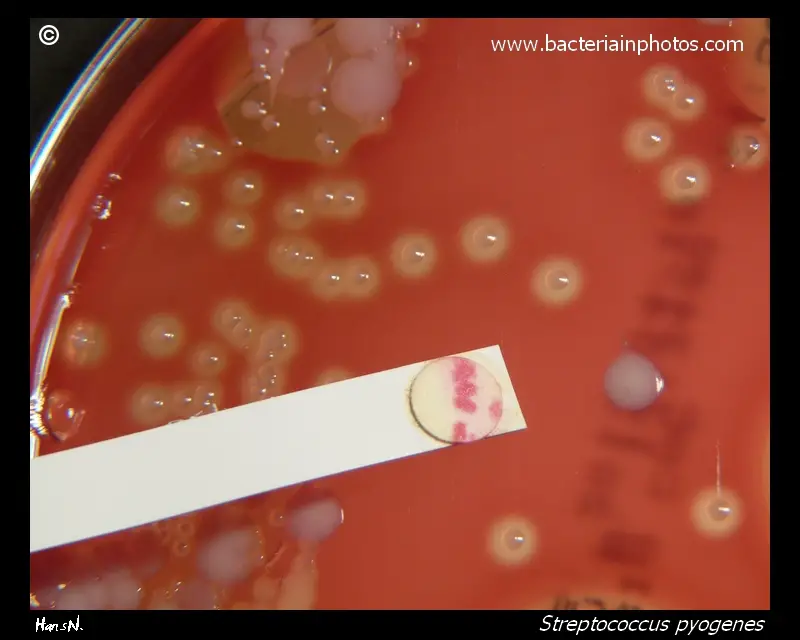
After adding PYR reagent bright pink or cherry-red colour is produced within 1–2 minutes. This indicates PYR positive.
PYR positive organism are–
- Group A streptococci like Streptococcus pyogenes.
- Enterococcus species like Enterococcus faecalis and Enterococcus faecium.
- Coagulase negative staphylococci like Staphylococcus haemolyticus Staphylococcus lugdunensis and Staphylococcus schleiferi.
- Staphylococcus intermedius group (SIG) like Staphylococcus pseudintermedius Staphylococcus delphini and Staphylococcus intermedius.
- Some Gram negative bacteria like Citrobacter Klebsiella Enterobacter Yersinia and Serratia species.
- Other organism like Aerococcus Gemella Lactococcus and Corynebacterium (Arcanobacterium) haemolyticum.
Negative result–
After adding PYR reagent no colour change is seen. Cream yellow orange or salmon colour may appear. Pale or faint pink colour is also taken as negative. Blue or blue-green colour can be produced due to indole cross reaction and it is interpreted as negative PYR.
PYR negative organism are–
- Group B streptococci like Streptococcus agalactiae.
- Group D non enterococci like Streptococcus bovis and Streptococcus equinus.
- Other Streptococcus species like Group C Group F Group G S. pneumoniae S. mitis S. milleri and viridans group streptococci.
- Staphylococcus aureus.
- Escherichia coli.
- Salmonella species.

PYR positive and PYR negative organism are shown in the following table–
| Category | Organism / Species |
|---|---|
| PYR Positive | Group A streptococci – Streptococcus pyogenes |
| Enterococcus species – Enterococcus faecalis, Enterococcus faecium, Enterococcus avium, Enterococcus durans | |
| Coagulase negative staphylococci – Staphylococcus haemolyticus, Staphylococcus lugdunensis, Staphylococcus schleiferi | |
| Staphylococcus intermedius group (SIG) – Staphylococcus pseudintermedius, Staphylococcus delphini, Staphylococcus intermedius | |
| Other streptococci – Streptococcus iniae, Streptococcus porcinus | |
| Some Gram negative bacteria – Citrobacter, Klebsiella, Enterobacter, Yersinia, Serratia species | |
| Other organism – Aerococcus viridans, Lactococcus garvieae, Gemella and most Corynebacterium (Arcanobacterium) haemolyticum | |
| PYR Negative | Group B streptococci – Streptococcus agalactiae |
| Group D non enterococci – Streptococcus bovis, Streptococcus equinus | |
| Other Streptococcus – Groups C F and G streptococci Streptococcus pneumoniae Streptococcus mitis Streptococcus milleri and viridans group streptococci | |
| Staphylococcus aureus | |
| Some coagulase negative staphylococci – Staphylococcus auricularis, Staphylococcus saprophyticus | |
| Escherichia coli | |
| Salmonella species |
Limitations of PYR Test
- It is a presumptive test only. It is used for preliminary identification and other biochemical serological or morphological test are required for confirmation.
- In rapid disk method heavy visible inoculum (paste) is required. If less inoculum is taken faint pink or false negative result may be obtained because enzyme is not sufficient.
- The disk should be moistened slightly. If the disk is flooded or over saturated the enzyme and hydrolyzed product can be diluted and false negative result is seen.
- The inoculum should be taken from pure colonies grown on non selective media like Blood agar. If inoculum is taken from selective media or tube biochemical agar enzyme synthesis may be suppressed and false negative reaction is produced.
- Indole producing organism (Escherichia coli Proteus) may give blue or blue-green colour due to indole cross reaction when grown on tryptophan rich media. It is interpreted as negative PYR but it can create confusion.
- Proper timing is important. If it is read too early the result can be inaccurate. If it is observed after 2–5 minutes non specific colour may develop and false positive can be interpreted.
- Some Group A streptococci (about 1–2%) may naturally lack the enzyme so false negative is obtained. Some Staphylococcus aureus may show weak pale pink reaction and it needs further confirmation (tube test).
- Some other organism also gives PYR positive like Aerococcus Lactococcus some Enterobacteriaceae and Corynebacterium (Arcanobacterium) haemolyticum. So it should be used in proper clinical context usually after confirming isolate is Gram positive catalase negative coccus.
- PYR reagent and disk are sensitive. It should be protected from light and stored at 2–8°C. Improper storage can lead to degradation and unreliable result.
Uses of PYR Test
Uses of PYR test are–
- It is used for rapid presumptive identification of Group A beta hemolytic streptococci like Streptococcus pyogenes.
- It is used for differentiating Enterococcus species (PYR positive) from Group D non enterococci like Streptococcus bovis (PYR negative).
- It is used for identification and differentiation of some coagulase negative staphylococci like Staphylococcus haemolyticus Staphylococcus lugdunensis and Staphylococcus schleiferi.
- It is used for distinguishing Staphylococcus aureus (usually PYR negative) from Staphylococcus intermedius group (SIG) which gives PYR positive.
- It is used for helping in confirmation of Escherichia coli (PYR negative) by separating it from other Gram negative indole positive lactose positive rods.
- It is used as a secondary marker for differentiating Enterobacteriaceae like Citrobacter species (PYR positive) from Salmonella species (mostly PYR negative).
- It is used for rapid identification (within few minutes) so targeted antimicrobial therapy is started and unnecessary broad spectrum antibiotic is avoided.
References
- Aryal, S. (2022, August 10). PYR Test- Principle, Uses, Procedure and Result Interpretation. Microbiology Info.
- Bhavana, R. N., & Chaitanya, R. K. (2022). Identification of coagulase negative staphylococcal species from bovine mastitis in India. Iranian Journal of Veterinary Research, 23(4), 358–362. https://doi.org/10.22099/IJVR.2022.43698.6406
- Chagla, A. H., Borczyk, A. A., Aldom, J. E., Rosa, S. D., & Cole, D. D. (1993). Evaluation of the L-pyrrolidonyl-beta-naphthylamide hydrolysis test for the differentiation of members of the families Enterobacteriaceae and Vibrionaceae. Journal of Clinical Microbiology, 31(7), 1946–1948.
- Chauhan, A. (2025, May 16). PYR Test: Staphylococcus & Bacterial Identification Guide. Flabs.
- Comprehensive Analysis of the Pyrrolidonyl Arylamidase (PYR) Test: Biochemical Principles, Clinical Application, and Diagnostic Paradigms. (n.d.).
- Compton, S. T., Kania, S. A., Robertson, A. E., Lawhon, S. D., Jenkins, S. G., Westblade, L. F., & Bemis, D. A. (2017). Evaluation of Pyrrolidonyl Arylamidase Activity in Staphylococcus delphini. Journal of Clinical Microbiology, 55(3), 859–864. https://doi.org/10.1128/JCM.02076-16
- Dahal, P. (2024, December 19). PYR Test: Principle, Media, Steps, Results, Uses. Microbe Notes.
- Dalynn Biologicals. (2014). PYR DISKS.
- De Paulis, A. N., Predari, S. C., Chazarreta, C. D., & Santoianni, J. E. (2003). Five-Test Simple Scheme for Species-Level Identification of Clinically Significant Coagulase-Negative Staphylococci. Journal of Clinical Microbiology, 41(3), 1219–1224. https://doi.org/10.1128/JCM.41.3.1219-1224.2003
- El Sanousi, S. M., Said, K. B., Elbager, S. G., Awad, A., Rodwan, K., & Eltom, K. H. (2015). A flow chart for the identification of Staphylococcus species. U. of K. J. Vet. Med. Anim. Prod., 6(2), 93-97.
- Facklam, R., Elliott, J., Shewmaker, L., & Reingold, A. (2005). Identification and Characterization of Sporadic Isolates of Streptococcus iniae Isolated from Humans. Journal of Clinical Microbiology, 43(2), 933–937. https://doi.org/10.1128/JCM.43.2.933-937.2005
- Hardy Diagnostics. (2020). PYR TEST KIT AND PYR REAGENT Instructions for Use.
- HiMedia Laboratories. (n.d.). PYR Reagent.
- Iorio, N. L. P., Ferreira, R. B. R., Schuenck, R. P., Malvar, K. L., Brilhante, A. P., Nunes, A. P. F., Bastos, C. C. R., & Santos, K. R. N. (2007). Simplified and Reliable Scheme for Species-Level Identification of Staphylococcus Clinical Isolates. Journal of Clinical Microbiology, 45(8), 2564–2569. https://doi.org/10.1128/JCM.00679-07
- ITEST plus, s.r.o. (n.d.). ITEST PYR test.
- Liofilchem. (2024, September 16). Peptidase A Test Instructions for Use.
- LSU School of Medicine. (1996). DM Lab 13: Gram Positive Pyogenic Cocci.
- MAST Group. (2020, June). MAST ID PYR Strips ETO7.
- Microbiology. (n.d.). Rapid Presumptive ID.
- Microxpress. (n.d.). PYR Reagent Intended Use.
- Pokhrel, P. (2015, July 7). PYR ( L-Pyrrolidonyl-β-Naphthylamide) Test- Principle, Uses, Procedure, Result Interpretation, Examples and Limitation. Microbiology Notes.
- Remel Inc. (2008, November 6). STREP ID QUAD.
- Remel Inc. (2009, August 31). PYR REAGENT.
- Remel Inc. (2021, March). PYR DISK w/ REAGENT Instructions for Use. ThermoFisher Scientific.
- Wikipedia contributors. (2025, October 22). Pyrrolidonyl-β-naphthylamide. In Wikipedia, The Free Encyclopedia.