Picric Acid Test is a chemical test. It is used for detection of reducing sugar in sample. These reducing sugar are glucose fructose and galactose.
It is a highly sensitive assay. It is based on the reducing nature of sugar. The sugar contain free aldehyde or ketone group. This group act as reducing agent in alkaline medium. The alkaline condition is formed by adding sodium carbonate.
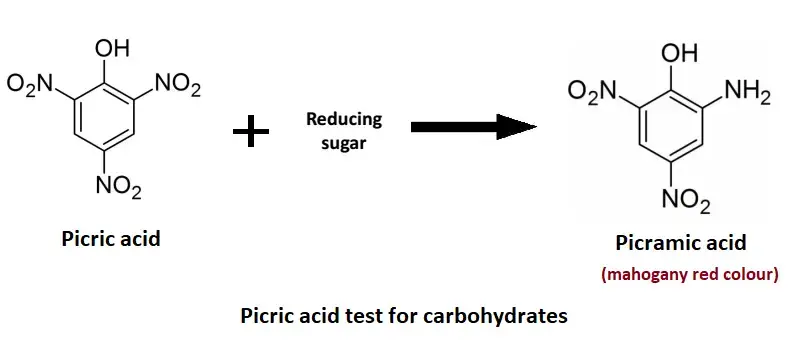
In this test the sample is heated with picric acid. Picric acid is a toxic yellow crystalline solid. If reducing sugar are present the yellow picric acid is reduced to picramic acid. The reaction is shown by formation of mahogany red colour.
Positive test is indicated by colour change from yellow to mahogany red. Negative test is indicated by no colour change and solution remain yellow. Non reducing carbohydrate such as sucrose starch and glycogen do not give this reaction.
It was used earlier in clinical blood and urine analysis. It is also used in food science for estimation of sugar and mineral composition in products like honey.
Objectives of Picric Acid Test
Objectives of Picric Acid Test are as follows–
- It is used for detection of reducing sugars such as glucose fructose and galactose in plant extract food products and honey.
- It is used for estimation of creatinine level in animal fluids human serum and urine (Jaffe reaction). It is important for kidney function test.
- It is used for detection of potassium ion (K+) when test is done in non alkaline or slightly acidic medium. Yellow precipitate is formed in presence of potassium.
- It is used for determination of hydrocyanic acid (HCN) or potassium cyanide in minute amount. It react with alkaline picric acid to form blood red isopurpuric acid.
- It is used for screening of alkaloids in plant extract. This is also referred to as Haggar’s test.
Principle of Picric Acid Test
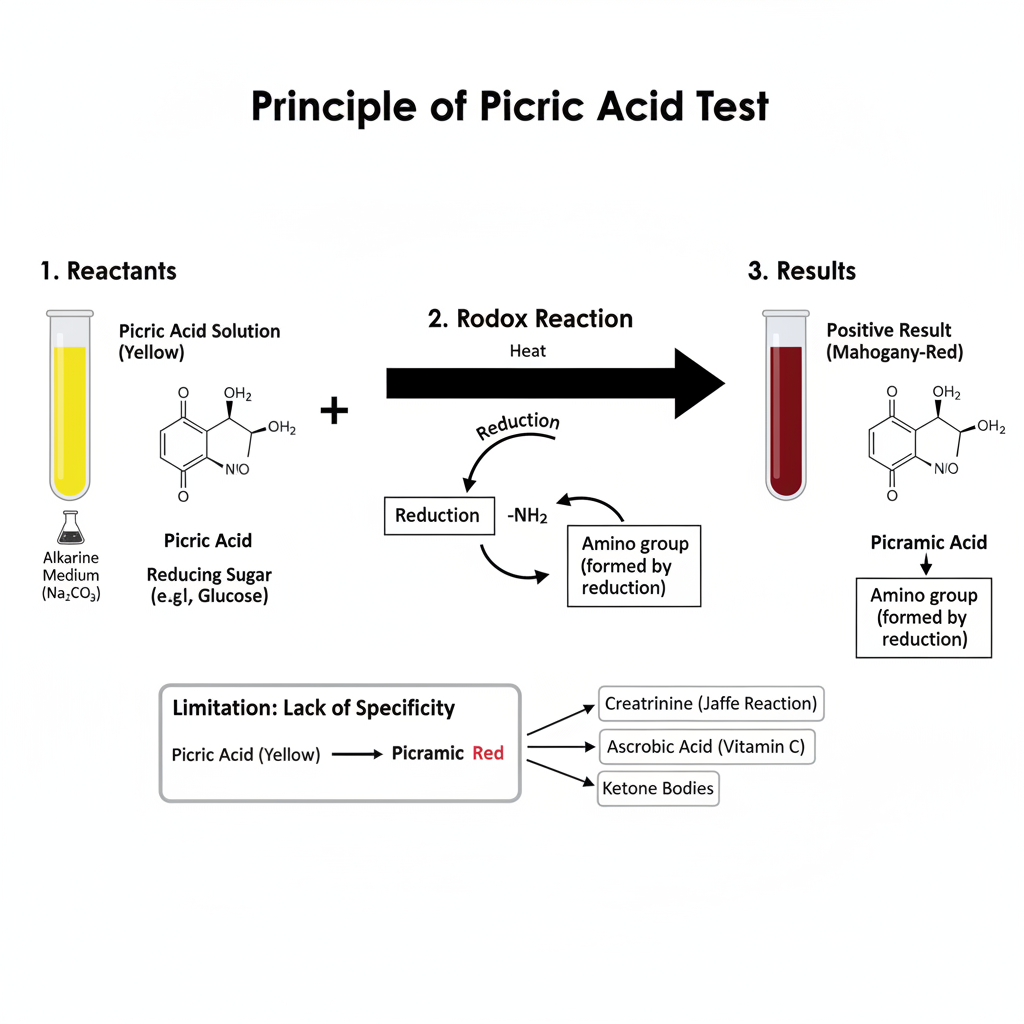
Principle of Picric Acid Test is based on oxidation reduction reaction. It is used to detect reducing sugar in given sample.
In alkaline medium the reducing sugar show reducing property. The alkaline medium is produced by addition of sodium carbonate. The reducing sugar contain free aldehyde or ketone group. These group is converted to reactive enediol form in alkaline condition.
On heating the reducing sugar reduce the picric acid. Picric acid (2,4,6-trinitrophenol) is yellow crystalline compound. It is reduced to picramic acid which is mahogany red colour.
The colour change from yellow to mahogany red is the indication of presence of reducing sugar. Non reducing sugar do not contain free aldehyde or ketone group so colour change is not produced and solution remain yellow.
Requirements for Picric Acid Test
Requirements for Picric Acid Test are as follows–
Reagents and solutions
- Saturated picric acid solution. It is prepared by dissolving 13 g picric acid in distilled water. It is boiled and then allowed to cool.
- Sodium carbonate solution (10% or 20%). It is added for making alkaline medium.
- Test sample. It may be 1% carbohydrate solution or natural sample like honey.
- Standard sugar solution (for quantitative test). It is required when exact quantity of sugar is to be measured (Lewis-Benedict method). It may be 0.08% or 0.1% glucose solution.
Apparatus and equipment
- Test tubes. Clean and heat resistant test tubes are used for mixing sample and reagents.
- Boiling water bath. It is used for heating the test tubes for few minutes so reaction is completed.
Procedure of Picric Acid Test
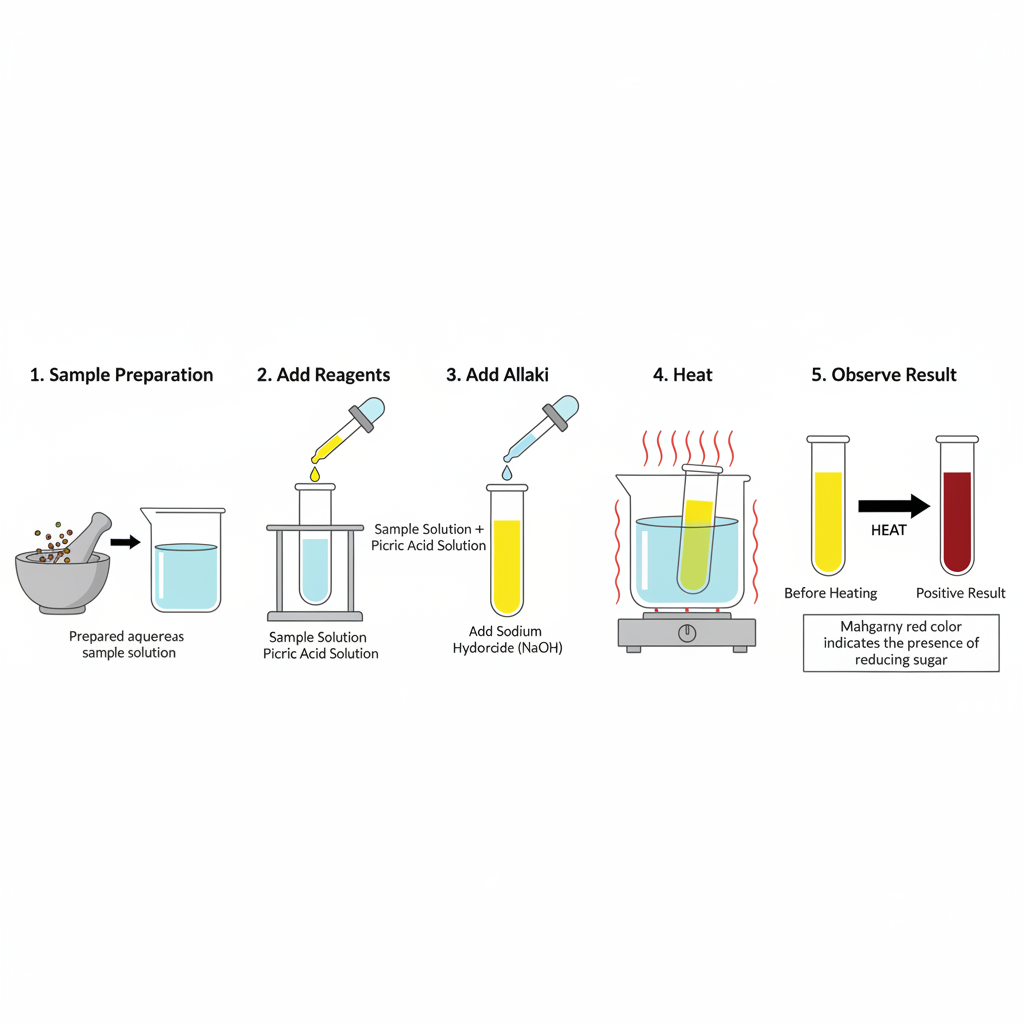
Procedure of Picric Acid Test are as follows–
- Step 1- Take 1 ml of sample solution (1% sugar solution or test sample) in a clean heat resistant test tube.
- Step 2- Add 1 ml of saturated picric acid solution in the same test tube.
- Step 3- Add 0.5 ml of 10% sodium carbonate solution. It is added for making alkaline medium. For Lewis-Benedict method 1 ml of 20% sodium carbonate is used.
- Step 4- The test tube is kept in boiling water bath (or heated at 60–70°C) for 2 to 5 minutes. For strict quantitative test the heating time is increased to 20 to 30 minutes so reduction reaction is completed.
- Step 5- The test tube is removed and colour is observed. Mahogany red colour indicate positive test for reducing sugar. In quantitative test the solution is cooled and diluted to fixed volume (10 ml) and absorbance is measured by spectrophotometer or colorimeter at 490 nm or 540 nm.
Result of Picric Acid Test

Result of Picric Acid Test are as follows–
Positive result (reducing sugar present)
- Mahogany red solution or red to brownish red colour is formed.
- Glucose– Red colour is observed.
- Galactose– Red colour is observed.
- Fructose– Red colour is observed.
- Lactose– Red colour is observed.
- Maltose– Brownish red colour is observed.
- Xylose– Mahogany red solution is observed.
Negative result (reducing sugar absent)
- No colour change is seen and solution remain yellow. No precipitation is formed.
- Sucrose– No precipitation or colour change.
- Glycogen– No precipitation or colour change.
- Starch– No precipitation or colour change.

Uses of Picric Acid Test
Uses of Picric Acid Test are as follows–
- It is used for detecting reducing sugars such as glucose fructose and galactose in food products plant extracts and honey.
- It is used for estimation of creatinine level in animal fluids human serum and urine. This is also known as Jaffe reaction.
- It was used earlier for measurement of blood glucose level in early 20th century.
- It is used for detecting potassium ion (K+) in samples like honey. Yellow precipitate is formed.
- It is used for determination of hydrocyanic acid (HCN) in minute amount (example fumigated insects studies).
- It is used for screening of alkaloids in plant extracts. This is referred to as Haggar’s test.
- It is used in gravimetric analysis for detection and quantification of alkaline earth metal ions such as calcium magnesium and barium. Insoluble picrate salts is formed and weighed.
- It is used in beverage industry for calculating sucrose content. Total reducing power of fruit juices and soft drinks is measured.
Advantages of Picric Acid Test
Advantages of Picric Acid Test are as follows–
- It is highly sensitive test. Minute quantity of reducing sugar is detected which may not be detected by copper reduction methods. It is also sensitive for trace amount of hydrocyanic acid.
- It is suitable for spectrophotometric estimation. The reaction give clear homogeneous mahogany red solution. Benedict and Fehling test produce opaque precipitate so precise colour measurement is difficult.
- Small volume of sample is required. Very less sample (example 0.2 ml blood) can be used which is useful for repeated measurement.
- Picric acid reagent is stable in solution. Fehling solution is required to be prepared freshly before test.
- Mild alkaline condition is used. Sodium carbonate is used for alkaline medium which is less corrosive than sodium hydroxide used in Fehling test. Sugar degradation by over oxidation is reduced.
- Reoxidation error is avoided. Uncontrollable reoxidation of cuprous oxide which occur in copper based methods is not seen.
- It is cost effective method. Picric acid and phenolic analogues are inexpensive and easily available for laboratory use.
Limitations of Picric Acid Test
Limitations of Picric Acid Test are as follows–
- It is not absolutely specific. The test is based on general nitro group reduction so false positive result can be formed and it is not only specific for glucose.
- Metabolic byproducts can interfere. Creatinine give red orange complex (Jaffe reaction) and uric acid acetone and acetoacetic acid can react with alkaline picrate causing overestimation of reducing sugar.
- Drugs can interfere. Ascorbic acid (Vitamin C) levodopa and beta lactam antibiotics (cephalosporins penicillins) act as reducing agent and mahogany red colour can be produced falsely.
- Bilirubin show negative interference in serum. It is oxidized to biliverdin in alkaline medium which changes absorbance and true result can be masked.
- Physical nature of sample affect reaction. Highly buffered alkaline or acidic urine and sample having high specific gravity can inhibit reaction or early colour formation can occur.
- Quantitative test is time consuming. Strict boiling step of 20 to 30 minutes is required so it is slower than modern enzymatic methods and automated analyser.
- Dry picric acid is explosive hazard. It is secondary high explosive and sensitive to heat shock and friction. Metal picrates can be formed with metal caps or shelving which are more unstable.
- Picric acid is toxic. Dermatitis and yellow staining of skin and hair can occur on contact. Ingestion or chronic exposure can cause gastrointestinal distress liver damage kidney damage and DNA damage (mutagenic effect).
Precautions of Picric Acid Test
Precautions of Picric Acid Test are as follows–
- Picric acid should not be allowed to dry. Dry picric acid is secondary high explosive and it is sensitive to heat shock and friction. It should be kept moistened with minimum 30% water.
- Reagent bottle should be stored carefully. Dry crust should not be formed around the cap.
- Metal contact should be avoided. Metal caps should not be used and it should not be kept on metal shelving. Metal picrates (lead iron copper picrates) can be formed which are more unstable and friction sensitive.
- Protective measures should be followed. Gloves and goggles is worn because skin and eye contact can cause severe dermatitis. Yellow or dark brown staining of skin and hair can occur for long time.
- Ingestion and skin absorption should be avoided. Nausea vomiting diarrhoea abdominal pain can occur and long term damage to liver kidney and respiratory organs can be caused.
- Adequate ventilation is required. It should be handled in well ventilated area (suction cupboard or fume hood) because fumes inhalation is harmful.
- Mutagenic risk should be considered. It should be handled with extreme caution because it is potential mutagen and DNA damage can be caused in living cells.
References
- Archana, A., Khutal, T., Bhand, A., & Gaikwad, S. (2025). A comprehensive review on picric acid: Synthesis, properties, and applications. International Journal of Pharmaceutical Sciences, 3(5), 5226-5231. https://doi.org/10.5281/zenodo.15564233
- Calo, F. (n.d.). Picric acid test and iodine test. Scribd.
- Fabin, M., Łapkowski, M., & Jarosz, T. (2023). Methods for detecting picric acid—A review of recent progress. Applied Sciences, 13(6), 3991. https://doi.org/10.3390/app13063991
- Hameed, O. M., Shaker, O. M., Ben Slima, A., & Makni, M. (2024). Biochemical profiling and physicochemical and biological valorization of Iraqi honey: A comprehensive analysis. Molecules, 29(3), 671. https://doi.org/10.3390/molecules29030671
- HOly makaroni. (n.d.). Picric acid test for reducing sugars. Scribd.
- Lolekha, P. H., Jaruthunyaluck, S., & Srisawasdi, P. (2001). Deproteinization of serum: Another best approach to eliminate all forms of bilirubin interference on serum creatinine by the kinetic Jaffe reaction. Journal of Clinical Laboratory Analysis, 15(3), 116-121. https://doi.org/10.1002/jcla.1013
- Saleem, A., Rafi, N., Qasim, S., Ashraf, U., & Virk, N. H. (2019). Synthesis of picric acid at domestic scales. International Journal of Innovations in Science and Technology, 1(2), 62-78. https://doi.org/10.33411/IJIST/2019010202
- Sinclair, W. B., & Ramsey, R. C. (1944). The picric acid method for determining minute amounts of hydrocyanic acid in fumigated insects. University of California Agriculture and Natural Resources Repository.
- Viraraghavan, S., & Blass, K. G. (1990). Effect of glucose upon alkaline picrate: A Jaffé interference. Journal of Clinical Chemistry and Clinical Biochemistry, 28(2), 95-105. https://doi.org/10.1515/cclm.1990.28.2.95
- VitaeGen Biotech. (n.d.). CARBOHYDRATE TEST PART 3- Anthrone test | Picric Acid Test | Carbohydrate Test [Video]. YouTube.
- Willaman, J. J., & Davison, F. R. (1924). Some modifications of the picric acid method for sugars. Journal of Agricultural Research, 28(5), 479-488.