What is Phenol-Sulfuric Acid Method for Total Carbohydrates?
Phenol-sulfuric acid method is a colorimetric method which is used for quantitative estimation of total carbohydrates. It was formalized by DuBois in 1956. It is simple rapid and reliable method. It can detect almost all type of carbohydrates like monosaccharides disaccharides oligosaccharides and polysaccharides.
In this method concentrated sulphuric acid is used as hydrolytic and dehydrating agent. The complex carbohydrate is hydrolysed to simple sugars and then dehydration is carried out. The sulphuric acid produces heat (exothermic reaction) and it helps in reaction. Pentose sugars are converted to furfural and hexose sugars are converted to hydroxymethylfurfural.
After this phenol reagent is added. The furfural and hydroxymethylfurfural condenses with phenol and yellow-orange (yellow-gold) coloured complex is formed. The intensity of colour formed is directly proportional to carbohydrate concentration in sample.
The absorbance is measured by spectrophotometer. Maximum absorbance is taken at 490 nm for hexoses and 480 nm for pentoses. The concentration is calculated by comparing sample absorbance with standard calibration curve. Glucose is commonly used as standard carbohydrate for calculation.
Objective of Phenol-Sulfuric Acid Method
Objective of phenol-sulphuric acid method are as follows-
- Detection of sugars– It is used to detect presence of carbohydrates (sugars) in given sample or solution.
- Quantitative estimation– It is used to measure total carbohydrate concentration (free as well as bound carbohydrates).
- Industrial and clinical analysis– It is used for determination of carbohydrate content in different applications like clinical biochemistry nutritional labelling metabolic studies and drug development.
- Biomass characterization– It is used as rapid screening method for total carbohydrate in biomass (example microalgae) for environmental monitoring and biofuel research.
- Chromatography monitoring– It is used to monitor column eluates for sugar containing compounds. Example lipopolysaccharides (LPS) and O-antigens can be detected during elution.
Principle of Phenol-Sulfuric Acid Method
Principle of phenol-sulphuric acid method is based on acid hydrolysis dehydration and colour formation. It is a colorimetric method for estimation of total carbohydrates. In this method concentrated sulphuric acid is used and it gives heat during dilution. This heat and acid hydrolyses complex carbohydrate into monosaccharides.
After hydrolysis the strong acid dehydrates the simple sugars. Pentose sugars (5 carbon) are converted to furfural. Hexose sugars (6 carbon) are converted to 5-hydroxymethylfurfural (HMF). These are reactive intermediate compounds and it is formed during dehydration step.
In second step phenol reacts with these furfural derivatives. Condensation reaction occurs and a stable yellow-orange or orange-red coloured complex is formed. The intensity of colour formed is directly proportional to total carbohydrate concentration present in sample.
The absorbance is measured by spectrophotometer. It is read at 490 nm for hexose rich sample and 480 nm for pentose rich sample. By this absorbance the carbohydrate concentration is calculated.
Requirements for Phenol-Sulfuric Acid Method
Requirements for phenol-sulphuric acid method are as follows-
Reagents and solutions are-
- Phenol solution– Usually 5% (w/v) aqueous phenol is prepared (some protocol uses 80% solution also).
- Concentrated sulphuric acid– Reagent grade concentrated acid is used (96–98%).
- Standard carbohydrate solution– Known concentration standard is used for calibration curve (glucose or glycogen is commonly used).
- Distilled or deionized water– It is used for preparation of solution and dilution of sample.
Equipment are-
- Spectrophotometer or microplate reader– It is used to measure absorbance at 480–490 nm.
- Vortex mixer– It is used for proper mixing of reagents and sample.
- Water bath or heat block– It is used to maintain temperature (25–30°C for colour stabilisation or heating upto 90–100°C depending on protocol).
Laboratory supplies are-
- Reaction vessels– Clean dry test tubes (16–20 mm internal diameter) with rack or acid compatible 96 well microtiter plate.
- Cuvettes– It is required if spectrophotometer is used.
- Pipetting tools– Adjustable micropipette with tips volumetric pipette Pasteur pipette or repipettor (fast delivery of acid is needed).
- Glassware– Conical flask (Erlenmeyer) and volumetric flasks are used for sample and standard preparation.
- Parafilm– It is used for sealing during mixing.
PPE and safety are-
- Gloves– Double gloves or acid resistant gloves (nitrile rubber or neoprene) is used (phenol is toxic and acid is corrosive).
- Eye and face protection– Safety glasses goggles or face shield is used.
- Body protection– Lab coat is used (rubber or neoprene apron can be used if splash risk is present).
- Fume hood– It is required for handling phenol and concentrated sulphuric acid safely.
Procedure of Phenol-Sulfuric Acid Method
Procedure of phenol-sulphuric acid method is as follows-
- Sample and standard are prepared. Unknown sample solution is prepared in water. Standard solutions (glucose series) are prepared and Blank is kept with distilled water. A fixed volume is pipetted in separate test tubes (example 1.0 mL).
- 5% phenol solution is added in each tube (example 1.0 mL) and it is mixed properly by vortex mixer.
- Concentrated sulphuric acid is added carefully and rapidly (example 5.0 mL) in a stream directly on liquid surface in each tube. The heat produced during dilution (exothermic) drives the reaction so rapid addition is important.
- The tubes are kept at room temperature for about 10 minutes.
- After this tubes are vortexed again and it is incubated in water bath at 25–30°C for 20 minutes so colour is developed uniformly and it is stabilised.
- The tubes are cooled to room temperature. Blank is used to set zero in spectrophotometer and absorbance is measured at 490 nm (or 480 nm for pentose/uronic acid).
- Standard calibration curve is prepared by standard absorbance and concentration. The unknown sample carbohydrate concentration is calculated from the curve.
Result and calculation of phenol-sulphuric acid method
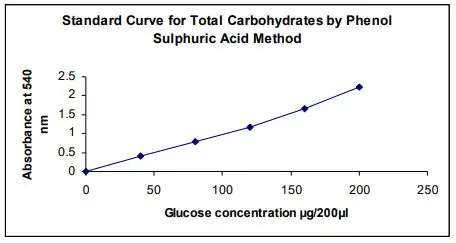
Result and calculation of phenol-sulphuric acid method are as follows-
- Result is obtained as absorbance (O.D.) value. It is read in spectrophotometer at 490 nm for hexose rich sample and 480 nm for pentose rich sample.
- Standard curve is prepared. Absorbance of standard (glucose) is plotted in Y-axis and concentration is plotted in X-axis.
- Linear equation is obtained from curve. The equation is as follows-
y = mx + c
(y is absorbance. x is carbohydrate concentration. m is slope. c is intercept) - Concentration of unknown sample is calculated by equation-
x = (y – c) / m
(y is absorbance of unknown sample) - If dilution is done then dilution factor is multiplied. Dilution factor is calculated as (final volume / initial volume). Final concentration = calculated concentration × dilution factor.
- If different monosaccharides are present then correction factor can be applied because colour intensity differs from glucose. Corrected sugar content is calculated as-
W = C × ∑(fi × Pi)
(C is initial calculated concentration. fi is correction factor. Pi is mass percentage of each sugar component) - Final result is expressed in unit like µg/mL or mg/g of original sample or µg/µg of protein (as per sample type and requirement).

Advantages of Phenol-Sulfuric Acid Method
Advantages of phenol-sulphuric acid method are as follows-
- Simple and rapid method– It is easy to perform and protocol is simple. Time required is less as compared to many other methods.
- High sensitivity and reliable– It is sensitive method and microgram quantity of carbohydrate can be detected. The result is reproducible and reliable.
- Broad applicability– Almost all classes of carbohydrates can be detected. Complex polysaccharides are broken down and total saccharides are measured.
- Stability– Reagents are stable and the yellow-orange coloured complex formed remains stable for several hours so reading can be taken with flexibility.
- Cost effective– It is inexpensive method as compared to enzymatic assay or HPLC method.
- High throughput use– It is suitable for large number of samples. It can be adapted to microplate format and screening can be done easily.
Limitations of Phenol-Sulfuric Acid Method
Limitations of phenol-sulphuric acid method are as follows-
- Lack of specificity– It gives total carbohydrate value but it cannot differentiate individual sugars present in mixture.
- Different colour response of sugars– Pentose hexose and uronic acid does not give same colour intensity at same concentration. If glucose is used as single standard then mixed sugar sample may give error (overestimation or underestimation).
- Interference by pigments and phytochemicals– Chlorophyll carotenoids (astaxanthin β-carotene) anthocyanins catechins and tannins can also develop colour with hot sulphuric acid. Due to this carbohydrate value may be shown very high.
- Oxidative interference– Residual oxidants in some sample (example persulfate in treated sludge) can interfere with colour development and result is affected.
- Matrix interference– High protein or lipid in sample can produce turbidity and spectrophotometric reading becomes inconsistent.
- Precision problem– The reaction depends on exothermic heat produced during rapid addition of sulphuric acid. If temperature acid addition and mixing is not controlled properly then reproducibility is reduced and error between replicates is increased.
- Safety hazard– Concentrated sulphuric acid is corrosive and it generates heat and splashing risk is present. Phenol is also toxic and corrosive and it can be absorbed through skin so careful handling is required.
Uses of Phenol-Sulfuric Acid Method
Uses of phenol-sulphuric acid method are as follows-
- General carbohydrate estimation– It is used for total carbohydrate and neutral sugar estimation. Glycogen and starch estimation is also done. It is used in oligosaccharides proteoglycans glycoproteins and glycolipids analysis.
- Food and beverage analysis– It is used to determine total carbohydrate for nutritional labelling and caloric calculation in products like soft drinks beer and fruit juices.
- Drug and vaccine development– It is used to evaluate glycosylation of therapeutic proteins. It is used to quantify capsular polysaccharides in glycoconjugate vaccine development and quality control (example pneumococcal vaccines).
- Biofuel research– It is used as rapid screening method to estimate total carbohydrate in algal and cyanobacterial biomass for bioethanol and biofuel production.
- Clinical and biological study– It is used in clinical biochemistry for carbohydrate estimation in body fluids like serum plasma and urine. It also supports metabolic studies.
- Environmental monitoring– It is used to estimate carbohydrate in environmental samples like marine sediments and treated wastewater sludge.
- Plant and fungal research– It is used for routine estimation of sugars and starch in woody plant tissues and fungi. It is also used for quality control and standardization of herbal medicines.
- Chromatography monitoring– It is used to monitor column eluates for sugar containing compounds. Example lipopolysaccharides (LPS) and O-antigens are detected during chromatography.
References
- BenchChem Technical Support Team. (2025). A comparative guide to glycogen quantification: Anthrone assay vs. alternatives. BenchChem.
- BenchChem Technical Support Team. (2025). Application notes and protocols for spectrophotometric determination of carbohydrates using the phenol-sulfuric acid method. BenchChem.
- BMH learning. (n.d.). Estimation of total carbohydrates by phenol sulphuric acid method | Total carbohydrate determination [Video]. YouTube.
- Cell Biolabs, Inc. (2015). Total carbohydrate assay kit (STA-682) product manual.
- ChemicalBook. (2022). Phenol sulfuric acid method.
- Chen, W., Gao, L., Song, L., Sommerfeld, M., & Hu, Q. (2023). An improved phenol-sulfuric acid method for the quantitative measurement of total carbohydrates in algal biomass. Algal Research, 70, 102986. https://doi.org/10.1016/j.algal.2023.102986
- Dr. Raki’s Just Biology. (n.d.). Estimation of total carbohydrates by phenol-sulphuric acid method//Microplate method//Standard graph [Video]. YouTube.
- Fan, J. (2015). Phenol-sulfuric acid mannose detection assay. Bowdish Lab, McMaster University.
- Fuentes, M. (n.d.). Dilution factor calculator. Hemocytometer.
- Hancock Lab. (n.d.). Phenol sulphuric acid carbohydrate assay.
- Masuko, T., Minami, A., Iwasaki, N., Majima, T., Nishimura, S.-I., & Lee, Y. C. (2005). Carbohydrate analysis by a phenol-sulfuric acid method in microplate format. Analytical Biochemistry, 339(1), 69–72. https://doi.org/10.1016/j.ab.2004.12.001
- Mettler Toledo. (n.d.). Determine total carbohydrate content by phenol sulphuric acid method.
- Natsuka, S. (2021). Color reaction of carbohydrates. In S. Nishihara, K. Angata, K. F. Aoki-Kinoshita, et al. (Eds.), Glycoscience protocols (GlycoPODv2). Japan Consortium for Glycobiology and Glycotechnology.
- Nicacio, S., Balasundaram, W. U., Bhingarkar, A., Cho, D., Ghayal, R., Datta, A., & Kapre, S. V. (2026). Automation of polysaccharide quantification: A rapid high-throughput assay enabled by liquid handling technology. BioTech, 15(1), 24. https://doi.org/10.3390/biotech15010024
- Nielsen, S. S. (2017). Total carbohydrate by phenol-sulfuric acid method. In S. S. Nielsen (Ed.), Food analysis laboratory manual (pp. 137–141). Springer. https://doi.org/10.1007/978-3-319-44127-6_14
- Nielsen, S. S. (2024). Total carbohydrate by phenol-sulfuric acid method. In S. S. Nielsen (Ed.), Nielsen’s food analysis laboratory manual (pp. 147–151). Springer. https://doi.org/10.1007/978-3-031-44970-3_17
- Prasanna, K., Narayanamma, N., Satya Sai, L. J. N. B., Prasad, I., Idupularajesh, & Teja, K. V. (2025). Estimation of carbohydrates by anthrone method in different juices. Indo American Journal of Pharmaceutical Sciences, 12(3), 137–140. https://doi.org/10.5281/zenodo.15020597
- Quansys Biosciences. (2023). Dilutions: Explanations and examples of common methods.
- Sigma-Aldrich. (2020). Total carbohydrate assay kit (MAK104) technical bulletin.
- Sigma-Aldrich. (2024). Total carbohydrate assay kit (MAK559) technical bulletin.
- Yue, F., Zhang, J., Xu, J., Niu, T., Lü, X., & Liu, M. (2022). Effects of monosaccharide composition on quantitative analysis of total sugar content by phenol-sulfuric acid method. Frontiers in Nutrition, 9, 963318. https://doi.org/10.3389/fnut.2022.963318
- Zavřel, T., Očenášová, P., Sinetova, M. A., & Červený, J. (2018). Determination of storage (starch/glycogen) and total saccharides content in algae and cyanobacteria by a phenol-sulfuric acid method. Bio-protocol, 8(15), e2966. https://doi.org/10.21769/BioProtoc.2966
- Zhang, W.-H., Wu, J., Weng, L., Zhang, H., Zhang, J., & Wu, A. (2020). An improved phenol-sulfuric acid method for the determination of carbohydrates in the presence of persulfate. Carbohydrate Polymers, 227, 115332. https://doi.org/10.1016/j.carbpol.2019.115332