What is Osazone Test?
Osazone test is a chemical test which is used for detection and differentiation of reducing sugars. Reducing sugar contains free carbonyl group (or potentially free carbonyl group) and it gives positive reaction. This test was developed by Emil Fischer. It is based on formation of osazone crystals and these crystals are observed under microscope.
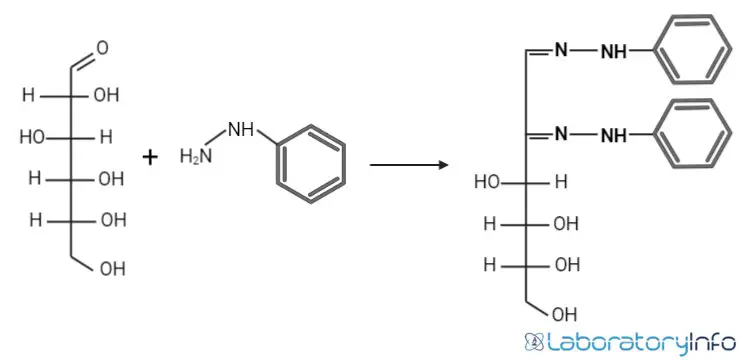
In this test the sugar solution is heated with excess phenylhydrazine reagent at boiling temperature. During reaction first two carbon atoms (C1 and C2) of sugar are involved. Condensation and oxidation occurs and a stable insoluble crystalline derivative is formed. This derivative is referred to as osazone. The crystals formed are yellow in colour and it can be identified by crystal shape.
Different sugars produces different osazone crystals. The time of formation melting point and shape of crystals are used for identification. Glucose forms needle shaped crystals. Maltose forms sunflower shaped crystals. Lactose forms cotton ball type crystals. This is used as simple visual method to identify the type of reducing sugar present in sample.
One limitation is also present. Since first two carbon atoms are modified during reaction sugars having same structure after C2 gives same osazone crystals. So glucose fructose and mannose forms same type of osazone crystals and it cannot be differentiated by this test.
Objectives of Osazone Test
Objectives of osazone test are as follows-
- Detection of reducing sugars– It is used to detect presence of carbohydrates having free carbonyl group (or potentially free carbonyl group).
- Differentiation of reducing and non reducing sugars– Reducing sugar forms osazone crystals but non reducing sugar like sucrose does not react in normal condition.
- Identification of specific reducing sugar– The crystal shape time of formation and melting point are observed and the specific sugar is identified.
- Differentiation of disaccharides– It is used to differentiate lactose and maltose (lactose gives cotton ball crystals and maltose gives sunflower crystals).
- Use in clinical and botanical sample analysis– It is used for analysis of sugars in biological fluids and it is also used to study distribution of sugars in plant tissues.
Principle of Osazone Test
Principle of osazone test is based on reaction of reducing sugar with phenylhydrazine and formation of osazone crystals. It occurs in acidic buffered medium and heating is required. The pH is maintained around 4.3 by sodium acetate and acetic acid. The mixture is heated in boiling water bath (100°C). Only reducing sugars reacts because free carbonyl group (aldehyde or ketone) is required for reaction.
The reaction is referred to as condensation–oxidation–condensation reaction. It is completed in three steps and three moles of phenylhydrazine is consumed for one mole of sugar. In first step phenylhydrazine condenses with carbonyl group of sugar and phenylhydrazone is formed.
In second step another molecule of phenylhydrazine acts as oxidizing agent. It oxidizes adjacent hydroxyl group (C2) and it is converted into carbonyl group. In third step a third phenylhydrazine molecule condenses with this new carbonyl group. A 1,2-bis-phenylhydrazone is formed and this is referred to as osazone. The yellow coloured crystals are produced and it is observed under microscope.
The main point is reaction involves only first two carbon atoms (C1 and C2) of sugar. So sugars which have same configuration after C2 gives same osazone crystals. Glucose fructose and mannose forms same osazone because structure from C3 onwards is same.
Requirements for Osazone Test
Requirements for osazone test are as follows-
Reagents and chemicals are-
- Test sample– Carbohydrate solution is taken (usually 1–2% in distilled water).
- Phenylhydrazine hydrochloride– It is main reagent which reacts with sugar to form osazone.
- Sodium acetate– It acts as buffer base and it liberates free phenylhydrazine from phenylhydrazine hydrochloride (osazone mixture can be prepared by mixing phenylhydrazine hydrochloride and sodium acetate).
- Glacial acetic acid– It is added to provide acidic medium (pH around 4.3) for reaction.
Materials and equipment are-
- Glassware– Clean dry test tubes and test tube stand is required.
- Handling tools– Pipettes are used for measurement and glass rod is used to transfer crystals on slide.
- Heating apparatus– Boiling water bath (100°C) is required for heating reaction mixture.
- Observation equipment– Microscope glass slide and cover slip is required to observe osazone crystals shape.
- Mixing equipment– Vortex mixer is used for proper mixing of reagent and sample.
Procedure of Osazone Test
Procedure of osazone test is as follows-
- 5 mL of carbohydrate test solution is taken in a clean dry test tube (1–2% solution is used).
- Dry osazone mixture is added (0.3–0.5 g) (phenylhydrazine hydrochloride and sodium acetate mixture). 3–5 drops of glacial acetic acid is added in same tube.
- The contents are mixed properly. If reagent is not dissolved at room temperature then it is warmed gently in water bath till clear solution is formed.
- The test tube is kept in boiling water bath (100°C) and time is noted.
- The tube is observed for yellow osazone crystal formation while heating. In monosaccharide like glucose and fructose crystals are formed within 5–10 minutes (solution is still hot).
- If crystals are not seen after 30–45 minutes then tube is removed and it is allowed to cool slowly at room temperature. Disaccharides like lactose and maltose forms crystal after cooling (it is soluble in hot water).
- A small amount of crystals is transferred on glass slide by glass rod. Cover slip is placed and crystals are observed under microscope (low to medium power) and shape is identified.
Result and Interpretation of Osazone Test
Result and interpretation of osazone test are as follows-
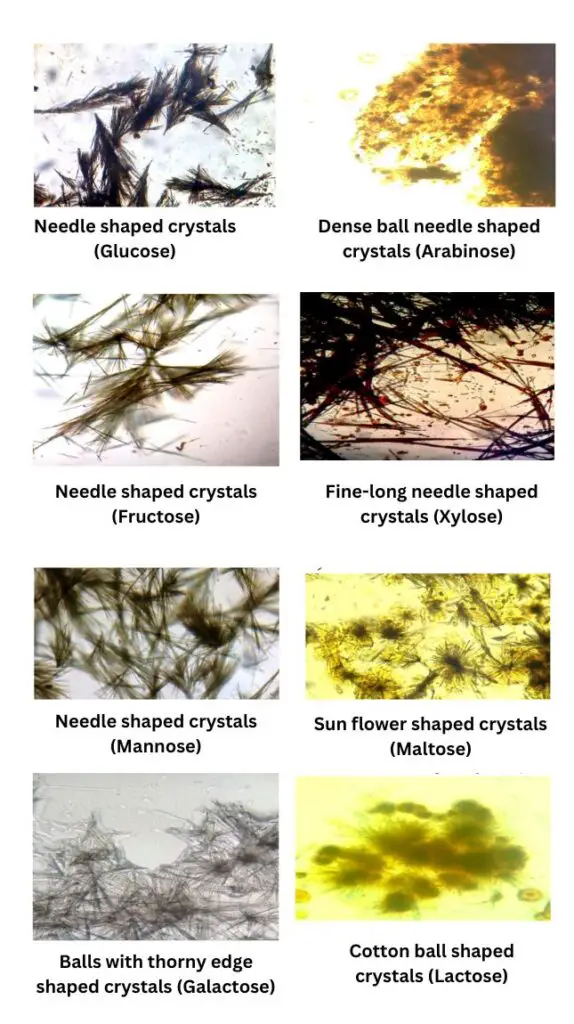
- Fructose– Yellow needle shaped crystals (sheaves type) is formed very fast. It is formed within 2–3 minutes (hot solution).
- Glucose– Yellow needle shaped crystals (broomstick or sheaves of wheat) is formed in about 4–5 minutes (hot solution).
- Xylose– Fine long intersecting needle shaped crystals is formed in about 6–7 minutes (hot solution).
- Mannose– Needle shaped crystals is formed (similar to glucose and fructose) and it takes around 10 minutes (hot solution).
- Arabinose– Dense ball of needles (dense cluster) is formed in about 10–11 minutes (hot solution).
- Galactose– Thorny ball shaped crystals (ball with thorny edges) is formed in about 15–20 minutes (hot solution).
- Maltose– Sunflower shaped (petal shaped) crystals is formed. It appears after cooling (because disaccharide osazone is soluble in hot water) and it is formed within 10–45 minutes on cooling.
- Lactose– Cotton ball shaped (powder puff) crystals is formed. It appears after cooling and it is formed within 20–45 minutes on cooling.
Uses of the Osazone Test
Uses of osazone test are as follows-
- Identification and differentiation of reducing sugars– It is used to detect reducing sugar and it helps to differentiate it from non reducing sugar.
- Differentiation of disaccharides– It is used to differentiate lactose and maltose by crystal shape (lactose gives cotton ball crystals and maltose gives sunflower crystals).
- Clinical diagnostic use– It is used to analyse biological fluids for specific sugars. It helps in screening of galactose (galactosemia) arabinose (autism) xylose (small bowel dysfunction) and glucose (renal glycosuria or impaired glucose tolerance).
- Food quality control– It is used to check authenticity of food products and to detect adulteration. Example honey adulteration by high fructose corn syrup rice syrup or cane sugar can be detected.
- Botany and agricultural research– It is used to study distribution of carbohydrates in plant tissues. It helps in fruit ripening study and starch accumulation mapping.
- Educational purpose– It is used in chemistry and biochemistry laboratory for teaching organic compound characterization and crystal formation.
Advantages of the Osazone Test
Advantages of osazone test are as follows-
- Cost effective and easy method– It is simple economical and quick test. Minimum equipment is required and reagents are easily available.
- Differentiation of reducing and non reducing sugars– Reducing sugar forms osazone crystals but non reducing sugar does not react in normal test condition.
- Visual identification– It gives clear yellow crystalline product and different sugars can be identified by crystal shape and time of formation.
- Differentiation of disaccharides– It is useful to differentiate lactose and maltose which is difficult by other simple test.
- Clinical diagnostic use– It is used as screening test for specific sugar in biological fluid and it helps in diagnosis of galactosemia small bowel dysfunction and intestinal candidiasis associated with autism.
- Use in plant tissue analysis– It is used in botany and agriculture for analysing carbohydrate content and distribution of sugars in plant sample.
- Detection of food adulteration– It is used in food industry to check authenticity of products. Adulteration of honey by commercial syrup can be detected.
- Modern adaptation possible– Microwave assisted heating can be used in modified method and it gives faster crystal formation (5–8 minutes) and it is energy efficient.
Limitations of the Osazone Test
Limitations of osazone test are as follows-
- False positive in non reducing sugar– Sucrose is non reducing sugar but if it is boiled for 30 minutes or more then hydrolysis can occur and osazone like crystals can be formed so false positive result is obtained.
- Not suitable for sugar mixture– If sample contains mixture of sugars then crystals are mixed and it can co-precipitate. Due to this interpretation becomes difficult and result is not reliable.
- Less sensitivity– High concentration of sugar is required (usually 1–2%) to get sufficient crystals for microscopic examination. Small amount of sugar may not give clear crystals.
- Epimer problem– Reaction involves first two carbon atoms so sugars having same configuration after C2 gives same osazone. Glucose fructose and mannose forms same crystals and it cannot be differentiated properly (only formation time can help but it is not always accurate).
References
- Aakash Educational Services Limited. (n.d.). Osazone formation – Definition, formation, tests of osazone, practice problems and FAQ in chemistry.
- Analytical monograph on the osazone reaction: Chemical principles, clinical diagnostics, and industrial methodologies. (n.d.).
- Laing, M. (2022, March 24). Different osazone crystals. Sciencing.
- Loma-Ang, S. A. (n.d.). Osazone test for sugar identification. Scribd.
- Osazone. (n.d.). In Wikipedia.
- Sapkota, A. (2022, September 5). Osazone test- definition, principle, procedure, result, uses. Microbe Notes.
- Shah, T. J. (2016). Utility of osazone test to identify sugars. Journal of Medical Science and Clinical Research, 4(12), 14361–14365. https://doi.org/10.18535/jmscr/v4i12.14.
- Sonkar, S. M., Sengupta, S., Sonkar, A., Bahri, S., & Sinha Roy, S. (2025). Osazone formation of sugars: A green chemistry strategy for the undergraduate laboratory. Asian Journal of Green Chemistry, 9(3), 246–254. https://doi.org/10.48309/AJGC.2025.488593.1592.
- Sugumaran, V. B., Silambanan, S., & Krithika. (2015). Osazones of the uncommonly encountered reducing sugars. International Journal of Interdisciplinary and Multidisciplinary Studies, 2(9), 24–29.
- Ziegler, F. E. (2016, April). Osazone formation.