One Step Growth Curve
The one-step growth curve is an experimental method used to study the life cycle of viruses inside a host cell. It was first developed by Emory Ellis and Max Delbrück in 1939. In this experiment, a large population of host cells is infected with viruses at the same time and the mixture is diluted so that newly released viruses cannot infect other cells. In this way only a single cycle of viral replication is observed. When the number of infectious virus particles is plotted against time, a characteristic growth curve is obtained. This curve generally shows three phases.
The first phase is the latent period. In this period the virus enters the host cell and loses its infectivity for a short time which is referred to as eclipse phase. During this phase viral nucleic acid and proteins are synthesized using the host cellular machinery and new virus particles are assembled inside the cell.
The second phase is the rise or burst period. In this step the infected host cells rupture and large numbers of newly formed viruses are released at the same time. Because of this sudden release the number of extracellular viruses increases rapidly.
The final phase is the plateau period. In this stage the number of virus particles remains constant because all infected cells have already lysed and no further infection takes place due to the dilution of the culture. The one-step growth curve helps in determining important properties of viruses such as the length of the latent period and the burst size which represents the number of virus particles produced from a single infected cell.
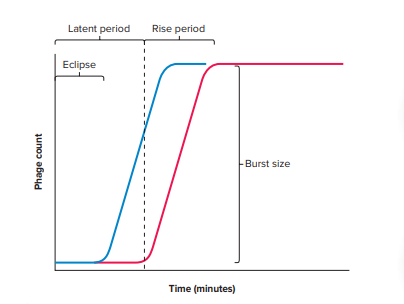
Phases of One Step Growth Curve
The phases of one-step growth curve represent the different stages of viral replication inside the host cell. These phases show how virus enters the cell, multiplies and releases new virus particles.
- Inoculation / Adsorption Phase – It is the initial stage of infection. In this step the virus particles attach to specific receptors present on the surface of host cell. After attachment the virus penetrates into the host cell and infection begins.
- Latent Period –It is the time interval between viral attachment and the release of new virus particles from the host cell. During this period the virus replicates inside the host cell but no extracellular viruses are detected. This period includes eclipse phase and intracellular accumulation phase.
- Eclipse Phase – In this step the virus uncoats after entering the host cell and releases its nucleic acid. The viral genome controls the host cellular machinery and synthesis of viral nucleic acid and proteins occurs. During this stage no complete infectious virions are present.
- Intracellular Accumulation Phase– In this phase the newly synthesized viral nucleic acids and structural proteins assemble to form mature virus particles. These virus particles temporarily accumulate inside the cytoplasm of host cell.
- Rise or Burst Period (Lysis Phase)– It is the stage where infected host cells rupture and release large number of newly formed viruses. Because of this sudden release the number of extracellular virus particles increases rapidly.
- Plateau or Stationary Period– It is the final stage of growth curve. In this phase the number of virus particles becomes constant because all infected host cells have already lysed and released their viruses. No further increase in virus number occurs.
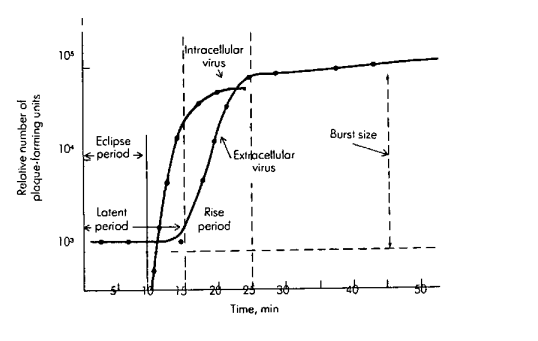
1. Inoculation / Adsorption Phase
- It is the initial stage of viral infection where virus particles (virions) are brought into contact with susceptible host cells.
- In this step the virion attaches to the host cell surface through specific receptor molecules present on the cell membrane.
- The viral surface proteins (antireceptors) bind with complementary receptors of host cell. This specific interaction is referred to as adsorption.
- The initial attachment between virus and host receptor is often reversible in nature.
- After successful attachment the virus penetrates the host cell.
- In bacteriophages the viral genome is injected through the bacterial cell wall into the host cytoplasm.
- In animal viruses the entry occurs either by membrane fusion or by endocytosis (engulfment of virus by host cell membrane).
- In one-step growth experiments this phase is synchronized so that many viruses attach to host cells at the same time (time zero).
- The multiplicity of infection (MOI) is controlled during this phase. MOI is the ratio of infectious virus particles to host cells.
- A high MOI ensures that almost every host cell is infected by virus particles.
- A low MOI ensures that each host cell is infected by only a single virus particle.
- The adsorption period is usually kept very short and normally lasts only few minutes.
- After adsorption the culture is diluted heavily to stop further infection by free virus particles.
- In some experiments the unadsorbed viruses are removed by centrifugation or neutralized by anti-phage serum or virucides.
2. Latent Period Phase
- It is the minimum time interval between adsorption of virus to the host cell and the first release of new viral progeny outside the cell.
- During this phase no infective virus particles are detected in the extracellular culture medium.
- The extracellular plaque count remains constant because newly formed virions are not yet released from the host cell.
- This phase includes the entire intracellular replicative cycle of the virus.
- After penetration the viral capsid is removed and the viral nucleic acid is released inside the host cell. This step is referred to as uncoating.
- The viral genome is then replicated inside the host cell with the help of host cellular machinery.
- Transcription of viral genes takes place and viral messenger RNA (mRNA) is produced.
- These viral mRNA molecules are translated by host ribosomes to form viral structural and enzymatic proteins.
- Newly synthesized viral nucleic acids and proteins are assembled to form new viral particles.
- The latent period is divided into two parts– Eclipse phase and Intracellular accumulation phase.
- Eclipse phase is the early stage after penetration where virus loses its infectivity due to uncoating.
- During eclipse phase no complete virions are detected either inside or outside the host cell.
- Viral genome replication and synthesis of viral proteins occurs during this period.
- Intracellular accumulation phase is the later stage where newly formed viral components assemble to form mature virions.
- These newly formed virions accumulate in large number within the host cell cytoplasm before release.
- The duration of latent period varies depending on virus and host type.
- In bacteriophages the latent period usually lasts about 20–60 minutes.
- In animal and plant viruses this period may extend from several hours to few days.
- The length of latent period is related to the burst size (number of virions released from a single infected cell).
a. Eclipse Phase
- It is the first stage of the latent period that begins immediately after penetration of the virus into the host cell.
- In this phase the viral capsid is removed and the viral nucleic acid is released inside the host cell. This step is referred to as uncoating.
- The eclipse phase ends when the first fully mature progeny virion is assembled inside the host cell.
- During this period the virus temporarily loses its infectivity. This condition is referred to as eclipse.
- No intact or complete virus particles are present either inside or outside the host cell during this stage.
- If the host cell is artificially lysed during this phase no functional virions will be detected.
- Although intact virions are absent the virus remains metabolically active within the host cell.
- The viral genome is replicated using the host cellular machinery.
- Viral messenger RNA (mRNA) is synthesized through transcription of viral genes.
- These viral mRNA molecules are translated by host ribosomes to produce viral proteins and capsid components.
- Newly synthesized viral nucleic acids and proteins accumulate in the host cytoplasm.
- The duration of eclipse phase varies depending on the type of virus.
- In bacteriophages the eclipse phase generally lasts about 10–15 minutes.
- In animal viruses the eclipse phase may extend for several hours or even days.
- The eclipse phase cannot be directly observed in a normal one-step growth experiment because extracellular viruses are measured.
- To study this phase the host cells are artificially lysed by methods such as chloroform treatment or sonic disruption in order to detect newly formed intracellular virions.
b. Intracellular Accumulation Phase
- It is the second stage of the latent period which occurs immediately after the eclipse phase.
- By the beginning of this phase viral nucleic acids and viral structural proteins are already synthesized inside the host cell.
- These viral components accumulate in large amount within the cytoplasm of the host cell.
- During this phase the viral genomes combine with capsid proteins to form complete viral particles.
- This process is referred to as assembly of virions.
- The viral components spontaneously organize and assemble into mature virus particles.
- As a result many complete progeny virions are formed inside the host cell.
- The newly formed virions accumulate in large numbers within the cytoplasm of the infected cell.
- During this period the virus particles remain inside the host cell and are not yet released outside.
- The number of mature virions inside the host cell gradually increases with time.
- These accumulated virions remain stored inside the host cell until the burst or lysis phase occurs.
3. Rise or Burst Period (Lysis Phase)
- It is the phase that occurs immediately after the latent period in the viral growth cycle.
- During this period a rapid increase in the number of free extracellular virus particles is observed.
- This sudden increase occurs due to the rupture of infected host cells. This process is referred to as lysis.
- During lysis the host cell membrane breaks open and releases all newly formed mature virions.
- As a result a large number of viral particles are released simultaneously into the surrounding medium.
- In enveloped viruses the release of virions occurs through budding instead of cell lysis.
- During budding the virus acquires a portion of the host cell membrane as its envelope.
- On a one-step growth curve this phase appears as a steep upward slope in viral count.
- The rise in viral titer indicates the release of progeny viruses from infected cells.
- The steepness of the rise period depends on the synchronization of infection among host cells.
- If all infected cells lyse at the same time the increase in viral particles appears almost instantaneous.
- However in most cultures the lysis of cells occurs at slightly different times producing a sloped rise curve.
- The timing of cell lysis is influenced by the physiological condition and metabolic activity of host cells.
- Certain viral proteins such as holins help in controlling the lysis of infected cells.
- The duration of rise period varies depending on the type of virus.
- In virulent bacteriophages such as T4 this phase may last only about 10 minutes.
- In eukaryotic viruses that release virions by budding the rise period may be more gradual and extended.
4. Plateau or Stationary Period
- It is the final stage of the one-step growth curve of virus.
- This phase occurs immediately after the rise or burst period.
- During this period all infected host cells have already undergone lysis.
- As a result all newly formed viral progeny are released into the surrounding medium.
- The number of extracellular virus particles becomes constant during this phase.
- The viral titer or plaque count shows no further increase.
- This condition is referred to as the plateau or stationary phase.
- The stabilization of viral count occurs because no more infected cells remain to release new virions.
- In experimental conditions the culture is usually diluted greatly which prevents further infection of new host cells.
- Sometimes the susceptible host cells are already exhausted in the culture.
- Because of this newly released viruses cannot infect additional host cells.
- The viral population therefore remains constant for a certain period.
- The final viral titer measured in this phase is used for calculating burst size.
- Burst size represents the average number of virions produced by a single infected host cell.
What is Burst Size?
Burst size is defined as the average number of new infectious virus particles produced from a single infected host cell during viral multiplication. It is the measure of reproductive capacity of a virus. During infection the virus enters into the host cell and its nucleic acid begins to control the host cellular machinery. In this step viral nucleic acid and structural proteins are synthesized and many new virus particles are formed inside the host cell. At the end of the replication cycle the host cell undergoes lysis and the newly formed viruses are released. The number of virus particles released from one infected cell is referred to as burst size.
In a one–step growth curve experiment burst size is calculated by dividing the total number of virus particles released during plateau phase by the number of initially infected host cells. The burst size is not constant and it may vary depending on the virus strain host cell condition and environmental factors such as temperature and nutrient availability. In many viruses the burst size may range from a few tens to several thousand virus particles produced from a single infected cell.
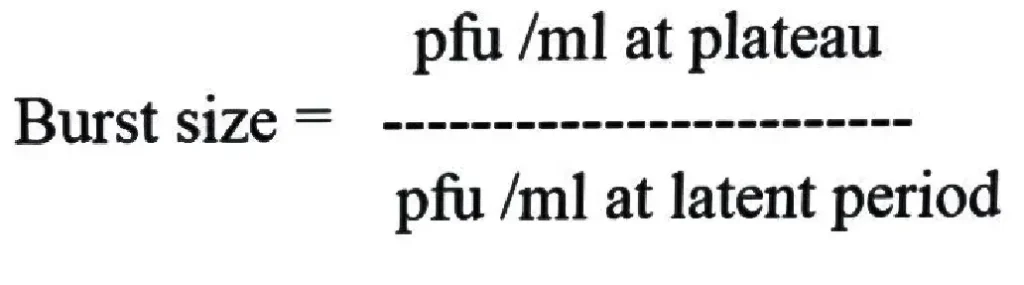
Significance of One-Step Growth Curve of Virus
The one-step growth curve experiment is important in virology because it helps in understanding the replication cycle and multiplication of viruses inside the host cell. Some of the main significance are–
- Understanding viral replication– It helps in explaining that viruses multiply inside the host cell by assembly of viral components rather than by binary fission like cellular organisms.
- Measurement of latent period – It is used to determine the length of latent period which represents the time between viral infection and release of new virus particles.
- Determination of burst size– It helps in calculating the burst size which represents the number of virus particles produced from a single infected host cell.
- Study of intracellular events– It provides information about different biochemical and molecular events that occur during the viral replication cycle inside the host cell.
- Proof of single particle infectivity– It demonstrates that a single virus particle is capable of infecting a host cell and initiating viral infection.
- Study of environmental effects– It helps in studying the effect of environmental factors such as temperature, nutrients and host cell condition on viral growth.
- Understanding viral growth and fitness – It provides information about viral multiplication efficiency and helps in understanding the interaction between virus and host cell population.
- Abedon, S. T. (2025). Dos and don’ts of bacteriophage one-step growth. Preprints.org. https://doi.org/10.20944/preprints202507.2624.v1
- Abedon, S. T., Hyman, P., & Thomas, C. (2003). Experimental examination of bacteriophage latent-period evolution as a response to bacterial availability. Applied and Environmental Microbiology, 69(12), 7499–7506. https://doi.org/10.1128/AEM.69.12.7499-7506.2003
- Ali, U. (n.d.). Enveloped vs. non-enveloped viruses: A visual MCAT breakdown. King of the Curve.
Azab, B. (2018). Lecture 3 topics [Lecture notes]. Doctor 2018. - Bacteriophage Ecology Group. (n.d.). Eclipse. Archaeal Viruses.
- Bacteriophage Ecology Group. (n.d.). Single-step growth. Archaeal Viruses.
- Barrick Lab. (n.d.). ProtocolsPhageBurstSize. TWiki.
- Cann, A. J. (2016). Replication. In Principles of Molecular Virology (pp. 105–133). Elsevier. https://doi.org/10.1016/B978-0-12-801946-7.00004-3
- Chandra, N. S. (n.d.). One step multiplication curve [Lecture notes]. Ram Lal Anand College, Department of Microbiology.
- Creative Biolabs. (n.d.). One-step growth curve of phage. BioPhage Technology.
- Difference between enveloped and non enveloped virus. (2012). r/biology – Reddit.
- Dominguez-Mirazo, M., Harris, J. D., Demory, D., & Weitz, J. S. (2024). Accounting for cellular-level variation in lysis: Implications for virus–host dynamics. mBio, 15(8), e01376-24. https://doi.org/10.1128/mbio.01376-24
- Fenner, F. (1999). Genetics of animal viruses. In Encyclopedia of Virology (pp. 606–613). Elsevier. https://doi.org/10.1006/rwvi.1999.0111
- Homework.Study.com. (n.d.). What is the difference between the eclipse phase and the latent phase of the virus replication cycle?
- Kakizoe, Y., Nakaoka, S., Beauchemin, C. A. A., Morita, S., Mori, H., Igarashi, T., Aihara, K., Miura, T., & Iwami, S. (2015). A method to determine the duration of the eclipse phase for in vitro infection with a highly pathogenic SHIV strain. Scientific Reports, 5, 10371. https://doi.org/10.1038/srep10371
- Kalatzis, P. G., Bastías, R., Kokkari, C., & Katharios, P. (2016). Isolation and characterization of two lytic bacteriophages, φSt2 and φGrn1; Phage therapy application for biological control of Vibrio alginolyticus in aquaculture live feeds. PLoS ONE, 11(3), e0151101.
- Kannoly, S., Oken, G., Shadan, J., Musheyev, D., Singh, K., Singh, A., & Dennehy, J. J. (2022). Single-cell approach reveals intercellular heterogeneity in phage-producing capacities. Microbiology Spectrum, 11(1), e02663-21. https://doi.org/10.1128/spectrum.02663-21
- Kuo, J., & Li, R. (2018). Growth curve and life cycle of viruses. Wolfram Demonstrations Project.
Liu, Y., Chang, S., Murphy, G., Ajayi-Akinsulire, E., Ardren, I., Guy, I., Johnston, K., Lee, S., & Russell, L. (2025). 7.4: Animal virus life cycle. Biology LibreTexts. - Lumen Learning. (n.d.). The viral life cycle. Microbiology.
- Middelboe, M., Chan, A. M., & Bertelsen, S. K. (2010). Isolation and life-cycle characterization of lytic viruses infecting heterotrophic bacteria and cyanobacteria.
- Middelboe, M., Chan, A. M., & Bertelsen, S. K. (2016). One-step growth experiments (bacteriophages). Protocols.io. https://doi.org/10.17504/protocols.io.dpw5pd
- Oka, Y., Miyakawa, K., Yamazaki, M., & Maruyama, Y. (2026). Feature-based growth curve classification enables efficient phage discrimination. Viruses, 18(1), 92. https://doi.org/10.3390/v18010092
- Open Educational Resources. (2019). 5.2 The viral life cycle. In Allied Health Microbiology.
Rybicki, E. P., & Kightley, R. (2015, January 29). A short history of the discovery of viruses – Part 3. ViroBlogy. - Sanjuán, R., & Domingo-Calap, P. (2016). Mechanisms of viral mutation. Cellular and Molecular Life Sciences, 73(23), 4433–4448. https://doi.org/10.1007/s00018-016-2299-6
- Shet, S. A. (n.d.). Programme: Bachelor of Science (Third Year) Subject: Microbiology Course Code: MID 105 Course Title: Virology Unit II: Bacteriophages [Lecture notes].
- Shin, H., Lee, J.-H., Yoon, H., et al. (2013). Genomic investigation of lysogen formation and host lysis systems of the Salmonella temperate bacteriophage SPN9CC. ResearchGate.
- The “one step growth curve”. (n.d.).
- Viral evolution. (2025, December 17). In Wikipedia.
- Viral latency. (n.d.). ViralZone – Expasy.
- Virology Research Services. (2022). Enveloped vs. non-enveloped viruses.
- Virus latency. (2026, February 27). In Wikipedia.
- Zath, G. K., Thomas, M. M., Loveday, E. K., Bikos, D. A., Sanche, S., Ke, R., Brooke, C. B., & Chang, C. B. (2024). Influenza A viral burst size from thousands of infected single cells using droplet quantitative PCR (dqPCR). PLoS Pathogens, 20(7), e1012257. https://doi.org/10.1371/journal.ppat.1012257