What is Nylander’s Test?
Nylander’s Test is a chemical test. It is used for qualitative detection of reducing sugar mainly glucose in biological fluids like urine. It was developed in 1880s by Emil Nylander. It was used earlier as screening test for diabetes mellitus and other metabolic disorder.
It is based on oxidation reduction reaction. In this test carbohydrate act as reducing agent and bismuth(III) ions act as oxidizing agent. Nylander reagent is an alkaline solution. It contain bismuth subnitrate potassium hydroxide and Rochelle salt (potassium sodium tartrate) which act as chelating agent.
In strongly alkaline medium (due to potassium hydroxide) the reducing sugar become reactive. Bismuth(III) ions is reduced to elemental metallic bismuth. The mixture is heated in boiling water bath for 2 to 5 minutes.
Positive test is indicated by dark brown colour and then heavy black precipitate is formed. This black precipitate is metallic bismuth. Negative test is indicated when solution remain clear or pale and precipitate is not formed.
It was favoured over Fehling test in earlier time. It show more selectivity for monosaccharides and false positive due to concentrated non sugar substances like uric acid and creatinine are reduced. The detection limit is around 0.1% to 0.5% glucose so it mostly indicate pathological level of sugar.
Limitation is present. Sulfur containing protein like albumin can give false positive. It react and bismuth sulfide is formed which give similar black precipitate.
Objectives of Nylander’s Test
Objectives of Nylander’s Test are as follows–
- It is used for detection of reducing sugar mainly glucose in biological fluids like urine.
- It is used for screening of metabolic disorder. Pathologic glucosuria is detected which indicate diabetes mellitus.
- It is used for differentiating cause of glucosuria. Diabetes renal glucosuria pregnancy and Fanconi syndrome can be differentiated.
- It is used as broad screening for reducing substances in special clinical cases. It is used in pediatric screening for galactosemia.
- It is used in histological diagnosis. Oxidation of reducing sugars (mono saccharides di saccharides and polysaccharides) is detected under microscope.
- It is used as teaching tool. Redox chemistry and metal tartrate coordination complex behaviour is studied.
- It is used in low resource condition. It is used when modern enzymatic reagent is not available or it degrade easily.
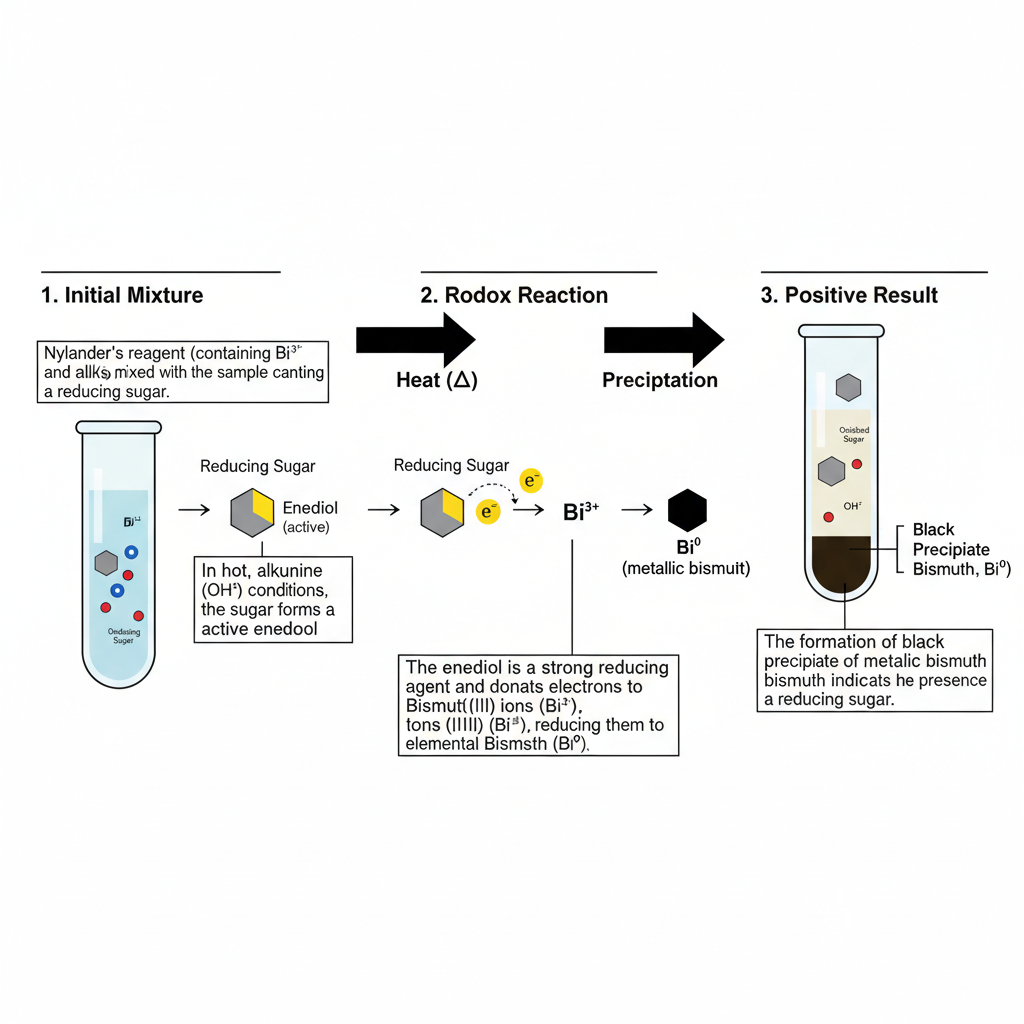
Principle of Nylander’s Test
Principle of Nylander’s Test is based on oxidation reduction reaction. It is used for detection of reducing sugar. In this reaction carbohydrate act as reducing agent and bismuth(III) ions act as oxidizing agent.
The test is carried out in strongly alkaline medium. Under this condition reducing sugar such as glucose or fructose undergo enolization and reactive intermediate is formed. These activated sugar reduce the soluble bismuth(III) ions present in Nylander reagent to elemental metallic bismuth.
Rochelle salt (potassium sodium tartrate) is present in reagent as chelating agent. It is used for keeping bismuth in dissolved form in alkaline solution. Premature precipitation of bismuth hydroxide is prevented.
During reaction sugar is oxidized and bismuth is reduced. The elemental bismuth is formed and it settle as dense black or dark brown precipitate. This formation of black precipitate is the indication of positive test for reducing sugar.
Requirements for Nylander’s Test
Requirements for Nylander’s Test are as follows–
- Nylander’s reagent is required. It is alkaline solution containing bismuth subnitrate (about 2.0% w/v) potassium sodium tartrate (Rochelle salt about 4.0% w/v) and strong alkali such as 10.0% potassium hydroxide or sodium hydroxide.
- Test sample is required (urine or carbohydrate solution).
- Sample pre treatment is required for urine. Protein should be removed before test. Acetic acid is added then boiling is done and filtration is carried out. This is done to avoid false positive due to sulfur containing proteins which form bismuth sulfide.
- Fixed reagent to sample ratio is required. Usually 1 part Nylander reagent and 10 part sample is taken (example 0.5 ml to 1.0 ml reagent with 5.0 ml carbohydrate solution or 1 ml reagent with 10 ml urine).
- Strong alkaline medium is required. It is needed for enolization of sugar so reducing action is produced.
- Boiling water bath is required for controlled heating. Direct flame heating should be avoided to prevent bumping and uneven heating.
- Strict reaction timing is required. Heating is done for exactly 2 to 5 minutes. Positive result (dense black or dark brown precipitate of metallic bismuth) should appear during heating. Colour change formed only after cooling is not taken as specific.
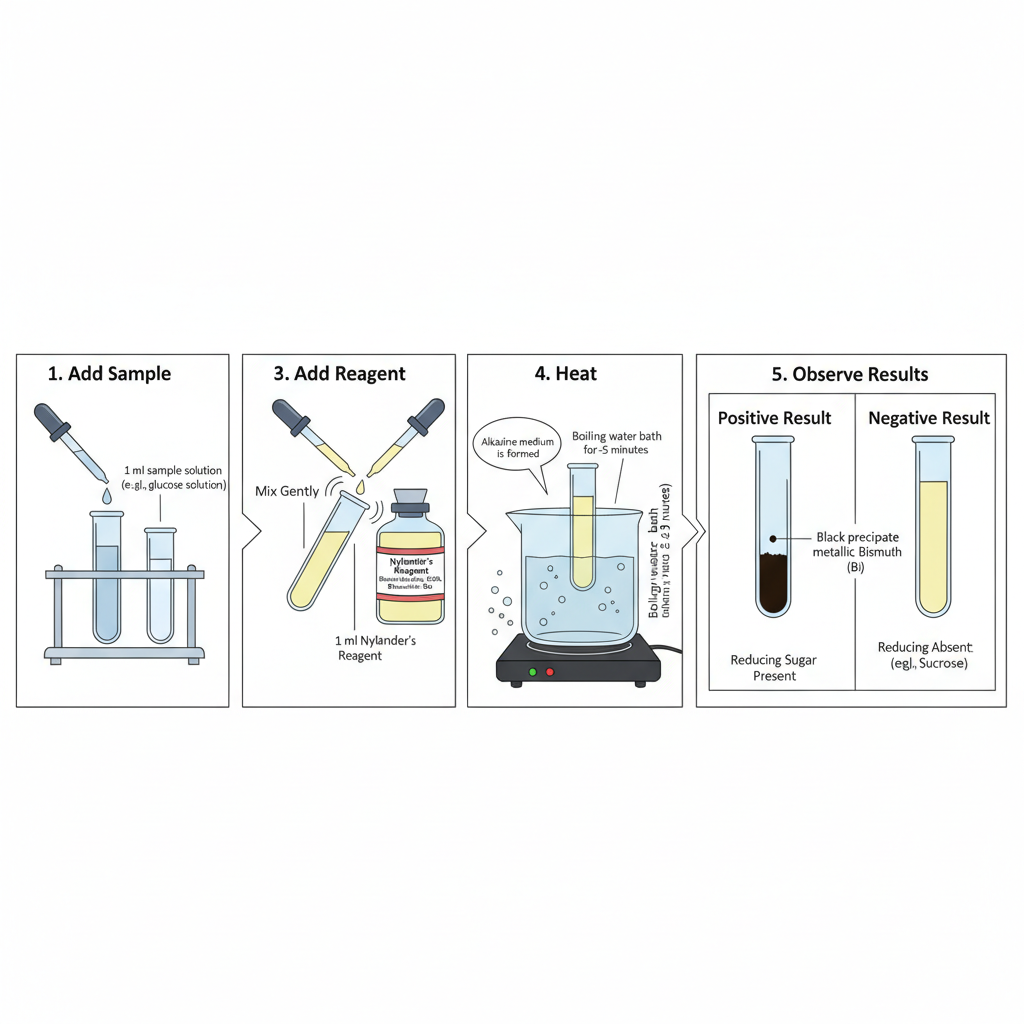
Step-by-Step Procedure of Nylander’s Test
Step-by-Step Procedure of Nylander’s Test are as follows–
- Step 1- If sample contain sulfur containing proteins then protein is removed first. The sample is acidified with acetic acid then it is boiled and filtered.
- Step 2- Take 5.0 ml of test solution (urine or 5% carbohydrate solution) in a heat resistant glass test tube.
- Step 3- Add 0.5 ml to 1.0 ml of Nylander’s reagent. Standard ratio is maintained as 1 part reagent and 10 part urine.
- Step 4- Mix the contents by gentle shaking so reagent is distributed uniformly.
- Step 5- Keep the test tube in boiling water bath for exactly 2 to 5 minutes. Direct heating over open flame should not be done because uneven heating and sample loss can occur and result may be inaccurate.
- Step 6- The colour change is observed during boiling. Positive test is indicated when solution turns dark brown and then black precipitate is formed and settle down. Dark colour which appear only after cooling is not taken as positive result and it is non specific.
Nylander’s Test Result
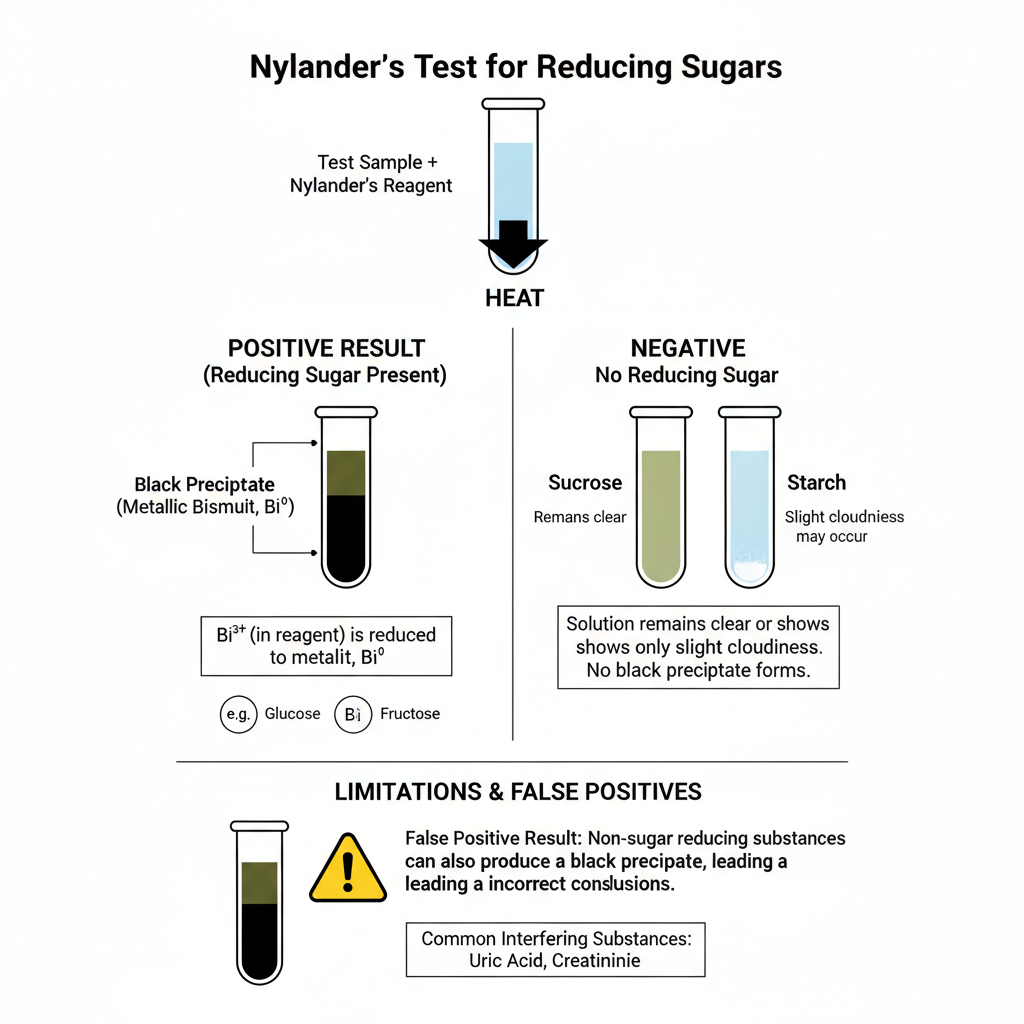
Nylander’s Test Result are as follows–
Positive result
- Solution turns dark brown and then black during heating.
- Heavy dense black or dark brown precipitate is formed. This precipitate is elemental bismuth.
- Glucose– Strong positive is seen and dense black precipitate is formed rapidly.
- Fructose– Olive green to dark brown or black colour is formed.
- Galactose– Olive green to dark brown or black colour is formed.
Negative result
- Solution remain clear or light yellow or pale green.
- Blackening is not seen and black precipitate is not formed. Cloudy white precipitate may be formed.
- Lactose– Negative result is obtained.
- Sucrose– Negative result is obtained.
- Starch– Negative result is obtained.
- Glycogen– Negative result is obtained.
- Maltose– Slow reduction can occur. Dark yellow or brownish colour may be formed so it is taken as weak positive or negative.
Invalid reaction
- Dark colour formed only after cooling is not taken as positive. It is non specific reaction.
False positive
- Sulfur containing proteins like albumin can give false positive. Bismuth sulfide is formed and black precipitate look similar to true positive.

Uses of Nylander’s Test
Uses of Nylander’s Test are as follows–
- It is used for diagnosis of metabolic disorder. Significant glycosuria (glucose in urine) is detected which indicate diabetes mellitus.
- It is used for differentiating causes of glucosuria. Diabetes mellitus renal glucosuria pregnancy and Fanconi syndrome can be differentiated.
- It is used for testing concentrated urine. False positive due to non sugar reducing substances like uric acid and creatinine are less so it is useful for concentrated urine sample.
- It is used for testing lactating patients. It is less affected by lactose so glucose in urine of lactating women can be detected.
- It is used for broad pediatric screening for reducing substances. It is used as backup method in cases like galactosemia screening.
- It is used as teaching model. Oxidation reduction (redox) reaction one electron transfer and metal tartrate coordination complex behaviour is studied.
- It is used in low resource condition. It is used when modern enzymatic strips are not available or storage condition is poor and strips degrade.
Advantages of Nylander’s Test
Advantages of Nylander’s Test are as follows–
- Nylander reagent is stable. It remain effective for many months when it is stored in amber coloured bottle and light induced reduction is prevented.
- It give clear end point. Dense black precipitate is formed so result interpretation is easy as compared to copper based test where colour change may be confusing.
- Pass through phenomenon is avoided. Black bismuth precipitate is cumulative and it remain stable after reduction so visual error and underestimation of high glucose seen in some copper test is reduced.
- False positive due to non sugar reducing substances is less. Uric acid and creatinine do not easily disturb the reagent so concentrated urine sample can be tested with less false positive.
- It show more selectivity for monosaccharides. Glucose is detected readily but lactose is less affecting the test so it is useful for lactating women or digestive problem cases.
- Detection limit is clinically useful. It detect around 0.1% to 0.5% glucose which match renal threshold so significant pathological glycosuria (diabetes mellitus) is detected and trace irrelevant positives are reduced.
Limitations of Nylander’s Test
Limitations of Nylander’s Test are as follows–
- Protein interference is major limitation. Sulfur containing proteins like albumin can give false positive. In alkaline medium and heating condition sulfur react and bismuth sulfide is formed. It give black precipitate which look same as sugar positive. So sample pre treatment is required (acidification boiling and filtration).
- Sensitivity is low to moderate. Detection limit is around 0.1% to 0.5% (100–500 mg/dL). Trace amount of glucose below 0.1% is not detected properly.
- Drug and chemical interference can occur. Ascorbic acid (Vitamin C) cephalosporin antibiotics chloral hydrate turpentine and pigments from senna or rhubarb can give false positive. Oxidizing agents can prevent reduction and false negative can be obtained.
- Strict timing is required. True positive should be seen during active heating. If dark colour appear only after cooling then it is invalid and non specific.
- Certain sugars are detected poorly. It is selective for monosaccharides like glucose but reducing disaccharides such as lactose and maltose give poor reaction.
Precautions of Nylander’s Test
Precautions of Nylander’s Test are as follows–
- Protein should be removed before test. Sample is acidified with acetic acid then it is boiled and filtered. Sulfur containing protein can form black bismuth sulfide precipitate and false positive is obtained.
- Boiling water bath is used for heating. Direct heating over open flame should not be done. Uneven heating and bumping can occur and sample loss or inaccurate result is obtained.
- Ratio and timing should be followed strictly. Reagent to sample ratio is maintained (1 part reagent and 10 part urine). Heating is done for exactly 2 to 5 minutes. False result can occur if it is not followed.
- Result should be observed during heating. Positive result is taken only when dark colour and black precipitate appear while solution is heated. Colour change during cooling is non specific and it is invalid.
- Personal protective equipment is used. Nylander reagent is corrosive because strong alkali and bismuth salts are present. Nitrile gloves goggles and lab coat is worn. If reagent contact skin or eye then washing with water for minimum 15 minutes is done and medical help is taken.
- Reagent storage should be proper. Amber coloured bottle is used to protect from light induced reduction. Compatible container (HDPE or alkali resistant glass) should be used because reagent can damage vessel.
- Waste disposal should be done properly. Test waste should be neutralized before disposal because reagent is highly alkaline and local regulation should be followed.
References
- Analytical biochemistry of bismuth-based reduction: A comprehensive evaluation of Nylander’s test for carbohydrates. (n.d.).
- Meeker, G. H. (n.d.). Comparison of the sensitivieness of the Fehling, the Nylander and the phenyl-hydrazine tests for the detection of dextrose in urine. The Journal of the American Pharmaceutical Association, 462–465.
- Sánchez-Viesca, F., & Gómez, R. (2022). The mechanism of Nylander’s test for glucose in urine. American Journal of Chemistry, 12(5), 91–93. https://doi.org/10.5923/j.chemistry.20221205.02
- Soriano, A. G. (n.d.). Carbohydrate qualitative tests overview. Scribd.
- Wikipedia contributors. (2024, April 13). Nylander’s test. In Wikipedia, The Free Encyclopedia. https://en.wikipedia.org/w/index.php?title=Nylander%27s_test&oldid=1218745223
- 200085-student. (n.d.). Nylanders test. Scribd.
- BYJU’S. (n.d.). Tests of carbohydrates. Retrieved from [Source URL not provided in excerpt]
- Chemistry Stack Exchange. (2025). Functional difference of Benedict’s solution and Fehling’s solution.
- Dela Cruz, G. (2023). Post-lab activity No. 12 test for carbohydrates [PDF document]. Scribd.
- Filo EdTech INC. (2025). nylander’s test for carbohydrates structures. Filo.
- Food Safety and Standards Authority of India. (2015). Manual of methods of analysis of foods milk and milk products.
- Food Safety and Standards Authority of India. (2016). Manual of methods of analysis of foods: Milk and milk products.
- Hernández-López, A., Sánchez Félix, D. A., Z. Z.-S., I. G.-B., T. D. D., & A. X. A.-A. (2020). Quantification of reducing sugars based on the qualitative technique of Benedict. ACS Omega, 5(50), 32403–32410. doi: 10.1021/acsomega.0c04467
- Lumongsud, M. A. C. (n.d.). Biochem lab tests explained | PDF | materials | atoms [PDF document]. Scribd. Retrieved from
- Morphisto. (n.d.). NYLANDER’s Reagenz. Retrieved from
- Nylander’s test. (2023). In Wikipedia. Retrieved from
- Nylander’s test. (2024). In Wikipedia. Retrieved from
- Nylanders test [PDF document]. (n.d.). Scribd. Retrieved from
- ResearchGate. (2022). (PDF) The mechanism of Nylander’s test for glucose in urine. Retrieved from [Source URL not provided in excerpt]
- The Nylander’s assay: Comprehensive review of the bismuth reduction principle for reducing sugar detection, mechanism, and analytical performance. (n.d.). Retrieved from [Source URL not provided in excerpt]
- preparation of special analytical reagents. (n.d.). In CRC handbook of chemistry and physics (91st ed.). CRC Press.
- Glucose. (2024). In Wikipedia. Retrieved from [Source URL not provided in excerpt]
- Glucose in urine test: MedlinePlus medical test. (2024). U.S. National Library of Medicine. Retrieved from
- Siemens Healthcare Diagnostics, Inc. (2016). 510(k) substantial equivalence determination decision memorandum assay only template (K161494). U.S. Food and DrugAdministration.Retrieved from
- Soriano, A. G. (n.d.). Post lab finals3 | PDF | glucose | starch [Presentation]. Scribd. Retrieved from
- Testbook. (n.d.). Fehling test – reaction, reagent, mechanism, and difference between Fehling’s and Benedict’s test. Retrieved from
- Thomas, L. (n.d.). Chapter 53: Laboratory organization. In Clinical laboratory diagnostics.: TH-Books.