What is Micropropagation?
Micropropagation is a technique used for rapid multiplication of plants by using very small pieces of plant tissue. It is also referred to as plant tissue culture. In this method a small part of plant such as cells, leaves or shoot tips are taken and used as explant. The explant is grown in an artificial nutrient medium under sterile laboratory conditions.
This process occurs due to the property of plant cells called cellular totipotency. It is the ability of a single plant cell to divide and develop into a complete plant. When the explant is placed on nutrient medium under controlled conditions of temperature light and humidity the cells divide repeatedly and many small plantlets is produced.
Micropropagation helps in producing large number of plants within short period of time. The plants produced by this method are genetically identical to the parent plant and mostly disease free. Because of these advantages this technique is widely used in agriculture horticulture forestry and in conservation of rare plant species.
Micropropagation Definition
Micropropagation is a method of rapidly multiplying plants by growing plant tissues, typically from the apical meristem, in sterile, controlled conditions to produce genetically identical clones. It is used for large-scale plant production, crop improvement, and maintaining genetic uniformity.
Stages of Micropropagation / Steps of Micropropagation
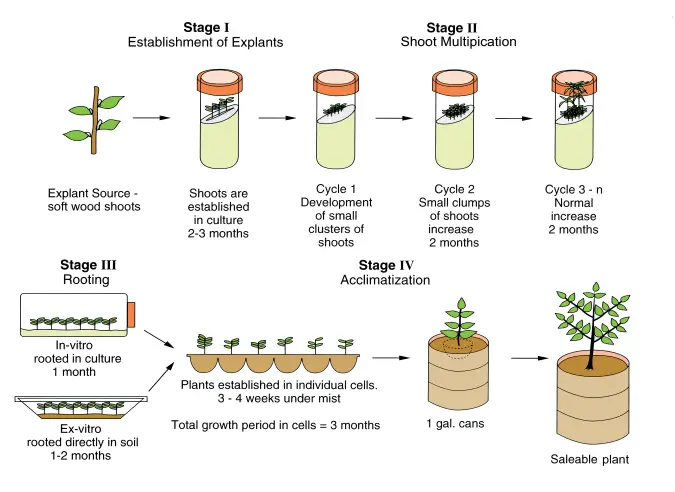
Micropropagation is carried out through several stages. In each stage specific development of plant tissues is carried out under controlled laboratory conditions. The stages were described by Murashige (1974). The stages are as follows–
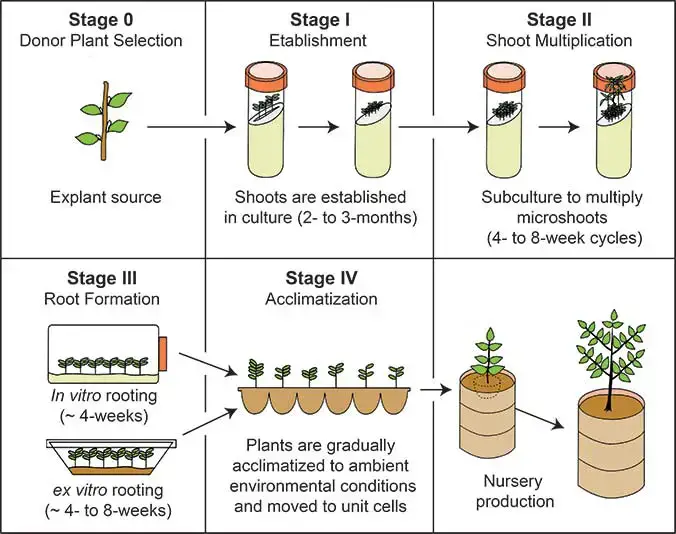
- Stage 0 – Selection of Stock Plant– In this stage a healthy and disease free stock plant is selected. Small plant parts called explants are obtained from this plant. The quality of stock plant is very important because it influences the success of culture.
- Stage I – Initiation of Culture– In this step small pieces of plant tissue such as apical meristem (about 5–10 mm) are removed from the stock plant. The explants are sterilized using disinfectants such as alcohol (70%), sodium hypochlorite or calcium hypochlorite for few minutes. After sterilization the explants are washed several times with distilled water and transferred to sterile nutrient medium to initiate culture.
- Stage II – Shoot Proliferation– In this stage the explants are inoculated into culture medium such as Murashige and Skoog (MS) medium. The culture is incubated at suitable temperature around 20–25°C. Shoots begin to develop from the explants. These shoots are transferred to another medium to induce formation of multiple shoots and large number of shoots is produced.
- Stage III – Rooting of Shoots– The developed shoots are separated and transferred to rooting medium. In this medium root formation occurs. By the end of this stage complete plantlets having both shoots and roots are formed inside culture tubes.
- Stage IV – Acclimatization– In this stage the plantlets are gradually transferred from laboratory conditions to external environment. The plantlets are acclimatized slowly to soil conditions by exposing them to lower humidity and normal light. Functional leaves and roots develop and the plantlets become capable of growing in soil.
Types of Micropropagation
Micropropagation can be carried out by several tissue culture techniques. In these methods small plant tissues or cells are grown under sterile laboratory conditions. Some of the important types are–
- Meristem Culture– In this method the meristematic tissue present at the tip of plant is cultured. These cells divide rapidly and develop into new plants. It is mainly used for producing virus free and disease free plants.
- Shoot Tip and Nodal Culture (Axillary Bud Proliferation)– In this method small shoot tips or nodal segments containing axillary buds are cultured. The buds develop into many shoots under suitable conditions. This method is widely used for clonal propagation of crops such as banana sugarcane and ornamental plants.
- Callus Culture– In this type the explant develops into an unorganized mass of cells called callus. The callus can later differentiate to form new plant tissues or organs. It is used for genetic studies somaclonal variation and production of secondary metabolites.
- Cell Suspension Culture– In this method isolated plant cells or small groups of cells are grown in liquid nutrient medium. The medium is kept in constant movement so that the cells remain suspended. This method is useful for large scale production of plant metabolites.
- Somatic Embryogenesis– In this process somatic cells are induced to form embryos. These embryos develop both shoot and root systems and finally grow into complete plants.
- Organogenesis (Organ Culture)– In this method plant tissues are induced to develop specific organs such as roots shoots or leaves. The organ formation occurs under the influence of plant growth regulators.
- Protoplast Culture– In this method plant cells without cell wall called protoplasts are cultured. The cell wall is removed by enzymatic treatment. This technique is useful for genetic engineering and fusion of cells from different plant species.
- Anther and Pollen Culture– In this method anthers or pollen grains are cultured in vitro. These cultures produce haploid plants which are useful in plant breeding for obtaining homozygous lines.
- Embryo Culture-0 In this technique embryos are removed from seeds and grown in artificial nutrient medium. It helps in the development of embryos that may fail to grow under natural conditions.
- Synthetic Seed Culture– In this method somatic embryos shoot tips or buds are enclosed in protective gel materials such as sodium alginate. These structures behave like seeds and can be stored transported and planted easily.
Advantages of Micropropagation
- Rapid mass multiplication – It is the method by which a very small piece of plant tissue is used to produce a large number of plants in a short time. Thousands or even millions of plants is produced from a single explant under laboratory conditions.
- Disease-free plant production – In this technique sterile culture conditions are maintained and meristem tissues are generally used. Because of this the plants obtained are free from viruses, bacteria and fungi.
- Year-round production – The process is carried out in controlled laboratory conditions. Because of this plant multiplication is not dependent on season, climate or geographical conditions.
- Genetic uniformity – The plants produced through micropropagation are genetically identical clones. These plants shows uniform growth, similar characters and stable yield.
- Propagation of difficult plants – Some plants are difficult to propagate by seeds or conventional vegetative methods. Micropropagation helps in multiplying such elite or sterile plant varieties.
- Conservation of endangered species – It is also used for the conservation of rare and endangered plant species. Large number of plants can be produced without disturbing the natural population.
- Efficient use of space and resources – A large number of plantlets can be produced in a small laboratory space. This reduces the requirement of agricultural land, water and fertilizers.
- Production of valuable metabolites – Plant tissue culture techniques can also be used for the production of important secondary metabolites. These compounds are useful in pharmaceutical, cosmetic and food industries.
- Easy transport of plant materials – The plantlets produced are grown in sterile and soil-free conditions. Because of this they can be transported easily to different countries without risk of spreading pests.
- Basis for genetic improvement – Micropropagation provides plant cells and tissues which can be used in genetic transformation and breeding programmes. It is useful in selecting improved plant varieties.
Disadvantages of Micropropagation
- High cost of establishment – Micropropagation requires special laboratory facilities and equipment. The culture room, laminar air flow cabinet, growth chambers and sterile instruments are required, therefore the initial setup cost is very high.
- Requirement of skilled labour – The technique is mostly carried out manually in laboratory conditions. Skilled technical persons are required for culture preparation, transfer of explants and maintenance of cultures, therefore labour cost is high.
- Risk of contamination – The nutrient medium used in tissue culture contains sugar and other nutrients. These conditions are also suitable for growth of bacteria and fungi, therefore contamination can easily occur and the whole culture may be destroyed.
- Somaclonal variation – Sometimes the plants produced through tissue culture may show genetic changes. These variations results in off-type plants which are not exactly similar to the parent plant.
- Difficulty in acclimatization – Plantlets produced in culture vessels grow in high humidity and controlled conditions. When these plantlets are transferred to soil or greenhouse they may suffer shock and high mortality can occur.
- Loss of genetic diversity – Micropropagation produces large number of genetically identical plants. Because of this the crop population becomes uniform and may become more susceptible to diseases and pests.
- Not suitable for all plant species – Some plants do not respond properly in tissue culture conditions. Woody plants and mature plants are often difficult to propagate by this method.
- Possibility of pathogen multiplication – If the mother plant used for culture is infected with virus or other pathogens, the infection can also be multiplied in the cultured plantlets. Therefore all the plants produced may carry the same infection.
References
- Arizton. (2024). Micropropagation market size, share, trends | global report 2027.
- Bhardwaj, S., Kumari, M., Chethan, T., Hosamani, M., Paul, M., Moirangthem, A., Dash, A. P., & Verma, S. (2025). Advances in micropropagation and tissue culture for horticultural crops: A review. Plant Cell Biotechnology and Molecular Biology, 26(11-12), 166–182. https://doi.org/10.56557/pcbmb/2025/v26i11-1210039
- Biology Discussion. (n.d.). The possible mechanisms causing somaclonal variation.
- Careers360. (n.d.). Micropropagation: Definition, introduction, parts, structure, characteristics.
- Catalina Island Conservancy. (n.d.). Creating a culture.
- Chokheli, V. A., Dmitriev, P. A., Rajput, V. D., Bakulin, S. D., Azarov, A. S., Varduni, T. V., Stepanenko, V. V., Tarigholizadeh, S., Singh, R. K., Verma, K. K., & Minkina, T. M. (2020). Recent development in micropropagation techniques for rare plant species. Plants, 9(12), 1733. https://doi.org/10.3390/plants9121733
- Danova, K., & Pistelli, L. (2022). Plant tissue culture and secondary metabolites production. Plants, 11(23), 3312. https://doi.org/10.3390/plants11233312
- de Oliveira, L. S., Leite, D. M., Mendes, F. M., Molinari, L. V., Brondani, G. E., Gonçalves, A. N., & de Almeida, M. (2024). Micropropagation and in vitro rejuvenation of Eucalyptus cloeziana F. Muell. 3 Biotech, 14(12), 292. https://doi.org/10.1007/s13205-024-04131-2
- Dorge, R. (2026). Global plant tissue culture market size, share 2025 – 2034. Custom Market Insights.
- Duta-Cornescu, G., Constantin, N., Pojoga, D.-M., Nicuta, D., & Simon-Gruita, A. (2023). Somaclonal variation—Advantage or disadvantage in micropropagation of the medicinal plants. International Journal of Molecular Sciences, 24(1), 838. https://doi.org/10.3390/ijms24010838
- Farrelly Mitchell. (n.d.). The advantages and disadvantages of micropropagation.
- Fazili, M. A., Bashir, I., Ahmad, M., Yaqoob, U., & Geelani, S. N. (2022). In vitro strategies for the enhancement of secondary metabolite production in plants: A review. Bulletin of the National Research Centre, 46(1), 35. https://doi.org/10.1186/s42269-022-00717-z
- Food and Agriculture Organization of the United Nations (FAO). (n.d.). Ch09. FAO Knowledge Repository.
- Hasnain, A., Naqvi, S. A. H., Ayesha, S. I., Khalid, F., Ellahi, M., Iqbal, S., Hassan, M. Z., Abbas, A., Adamski, R., Markowska, D., Baazeem, A., Mustafa, G., Moustafa, M., Hasan, M. E., & Abdelhamid, M. M. A. (2022). Plants in vitro propagation with its applications in food, pharmaceuticals and cosmetic industries; current scenario and future approaches. Frontiers in Plant Science, 13, 1009395. https://doi.org/10.3389/fpls.2022.1009395
- Lab Associates B.V. (n.d.). Somaclonal variation in plant tissue culture.
- Liao, Z., Chen, M., Sun, X., & Tang, K. (2006). Micropropagation of endangered plant species. In Plant Cell Culture Protocols (pp. 179-185). Humana Press. https://doi.org/10.1385/1-59259-959-1:179
- Majumder, S., Igamberdiev, A. U., & Debnath, S. C. (2025). Somaclonal variation and clonal fidelity in commercial micropropagation: Challenges and perspectives. Agronomy, 15(6), 1489. https://doi.org/10.3390/agronomy15061489
- Malá, J., & Bylinský, V. (2004). Micropropagation of endangered species Daphne cneorum. Biologia Plantarum, 48(4), 633–636. https://doi.org/10.1023/B:BIOP.0000047167.18592.f7
- Market Reports World. (2026). *Plant tissue culture market size & trends research *.
- Mukhopadhyay, R. (2023). Micropropagation for the improved production of secondary metabolites. In Plants as Bioreactors for Industrial Molecules (pp. 161–184). https://doi.org/10.1002/9781119875116.ch6
- Murthy, H. N., Joseph, K. S., Paek, K. Y., & Park, S. Y. (2023). Bioreactor systems for micropropagation of plants: Present scenario and future prospects. Frontiers in Plant Science, 14, 1159588. https://doi.org/10.3389/fpls.2023.1159588
- P&S Intelligence. (n.d.). Micropropagation market size, share, and trends analysis.
- Pawłowska, B., & Ptak, A. (2023). Application of in vitro culture and biotechnology for the protection of endangered national plant species in Poland. Acta Societatis Botanicorum Poloniae, 92(1). https://doi.org/10.5586/asbp.9202
- Research and Markets. (2026). Micropropagation market size, competitors & forecast to 2030.
- Sathee Forum. (n.d.). Discuss the advantages and limitations of tissue culture techniques such as micropropagation and somatic hybridization?
- Sidhu, Y. (2011). In vitro micropropagation of medicinal plants by tissue culture. The Plymouth Student Scientist, 4(1), 432–449.
- Singh, A. (2025, November 6). Can we scale up tree propagation with tissue culture? Plant Cell Technology.
- Singh, A. (2025, November 11). When is somaclonal variation a tool, not a flaw? Plant Cell Technology.
- Sota, V., Wilms, H., Yucesan, B., Mendi, Y. Y., Christie, B., Nisler, J., Aslan, Ş. E., Purmale-Trasune, L., Silvestri, C., Werbrouck, S. P. O., Fischerová, L., Aronen, T., Cvjetković, B., & Lambardi, M. (2025). Challenges in the micropropagation of economically important fruit species in Europe. Plant Cell, Tissue and Organ Culture, 162(3), 53. https://doi.org/10.1007/s11240-025-03165-5
- SynTech Research. (n.d.). Banana crop health: How SynTech Research is combating global banana diseases.
- The Business Research Company. (2025). Micropropagation global market report 2025.
- The Business Research Company. (2026). Micropropagation market share, size, trends, analysis, report 2035.
- The Royal Horticultural Society. (n.d.). Micropropagation.
- Valliath, A. S., & Mondal, R. (2023). Micropropagation of strawberry crop (Fragaria ananassa): A review. Bhartiya Krishi Anusandhan Patrika, 38(1), 41–44. https://doi.org/10.18805/BKAP529
- Vidal, N., & Sánchez, C. (2019). Use of bioreactor systems in the propagation of forest trees. Engineering in Life Sciences, 19(12), 896–915. https://doi.org/10.1002/elsc.201900041
- Voora, V., Larrea, C., Luna, E., Bermudez, S., & Farrell, J. J. (2023). Global market report: Banana prices and sustainability. International Institute for Sustainable Development.
- Wang, X., Wu, R., Lin, X., Bai, Y., Song, C., Yu, X., Xu, C., Zhao, N., Dong, Y., & Liu, B. (2013). Tissue culture-induced genetic and epigenetic alterations in rice pure-lines, F1 hybrids and polyploids. BMC Plant Biology, 13, 77. https://doi.org/10.1186/1471-2229-13-77
- Zamani, M., Sonboli, A., Goldansaz, M., & Mirjalili, M. H. (2024). In vitro micropropagation and conservation of endangered medicinal plant Nepeta asterotricha Rech.f. (Lamiaceae): Genetic fidelity, phytochemical and biological assessment. Physiology and Molecular Biology of Plants, 30(1), 67–80. https://doi.org/10.1007/s12298-024-01416-x