What is Hydrogen Sulfide (H2S) Test?
The Hydrogen Sulfide (H2S) test is a routine biochemical method used in bacteriology, it is the process by which the capacity of a strain to produces hydrogen sulfide gas is detected in selective or differential media, this is referred to as an important aid for separating Salmonella and Proteus species that are H2S-positive from Shigella and Escherichia coli that is H2S-negative.
It is the principle that certain microorganisms breakdown sulfur-containing compounds such as cysteine or thiosulfate and the sulfur is reduced into colorless H2S during their metabolism, the gas itself cannot be seen therefore heavy metal salts (ferrous or lead) are supplied in the medium so the liberated H2S is immediately trapped and a black insoluble precipitate is formed.
The reaction is as follows– H₂S released from the growing cells combine with Fe²⁺ ion and ferrous sulfide is deposited as blackening, absence of this color means the organism failed to liberate the gas, it is the direct visual evidence used for reporting.
SIM medium, Triple Sugar Iron agar and occasionally a lead acetate paper over cysteine peptone water are the usual media, these are prepared with sodium thiosulfate as the major sulfur source and ferrous ammonium sulfate as indicator, the low agar concentration in SIM also permits observation of motility together with H2S.
In this step a well-isolated colony is touched by a straight inoculating wire, the wire is stabbed once through the center of SIM tube or into the butt of TSI slant, tubes is incubated at 35-37 °C for about 18–24 h, if paper strip is employed it is moistened with sterile water and fixed at the neck so the rising gas react upon it.
Black color develops along the stab line or throughout the butt whenever motile culture spread, no blackening but only growth indicates a negative result, it is the simple observation that can be made without additional reagent.
Positive test therefore signifies liberation of hydrogen sulfide and supports identification of Salmonella or Proteus in clinical, food or water samples, it is often kept together with indole, motility, carbohydrate reaction and urease pattern for presumptive grouping.
The reaction is influenced by size of inoculum and period of incubation, over-grown cultures sometimes gives weak or diffused precipitate, a few Citrobacter strains may also turn positive so it is the good practice that the H2S test is interpreted along with other biochemical evidence.
Principle of Hydrogen Sulfide (H2S) Test
Hydrogen sulfide (H2S) test is based on the ability of microorganism to reduce the sulphur containing compounds and produce hydrogen sulfide gas. It is mainly done by enzymatic activity of bacteria.
In this process bacteria decomposes organic sulphur containing amino acids like cysteine and methionine and H2S is produced. It can also be produced by reduction of inorganic sulphur compounds such as thiosulfate sulfate and sulfite.
Hydrogen sulfide is a colourless gas so it cannot be seen directly. Therefore the medium or strip is containing heavy metal salt indicator like iron salts (ferrous ammonium sulfate) or lead acetate.
When H2S is produced it reacts with metal ions and insoluble black precipitate is formed. The precipitate is iron sulfide (FeS) or lead sulfide (PbS).
This black coloration in medium or on treated indicator paper is referred to as positive result for hydrogen sulfide production.
Objective of Hydrogen Sulfide (H2S) Test
- To detect hydrogen sulfide production in bacteria. It is used to determine that bacterial strain is able to metabolize and reduce sulphur containing compounds and H2S gas is produced.
- To presumptively identify bacteria. It is helpful in identification and characterization of unknown pathogenic and non-pathogenic bacterial isolates.
- To differentiate enteric pathogens. It is used to differentiate members of Enterobacteriaceae family from other Gram negative bacilli and to separate H2S positive pathogens (Salmonella, Proteus) from H2S negative pathogens (Shigella).
- To identify fastidious or non-fermenting organisms. It is used in identification of bacteria like Brucella, Francisella, Shewanella and Bacteroides species.
- To distinguish specific Gram positive bacteria. It is used to separate Lactobacilli (H2S negative) from Erysipelothrix species (H2S positive).
- To test environmental water quality. It is used as low cost portable screening tool for detection of fecal coliform contamination and to check microbiological safety of drinking water in resource limited conditions.
Requirements for Hydrogen Sulfide (H2S) Test
- Culture media for tube methods– SIM (Sulfide Indole Motility) medium. KIA (Kligler’s Iron Agar). TSI (Triple Sugar Iron) agar. Lead acetate agar.
- Culture media for plate methods– Salmonella-Shigella (SS) agar. Deoxycholate citrate agar (DCA). Bismuth sulfite (BS) medium. Xylose-lysine deoxycholate (XLD) agar. Hektoen enteric (HE) agar.
- Culture media for lead acetate paper method– Nutrient broth. Peptone water. Tryptic soy broth.
- Reagents and indicators– Heavy metal salts in medium (ferrous sulfate, ferric ammonium citrate, ferric citrate, peptonized iron). Lead acetate test strips or indicator paper (for lead acetate paper method).
- Equipment– Test tubes and petri plates. Inoculating wire/needle and inoculating loop. Incubator (35°C to 37°C). Bunsen burner. Autoclave. Weighing machine.
- General laboratory materials– PPE and basic laboratory materials is required for safe handling.
- Test organisms for quality control– Positive control (H2S producers) Proteus mirabilis, Proteus vulgaris or Salmonella species (Salmonella Typhimurium). Negative control (H2S non-producers) Shigella flexneri or Escherichia coli.
Culture Media Used for H2S Test
1. Sulfide-Indole-Motility (SIM) Medium
Composition (per 1000 mL)–
- HM Peptone B / Beef extract– 3.0 g
- Peptone– 30.0 g
- Peptonized iron or Ferrous ammonium sulfate– 0.02 to 0.2 g
- Sodium thiosulfate– 0.025 g
- Agar– 3.0 to 3.5 g
- Distilled water– 1000 mL (Final pH 7.3)
Preparation steps–
- Required amount of SIM agar powder is measured and it is mixed in required volume of water.
- It is stirred well and it is heated to boiling for complete dissolution.
- About 5 mL of medium is dispensed in each test tube and it is loosely capped.
- Tubes are autoclaved at 121°C and 15 lbs pressure for 15 minutes.
- Tubes are allowed to cool and solidify in upright position so deep agar butt is formed.
2. Triple Sugar Iron (TSI) Agar
Composition (per 1000 mL)–
- Pancreatic digest of casein– 10.0 g
- Peptic digest of animal tissue– 10.0 g
- Glucose– 1.0 g
- Lactose– 10.0 g
- Sucrose– 10.0 g
- Ferrous sulfate or Ferrous ammonium sulfate– 0.2 g
- NaCl– 5.0 g
- Sodium thiosulfate– 0.3 g
- Phenol red– 0.024 g
- Agar– 13.0 g
- Distilled water– 1000 mL (Final pH is maintained around 7.3)
Preparation steps–
- All ingredients are combined and pH is adjusted to 7.3.
- The mixture is boiled so agar is dissolved completely and then it is dispensed into tubes.
- Sterilization is done by autoclaving at 121°C for 15 minutes.
- Tubes are cooled in slanted position to form 2.5 cm butt and 3.8 cm slant.
Note– Kligler’s iron agar (KIA) is similar medium but it contains only glucose and lactose.
3. Hektoen Enteric (HE) Agar
Composition (per 1000 mL)–
- Proteose peptone– 12.0 g
- Yeast extract– 3.0 g
- Lactose– 12.0 g
- Sucrose– 12.0 g
- Salicin– 2.0 g
- Bile salt mixtures– 9.0 g
- Sodium chloride– 5.0 g
- Sodium thiosulfate– 5.0 g
- Ferric ammonium citrate– 1.5 g
- Acid fuchsin– 0.1 g
- Bromothymol blue– 0.065 g
- Agar– 15.0 g
- Distilled water– 1000 mL (Final pH 7.5)
Preparation steps–
- Powder components are mixed in sterile distilled water and it is stirred well.
- Heating is done to boiling so components is dissolved completely.
- This medium is not autoclaved.
- The medium is cooled to about 40–45°C and it is poured into sterile petri plates.
- Plates are allowed to cool and solidify at room temperature.
4. Nutrient Broth / Peptone Water (For Lead Acetate Paper Method)
Composition (per 1000 mL)–
- Peptone– 5.0 g
- Yeast extract– 1.5 g
- Beef extract– 1.5 g
- Sodium chloride– 5.0 g
- Water– 1000 mL (Final pH 7.4)
- Cysteine (optional)– 0.01% can be added for enhancing H2S production.
Preparation steps–
- Broth powder is mixed in required volume of water and it is stirred well.
- Heating is done to boiling for complete dissolution.
- 5 to 10 mL of medium is dispensed into test tubes and it is loosely capped or plugged.
- Tubes are autoclaved at 121°C and 15 lbs pressure for 15 minutes.
Procedure of Hydrogen Sulfide (H2S) Test
1. Tube method (SIM, TSI or KIA)
- Fresh pure culture of test organism (18 to 24 hours old) is taken.
- A well isolated colony is touched by sterile inoculating needle/wire.
- For SIM medium (agar deep)–
- The tube is inoculated by stabbing straight down in centre of medium up to two-thirds or more than halfway to bottom.
- Needle is withdrawn carefully along the same path.
- For TSI or KIA medium (slant and butt)–
- The butt is stabbed first.
- Then the surface of slant is streaked.
- Loose cap is kept on the tube so proper aerobic condition is maintained.
- Tubes are incubated at 35°C to 37°C for 18 to 48 hours.
- After incubation tubes are observed for black precipitate formation (blackening along stab line or whole medium becomes black). It is referred to as positive result.
2. Lead acetate paper method
- Liquid basal medium like nutrient broth or peptone water is taken and it is inoculated with loopful of test organism (0.01% cysteine can be added).
- Lead acetate indicator paper strip is inserted in neck of tube and it is fixed between plug/cap and inner wall.
- Paper strip is kept just above broth and it should not touch the liquid medium (lead is toxic to many bacteria and growth can be inhibited).
- Tube is incubated aerobically at 35°C to 37°C for 18 to 24 hours.
- For slow growing organisms observation can be done daily up to 7 days.
- Lower part of lead acetate strip is examined. Brownish black or black discoloration indicates positive H2S test.
3. Plate method (HE, SS, BS or XLD agar)
- A well isolated colony from fresh culture is touched by sterile inoculating loop.
- Bacteria is streaked on agar plate for getting well isolated single colonies.
- Plates are incubated aerobically at 35±2°C for about 24 hours.
- Colony appearance is observed. Black coloured colonies or colonies with distinct black centres (coloured or colourless) indicates positive H2S reaction.
Result and Interpretation of Hydrogen Sulfide (H2S) Test
Positive result (H2S is produced)
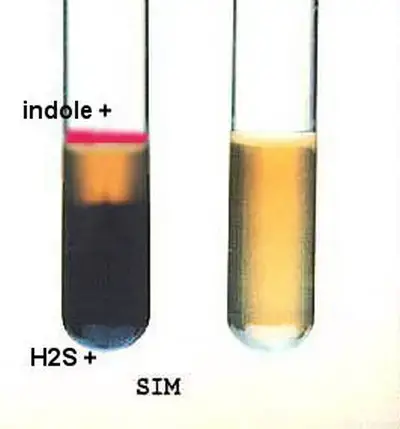
- SIM medium– Agar becomes black along the stab line or whole tube becomes black. It indicates H2S production.
- TSI / KIA agar– Butt portion becomes black. Yellow colour due to carbohydrate fermentation can be masked by this blackening.
- Lead acetate paper– Lower part of indicator paper strip shows brown colour to black discoloration. It is referred to as positive test.
- Plate media (HE, DCA, BS agar)– Colonies appears black or colonies shows distinct black centres. It indicates H2S positive organism.
Negative result (H2S is not produced)
- SIM medium– No blackening is seen and medium remains yellow/amber colour. It indicates H2S is not produced.
- TSI / KIA agar– No blackening in butt portion is observed.
- Lead acetate paper– No colour change is seen on indicator paper strip and blackening is absent.
- Plate media (HE, DCA, BS agar)– Colonies remains colourless or pink or yellow and black coloration is not present. It indicates negative H2S reaction.





List of H2S Test Positive and negative Bacteria
- H2S positive bacteria (Hydrogen sulfide producers)–
- Salmonella species (Salmonella Enteritidis, Salmonella Typhimurium).
- Proteus species (Proteus mirabilis, Proteus vulgaris).
- Citrobacter species (Citrobacter freundii) (reaction can be variable).
- Edwardsiella tarda.
- Shewanella species.
- Brucella species.
- Francisella tularensis.
- Erysipelothrix rhusiopathiae.
- Campylobacter species.
- Staphylococcus saprophyticus.
- Clostridium species (some strains).
- H2S negative bacteria (Hydrogen sulfide non-producers)–
- Escherichia coli.
- Shigella species.
- Klebsiella pneumoniae.
- Staphylococcus aureus.
- Pseudomonas aeruginosa.
- Neisseria gonorrhoeae.
- Vibrio cholerae.
- Yersinia pestis.
Quality Control organisms of Hydrogen Sulfide (H2S) Test
- Positive controls (H2S producers)– Salmonella serotype Enteritidis (ATCC 13076). Salmonella serotype Typhimurium (ATCC 14028). Proteus mirabilis (ATCC 29906). Proteus vulgaris.
- Negative controls (H2S non-producers)– Escherichia coli (ATCC 25922). Shigella flexneri (ATCC 12022). Enterobacter cloacae.
Precautions of H2S Test
- Appropriate medium should be selected. Media like DCA, SS agar and XLD medium should be avoided when Enterobacterales other than Salmonella and Shigella is tested.
- Sucrose interference should be avoided. Sucrose containing medium (TSI agar) should not be used for sucrose fermenter organism because sucrose fermentation can suppress H2S producing enzyme mechanism and false negative result can be obtained.
- Tube media should be inspected before use. Cracks in medium should be checked and cracked tubes should not be used for inoculation.
- Correct inoculation tool should be used. Straight inoculating wire/needle is used for stabbing deep tube media and inoculating loop is not preferred.
- Stabbing should be done carefully. Needle should be withdrawn along same path and lateral movement should be avoided because it can be mistaken as positive motility.
- Aerobic conditions should be maintained. Caps should not be tightened fully during incubation and loose cap is kept for proper aerobic condition.
- Some media should not be autoclaved. HE agar, DCA and Bismuth sulfite (BS) agar is not autoclaved.
- Sterility should be maintained. For those media which are not autoclaved sterile equipment is used and preparation is done in sterile zone.
- In plate method inoculum should be taken in small amount and proper spacing is maintained between streaks so well isolated colonies are obtained.
- In lead acetate paper method paper strip should hang slightly above the liquid and it should not touch broth because lead is toxic to bacteria and growth can be inhibited which gives false negative result.
Uses of Hydrogen Sulfide (H2S) Test
- Differentiation of enteric bacteria is done. It is mainly used to differentiate members of Enterobacteriaceae family and to distinguish H2S producing pathogens (Salmonella, Proteus) from non-producers (Shigella, Escherichia coli).
- Clinical diagnosis of infections is supported. By identification of specific bacterial isolates it helps in diagnosis of pathogens responsible for gastrointestinal infections (Salmonellosis, Typhoid fever) urinary tract infection and wound infection.
- Identification of fastidious and non-fermenting bacteria is done. Lead acetate paper method (highly sensitive) is used for identification of organisms like Brucella species, Francisella tularensis, Shewanella and Erysipelothrix rhusiopathiae.
- Environmental water quality testing is done. It is used as simple low cost portable screening tool for detection of fecal coliform contamination in drinking water and it is helpful in assessing water safety and diarrheal disease risk in field condition and resource limited settings.
Limitations of Hydrogen Sulfide (H2S) Test
- It is presumptive test and it is not confirmatory. For complete identification supplementary biochemical immunological or molecular tests are required.
- Sucrose inhibition can occur. In sucrose containing medium (TSI agar) sucrose fermentation can suppress the enzyme mechanism for H2S production and false negative result is obtained.
- Acidity requirement is present in KIA and TSI. Acidic condition is required for H2S reduction reaction and non-fermenting bacteria may not produce sufficient acid so visible reaction is not seen even if sulphur reduction capacity is present.
- Indicator toxicity is one limitation. Lead acetate is most sensitive indicator but it is toxic to many bacteria and if paper strip touches liquid medium growth can be inhibited and false negative result can be seen.
- Fastidious organisms may not grow properly. Some fastidious bacteria struggles to grow in basic media like SIM nutrient broth or DCA so result can be affected.
- Extended incubation time is required for slow growing organisms. Many enteric bacteria gives H2S within 24 hours but Campylobacter or Brucella may require 3 to 7 days for visible positive reaction.
- Media composition can affect the result. Peptone type matters because different peptones contains different amount of cysteine (substrate) so variable result or false negative result can be obtained.
- In water testing false positive result can occur. Non-fecal sulfate reducing bacteria from soil existing environmental sulfides or microbially induced corrosion in metal pipes can give positive reaction.
- Fecal origin cannot be differentiated. Positive H2S test in water screening cannot discriminate human fecal bacteria and animal fecal bacteria.
- Non-bacterial pathogens cannot be detected. This test detects only H2S producing bacteria and it is not used for virus or parasite detection.
- Lack of standardization is present. Different variations of test (media formulation sample volume incubation temperature) is used and sensitivity varies so comparison becomes difficult.
References
- Anishchenko, E., Vigorito, C., Mele, L., Lombari, P., Perna, A. F., & Ingrosso, D. (2019). Novel applications of lead acetate and flow cytometry methods for detection of sulfur-containing molecules. Methods and Protocols, 2(1), 13.
- Aryal, S. (2022). Hydrogen sulfide test – Principle, procedure, uses and interpretation. MicrobiologyInfo.com.
- Bai, P. (2016). The solid state reaction formula between Fe and H2S? ResearchGate.
- BMH learning. (n.d.). Hydrogen sulphide production test | H2S production test | [Video]. YouTube.
- Comprehensive analytical review of the hydrogen sulfide (H2S) production test in diagnostic microbiology: Principles, biochemical pathways, and clinical methodologies. (n.d.).
- Dahal, P. (2024). Hydrogen sulfide (H2S) test: Principle, procedure, results. Microbe Notes.
- DrChika. (2023). Hydrogen sulphide test. Microbiology Class.
- Gupta, S. K., Sheikh, M. A., Islam, M. S., Rahman, K. S., Jahan, N., Rahman, M. M., Hoekstra, R. M., Johnston, R., Ram, P. K., & Luby, S. (2007). Usefulness of the hydrogen sulfide test for assessment of water quality in Bangladesh. Journal of Applied Microbiology, 104, 388–395.
- Han, S., Li, Y., & Gao, H. (2022). Generation and physiology of hydrogen sulfide and reactive sulfur species in bacteria. Antioxidants, 11(12), 2487.
- Hartline, R. (2023). 1.23: SIM deep tests. In Microbiology Laboratory Manual. Biology LibreTexts.
- Hine, C., & Mitchell, J. R. (2017). Endpoint or kinetic measurement of hydrogen sulfide production capacity in tissue extracts. Bio-Protocol, 7(13), e2382.
- Hydrogen sulfide. (2026). In Wikipedia.
- Hydrogen sulfide production test. (n.d.). VUMIE.
- Instituto Valenciano de Microbiología (IVAMI). (2018). Hydrogen sulfide (H2S)-producing bacteria (sulfate reducers, sulfite reducers, sulfur reducers, and other molecules with sulfur) – Qualitative and quantitative culture; Molecular identification (PCR and sequencing).
- Kaiser, G. (2024). 8.5: Indole and hydrogen sulfie production w. procedures and results. In Microbiology Labs II. Biology LibreTexts.
- Kimura, H. (2014). Production and physiological effects of hydrogen sulfide. Antioxidants & Redox Signaling, 20(5), 783–793.
- Kolluru, G. K., Shen, X., Bir, S. C., & Kevil, C. G. (2013). Hydrogen sulfide chemical biology: Pathophysiological roles and detection. Nitric Oxide, 35, 5–20.
- Lehman, D. (2005). Triple sugar iron agar protocols. American Society for Microbiology.
- Li, K., Xin, Y., Xuan, G., Zhao, R., Liu, H., Xia, Y., & Xun, L. (2019). Escherichia coli uses separate enzymes to produce H2S and reactive sulfane sulfur from L-cysteine. Frontiers in Microbiology, 10, 298.
- Macabrey, D., Joniová, J., Gasser, Q., Bechelli, C., Longchamp, A., Urfer, S., Lambelet, M., Fu, C.-Y., Schwarz, G., Wagnières, G., Déglise, S., & Allagnat, F. (2022). Sodium thiosulfate, a source of hydrogen sulfide, stimulates endothelial cell proliferation and neovascularization. Frontiers in Cardiovascular Medicine, 9, 965965.
- Mathews, E., Barnett, D., Petrovski, S., & Franks, A. (2018). Four common mechanisms of H2S control: a Inhibition of sulfate (SO4²⁻) reduction using alternative electron acceptors. ResearchGate.
- Rickard, D. (1995). Kinetics of FeS precipitation: Part 1. Competing reaction mechanisms. Geochimica et Cosmochimica Acta, 59(21), 4367-4379.
- Shibuya, N., & Kimura, H. (2013). Production of hydrogen sulfide from D-cysteine and its therapeutic potential. Frontiers in Endocrinology, 4, 87.
- Sigma-Aldrich. (2013). 06728 Hydrogen sulfide test strips (Lead acetate test strips, H2S test strips).
- Sobsey, M. D., & Pfaender, F. K. (2002). Evaluation of the H2S method for detection of fecal contamination of drinking water. World Health Organization.
- Stein, A., & Bailey, S. M. (2013). Redox biology of hydrogen sulfide: Implications for physiology, pathophysiology, and pharmacology. Redox Biology, 1(1), 32–39.