Fehling’s test is a chemical analytical test that is used to identify reducing sugars and it is also used to differentiate aldehyde and ketone functional groups. This test was developed in 1849 by German chemist Hermann von Fehling. It is carried out by using Fehling’s reagent which is prepared by mixing two solutions just before use. These are Fehling’s A (deep blue aqueous copper(II) sulphate solution) and Fehling’s B (colourless alkaline solution containing sodium hydroxide and potassium sodium tartrate which is also known as Rochelle salt). After mixing a deep blue copper-tartrate complex is formed and it acts as a mild oxidizing agent.
In this test the sample is added to Fehling’s solution and it is heated gently in a water bath. If reducing sugar or an aliphatic aldehyde is present then a redox reaction occurs where aldehyde is oxidized into carboxylic acid and Cu2+ ions are reduced to Cu+ ions. The result is confirmed when the blue colour changes to brick red or reddish brown precipitate of copper(I) oxide (Cu2O). If the solution remains blue then the test is negative and it indicates ketone, non-reducing sugar like sucrose or an aromatic aldehyde like benzaldehyde.
Fehling’s test is used in laboratory and also in medicine and industry. It was used earlier for detection of excess glucose in urine for diagnosis of diabetes. It is also used in food and beverage industries to monitor sugar content and to measure breakdown of starch into glucose syrups.
Objectives of Fehling’s Test
- To detect the presence of carbohydrates in a solution.
- To identify and differentiate between reducing sugar and non-reducing sugar.
- To detect the presence of aldehyde group (–CHO) in organic compounds.
- To distinguish between aldehydes and ketones in carbohydrates and organic compounds since aliphatic aldehydes reacts but most ketones does not react.
- To detect glucose in urine or blood for medical diagnosis and screening of diabetes.
- To monitor sugar content and hydrolysis of starch in food and beverage industries and it helps in measuring reduced sugar and calculating Dextrose Equivalent (DE).
Principle of Fehling’s Test
The principle of Fehling’s test is based on a redox reaction for detection of aliphatic aldehydes and reducing sugars. When the test sample is heated with Fehling’s reagent (deep blue alkaline solution containing copper(II) ions stabilized by tartrate ions) the aldehyde group acts as a reducing agent. In this reaction the aldehyde is oxidized into the corresponding carboxylic acid (or carboxylate form) and at same time the blue Cu2+ ions are reduced to Cu+ ions.
Due to this reduction an insoluble brick red precipitate of copper(I) oxide (Cu2O) is formed and it indicates positive result. Ketones generally do not show this reduction (except alpha-hydroxy ketones) and aromatic aldehydes also cannot reduce the copper ions properly so the solution remains blue and the test is considered negative.
Fehling’s test reaction
The reaction of Fehling’s test is a redox reaction that occurs in strongly alkaline condition. When a sample containing an aliphatic aldehyde or reducing sugar is heated with Fehling’s reagent the aldehyde group donates electrons and it is oxidized to the corresponding carboxylate ion (RCOO⁻). At the same time the deep blue copper(II) ions (Cu²⁺) present in the reagent (stabilized by tartrate ions) accept the electrons and it is reduced to copper(I) ions (Cu⁺). Under alkaline condition these Cu⁺ ions react immediately with hydroxide ions and copper(I) oxide (Cu2O) is formed as an insoluble precipitate.
The reaction is as follows-
RCHO + 2Cu²⁺ + 5OH⁻ → RCOO⁻ + Cu2O↓ + 3H2O
Due to formation of insoluble Cu2O the colour changes from deep blue to brick red or reddish brown precipitate and it indicates positive result.
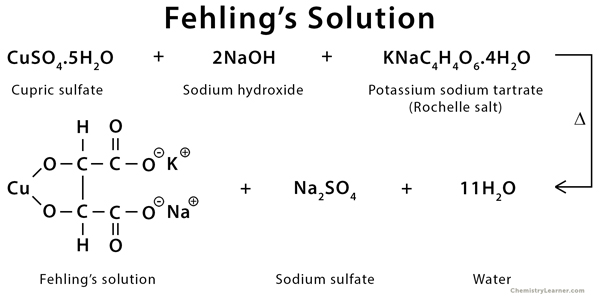
What is Fehling’s Solution?
- Fehling’s solution is a deep blue chemical reagent that is used for detection of aliphatic aldehydes and reducing sugars. It is also used to distinguish them from ketones and non-reducing sugars.
- The complete reagent is unstable and it decomposes slowly on standing. Because of this it is prepared freshly just before use by mixing equal volume of two solutions i.e. Fehling’s A and Fehling’s B.
- Fehling’s A is a deep blue aqueous solution of copper(II) sulphate (CuSO4). It provides the active oxidizing copper ions (Cu2+).
- Fehling’s B is a colourless alkaline solution containing sodium hydroxide (NaOH) and potassium sodium tartrate (Rochelle salt).
- When Fehling’s A and Fehling’s B are mixed, tartrate ions act as a chelating agent and it binds with copper(II) ions forming a soluble deep blue copper-tartrate complex (bistartarocuprate(II) complex).
- This complex formation prevents precipitation of copper as copper hydroxide in strong alkaline medium and the reagent remains effective as a mild oxidizing agent for the test.
Requirements for Fehling’s Test
- Fehling’s Solution A– Aqueous solution of copper(II) sulphate pentahydrate (CuSO4.5H2O).
- Fehling’s Solution B– Colourless aqueous solution containing potassium sodium tartrate (Rochelle salt) and strong alkali such as sodium hydroxide (NaOH) or potassium hydroxide (KOH).
- Fehling’s reagent– Freshly prepared mixture made by mixing equal volume of Fehling’s A and Fehling’s B just before use.
- Test sample– The substance to be tested (generally prepared as 5% solution) like glucose, fructose, sucrose or starch.
- Control sample– Distilled water.
- Test tubes.
- Test tube stand.
- Pipette / droppers.
- Water bath (for gentle heating for few minutes).
- Safety equipments– gloves and safety goggles because Fehling’s solution is toxic and corrosive.
Fehling’s Test Procedure
- Preparation of Fehling’s reagent– Equal volume of Fehling’s A (blue copper(II) sulphate solution) and Fehling’s B (colourless alkaline sodium potassium tartrate solution) is mixed to prepare deep blue Fehling’s reagent. It is prepared freshly just before use because the mixed reagent is unstable and on standing it can decompose forming copper hydroxide or black copper oxide.
- Preparation of sample- Small amount of the test sample is taken in a clean dry test tube (about 1 mL or few drops). Another test tube containing distilled water is taken as control.
- Addition of reagent– Freshly prepared Fehling’s reagent is added into the test tube containing sample and it is mixed properly.
- Heating– The test tube is kept in a hot water bath (about 60°C to 100°C) and it is heated gently for few minutes. Direct heating over flame is avoided because the copper-tartrate complex may break down to black copper(II) oxide and it can mask the positive result.
- Observation– Formation of brick red or reddish brown precipitate indicates positive result and it confirms presence of aliphatic aldehyde or reducing sugar. If the solution remains deep blue without colour change then it indicates negative result.
Result of Fehling’s Test
- Positive result– The deep blue solution changes colour and brick red or reddish brown precipitate is formed (copper(I) oxide– Cu2O). It indicates presence of aliphatic aldehydes or reducing sugars like glucose, fructose, lactose and maltose.
- Negative result- No colour change is seen and the solution remains deep blue. It indicates absence of aliphatic aldehydes and reducing sugars. It may be due to ketone, non-reducing sugar (sucrose) or aromatic aldehyde (benzaldehyde).

Uses of Fehling’s Test
- It is used to distinguish between aldehydes and ketones in organic chemistry. Aliphatic aldehydes reacts with Fehling’s solution but ketones generally does not react.
- It is used as a general qualitative test to identify reducing sugars (monosaccharides and other reducing sugars) and to differentiate them from non-reducing carbohydrates.
- It is used in medical diagnosis to detect excess glucose in urine and it helps in diagnosis and monitoring of diabetes.
- It is used in industries to measure starch breakdown. It measures the amount of reducing sugars formed during conversion of starch into glucose syrups and maltodextrins and it helps in determining Dextrose Equivalent (DE).
- It is used in food and beverage industries to determine and monitor the sugar content in different food and beverage products.
- It is sometimes used in forensic investigations to detect biological residues like presence of blood stains.
Advantages of Fehling’s Test
- It is simple and low cost test. It is quick and easy to perform and no special equipment is required.
- The result is clear by visual observation. Brick red precipitate is formed when reducing group is present.
- It is reliable for differentiation. It is specific for aliphatic aldehydes and it helps in distinguishing them from ketones and aromatic aldehydes.
- The reaction is fast and vigorous because Fehling’s reagent is strongly alkaline. It is faster than some other similar test like Benedict’s test.
- It is used as a screening test in different sectors. It is useful for monitoring sugar content and starch hydrolysis in food production and it was also used earlier for diabetes diagnosis.
- It has educational value in chemistry practical. It helps in understanding oxidation reaction, copper complex formation and sugar structure by simple colour change.
- Reproducibility is good because tartrate ligand stabilizes Cu2+ ions in alkaline medium and the test gives reliable result.
Limitations of Fehling’s Test
- It fails to detect aromatic aldehydes (benzaldehyde etc.) because the aromatic ring stabilizes the carbonyl group and it does not react properly with the mild oxidizing agent.
- It cannot detect most ketones. Only alpha-hydroxy ketones may give positive result but other ketones generally does not react.
- Highly alkaline pH is required for the test. Under acidic condition the test fails because copper(II) ions becomes stabilized and reaction does not occur.
- Fehling’s reagent is unstable after mixing. It cannot be stored in premixed form and it must be prepared freshly by mixing Fehling’s A and Fehling’s B just before use.
- Interference is common in biological samples. Other reducing agents like uric acid, Vitamin C (ascorbic acid) and some antibiotics (streptomycin, cephalosporins) can give false positive or false negative result.
- Specificity is limited. It only shows presence of reducing sugar but it cannot differentiate between different reducing sugars.
- It is mainly a qualitative test and quantitative accuracy is low. It is not highly sensitive for precise measurement of sugar concentration.
- Safety hazard is present. Fehling’s solution contains sodium hydroxide so it is corrosive and toxic to skin and eyes. Copper salts are also toxic and it can harm environment and aquatic life.
References
- ALLEN Career Institute. (n.d.). Assertion:-Aliphatic aldehyde can be diffrentiate from aromatic aldehyde by Fehling solution. Reason:- This is because aliphatic aldehyde reduce fehling solution but aromatic aldehyde donot. Retrieved from https://allen.in/dn/qna/646658029
- ALLEN Career Institute. (n.d.). Fehling’s solution: Preparation, tests and application. Retrieved from https://allen.in/jee/chemistry/fehlings-solution
- babyshark7379. (n.d.). Lobry de Bruyn rearrangement [PDF]. Scribd. Retrieved from https://www.scribd.com/document/970227966/Lobry-de-Bruyn-Rearrangement
- BYJU’S. (n.d.). Fehling test. Retrieved from https://byjus.com/chemistry/fehling-test/
- BYJU’S. (n.d.). Fehlings solution. Retrieved from https://byjus.com/jee/fehlings-solution/
- Chemistry Stack Exchange. (2016, February 7). Why does benzaldehyde not respond to Fehling’s test? Retrieved from https://chemistry.stackexchange.com/q/44872
- Chemistry Stack Exchange. (2016, November 2). Functional difference of Benedict’s solution and Fehling’s solution. Retrieved from https://chemistry.stackexchange.com/q/62034
- Comprehensive analysis of Fehling’s test: Principles, methodologies, and analytical implications in carbonyl chemistry. (n.d.).
- Diagnox Health. (n.d.). What can cause false results on a protein urine test? Retrieved from https://www.diagnoxhealth.com/product-faqs/what-can-cause-false-results-on-a-protein-urine-test
- ECHEMI. (n.d.). Why does fructose reduce Tollen’s reagent and Fehling’s solution? Retrieved from https://www.echemi.com/community/why-does-fructose-reduce-tollen-s-reagent-and-fehling-s-solution_mjart2204292771_773.html
- Filo. (2025, May 1). What is the composition fehling reagent.
- Filo. (2025, June 13). Fehling’s test is used to detect: All types of aldehydes Aromatic aldehy..
- Filo. (2026, January 4). Question: Why do aromatic aldehydes not give a reddish-brown precipitate ..
- Fleischer, H. (2019). The iodine test for reducing sugars – A safe, quick and easy alternative to copper(II) and silver(I) based reagents. World Journal of Chemical Education, 7(2), 45-52. https://doi.org/10.12691/wjce-7-2-3
- Foist, L. (n.d.). Reducing vs. non-reducing sugar | Definition & comparison [Video]. Study.com. Retrieved from https://study.com/academy/lesson/video/reducing-vs-non-reducing-sugars-definition-comparison.html
- GeeksforGeeks. (2025, July 23). Fehling’s solution. Retrieved from https://www.geeksforgeeks.org/chemistry/fehlings-solution/
- Interferences: False positive false negative urine samples with a … (n.d.).
- LibreTexts. (2019, June 5). 16.2: Oxidation of aldehydes. Chemistry LibreTexts. Retrieved from https://chem.libretexts.org/Courses/Purdue/Purdue%3A_Chem_26605%3A_Organic_Chemistry_II_(Lipton)/Chapter_16._Oxidation_and_Reduction/16.2%3A_Oxidation_of_Aldehydes
- LUMITOS AG. (n.d.). Lobry-de Bruyn-van Ekenstein transformation. chemeurope.com. Retrieved from https://www.chemeurope.com/en/encyclopedia/Lobry-de_Bruyn-van_Ekenstein_transformation.html
- Mohamed, A. M. H. (n.d.). CHEMISTRY 2 (BIOCHEMISTRY). Department of Biochemistry, Benha University, Agriculture College.
- National Center for Biotechnology Information. (n.d.). Interference in autoanalyzer analysis – PMC – NIH.
- Quick Biochemistry Basics. (n.d.). Difference between fehling and benedict test [Video]. YouTube. Retrieved from https://www.youtube.com/watch?v=3yvsoWgtyE0
- Rani anak mat. (2022, October 20). Fehling’s test for carbohydrate analysis [PDF]. Scribd. Retrieved from https://www.scribd.com/document/612264054/analysis-biomolecule-lab-report-2
- Sapkota, A. (2022, September 5). Fehling’s test- Definition, principle, procedure, result, uses. Microbe Notes. Retrieved from https://microbenotes.com/fehlings-test/
- Shang, K., Wang, S., Chen, S., & Wang, X. (2022). Sensitivity detection of uric acid and creatinine in human urine based on nanoporous gold. Biosensors, 12(8), 588. https://doi.org/10.3390/bios12080588
- Syal, K., Banerjee, D., & Srinivasan, A. (2013). Creatinine estimation and interference. Indian Journal of Clinical Biochemistry, 28(2), 210–211. https://doi.org/10.1007/s12291-013-0299-y
- Taylor & Francis. (n.d.). Fehling’s solution – Knowledge and references.
- Testbook. (n.d.). Fehling test – Reaction, reagent, mechanism, and difference between Fehling’s and Benedict’s test.
- Testbook. (n.d.). [Solved] Assertion (A) : Aromatic aldehydes do not respond to Fehling. Retrieved from https://testbook.com/question-answer/assertion-a-aromatic-aldehydes-do-not-respond–687622fbfe95b46dc02cc166
- Testbook. (n.d.). Understanding Fehling’s solution: Formation, test procedure, reactions & uses.
- Vedantu. (n.d.). Fehling test – Principle, procedure, result & applications. Retrieved from https://www.vedantu.com/chemistry/fehling-test
- Wikipedia contributors. (2025, December 2). Lobry de Bruyn–Van Ekenstein transformation. In Wikipedia, The Free Encyclopedia. Retrieved from https://en.wikipedia.org/wiki/Lobry_de_Bruyn%E2%80%93Van_Ekenstein_transformation
- Wikipedia contributors. (2026, February 28). Fehling’s solution. In Wikipedia, The Free Encyclopedia. Retrieved from https://en.wikipedia.org/wiki/Fehling%27s_solution
- Zhang, Y., & Chen, Q. (2020, July 21). Improving measurement of reducing sugar content in carbonated beverages using Fehling’s reagent. Journal of Emerging Investigators.