What is Bombardment (Biolistics) Method?
Bombardment (Biolistics) method is a direct physical technique used to introduce foreign DNA into living cells. It is also referred to as the gene gun method. In this process, the desired DNA is first coated on very small and dense metal particles such as gold or tungsten. These DNA coated particles are then loaded into a special device called gene gun. The particles are accelerated at very high velocity with the help of pressurized gas such as helium. Due to the high kinetic energy, these microprojectiles penetrate the cell wall and plasma membrane of the target cells and the DNA is delivered directly inside the cell without causing severe damage. This method does not depend on any biological vector and therefore it is useful for organisms that are resistant to bacterial transformation. It is widely used for the transfer of genes into plant tissues, animal cells, microbes and also into organelles such as chloroplasts.
Principle of Bombardment (Biolistics) Method
The principle of bombardment (biolistics) method is based on the direct physical delivery of foreign genetic material into the cells. In this method, microscopic particles of heavy metals such as gold or tungsten are coated with the desired DNA or RNA molecules. These coated particles are then accelerated to very high velocity by using a gene gun device with the help of high pressure helium gas. The sudden force generated inside the chamber propels the microprojectiles towards the target cells. Due to the high kinetic energy, the particles penetrate the cell wall and plasma membrane and the genetic material is deposited inside the cell. This method depends on physical force rather than biological infection and therefore it can bypass the cellular barriers. Because of this property, the method can be used for the transformation of different tissues, intracellular organelles and also plant species that are resistant to other biological gene transfer methods.
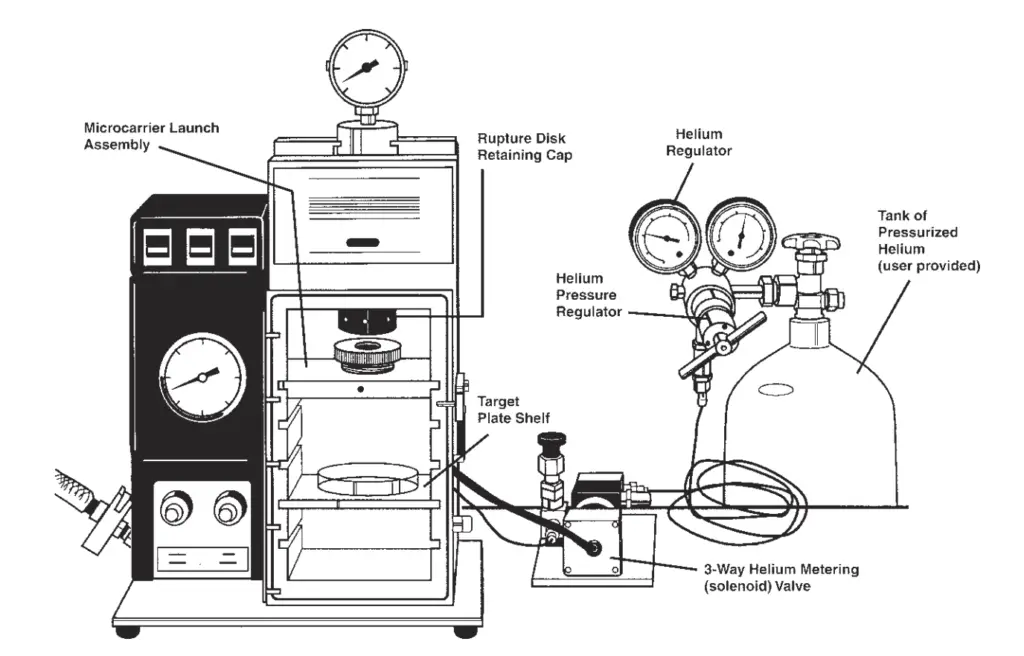
Materials Required for Bombardment (Biolistics) Method
The important materials required for bombardment (biolistics) method are as follows–
– Gene gun device – It is the particle delivery system used for accelerating the DNA coated particles into the target cells.
– Helium gas supply system – High pressure helium gas cylinder with regulator is used to generate the force required for particle acceleration.
– Vacuum chamber and vacuum pump – It is used to create partial vacuum inside the bombardment chamber for proper particle movement.
– Microcarriers (gold or tungsten particles) – These are microscopic heavy metal particles that act as carriers for the foreign DNA.
– Macrocarrier discs – Small plastic discs on which the DNA coated microcarriers are loaded before bombardment.
– Rupture discs – These are thin membranes that burst at specific helium pressure to produce the shock wave required for particle acceleration.
– Stopping screens – Fine metal mesh that stops the macrocarrier but allows the microscopic particles to pass towards the target cells.
– Nucleic acids (DNA or RNA) – The foreign genetic material that is to be introduced into the cells.
– Calcium chloride (CaCl₂) – It is used to help the binding of DNA onto the metal microcarrier particles.
– Spermidine solution – It helps in condensing and stabilizing the DNA around the metal particles.
– Ethanol – Used for washing and sterilizing the particles and equipment during the preparation process.
– Target biological material – Living cells or tissues such as plant callus, embryos, microbes or animal cells which receive the foreign DNA.
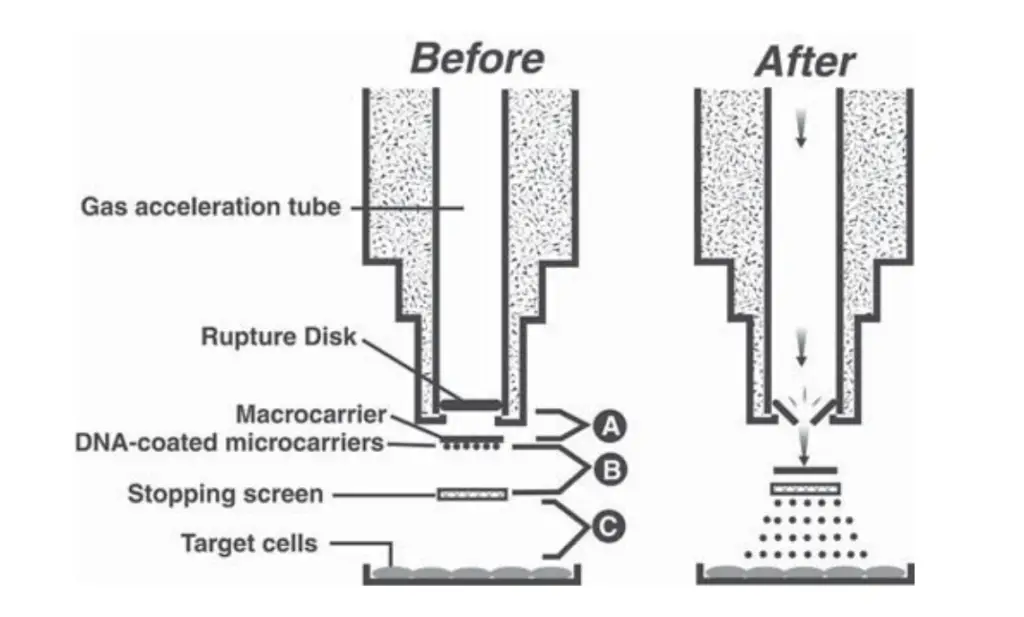
Protocol for Bombardment (Biolistics) Method
The bombardment method is a physical method of gene transfer in which DNA-coated microprojectiles are accelerated into plant cells or tissues using a gene gun. This process is referred to as biolistic transformation.
The protocol is as follows–
- Preparation and sterilization of equipment– The interior of the gene gun bombardment chamber (Bio-Rad PDS-1000/He) is cleaned with 70% ethanol. The rupture disks, stopping screens, macrocarriers and macrocarrier holders are soaked in 70% or 100% ethanol and then allowed to air dry in laminar flow hood.
- Preparation of microcarriers– Gold or tungsten microparticles (about 30–60 mg) are taken in a microfuge tube. These particles are vortexed with 70% ethanol and centrifuged to pellet the particles. The supernatant is discarded and the particles are washed with sterile water or absolute ethanol.
- Resuspension of microcarriers– After washing, the microcarriers are resuspended in 50% sterile glycerol or 100% ethanol at a concentration of about 60 mg/mL.
- Dispersion of microcarriers– The microcarrier suspension is vortexed for several minutes. This step is carried out so that the particles remain evenly dispersed and aggregation of particles is avoided.
- Transfer of microcarrier suspension – About 50 µL of the prepared microcarrier suspension is transferred into a fresh microcentrifuge tube.
- Addition of plasmid DNA– While vortexing continuously, plasmid DNA (about 5 µL containing approximately 1 µg/µL DNA) is added to the suspension.
- Addition of calcium chloride– Calcium chloride (CaCl₂) solution (about 50 µL of 2.5 M solution) is added dropwise into the vortexing mixture.
- Addition of spermidine– Spermidine solution (about 20 µL of 0.1 M) is added. These reagents facilitate precipitation of DNA onto the surface of microcarriers.
- Mixing and precipitation of DNA– The mixture is vortexed for about 2–3 minutes and then allowed to settle for about 1 minute. This step results in coating of DNA on microcarrier particles.
- Pelleting of DNA-coated microcarriers– The mixture is centrifuged briefly so that DNA-coated microcarriers are pelleted. The supernatant is discarded carefully.
- Washing of pellet – The pellet is washed once with 70% ethanol and then washed with 100% ethanol one or two times. The supernatant is discarded each time without disturbing the pellet.
- Final suspension of coated particles– The final pellet is gently resuspended in a small volume (about 48–60 µL) of 100% ethanol.
- Loading of macrocarrier disk– While vortexing the suspension, about 6–10 µL of the DNA-microcarrier mixture is pipetted onto the center of a sterile macrocarrier disk. The ethanol is allowed to evaporate completely for a few minutes.
- Adjustment of helium pressure– The main valve of the helium cylinder is opened. The regulator pressure is adjusted approximately 200 psi higher than the rupture disk rating.
- Installation of rupture disk– A sterile rupture disk is placed into the retaining cap and tightly fixed onto the gas acceleration tube of the bombardment chamber.
- Assembly of launch unit– A sterile stopping screen is placed in the microcarrier launch assembly. The macrocarrier holder containing the DNA-coated macrocarrier is positioned above the screen and the assembly is secured.
- Placement of target tissues– The Petri dish containing the target tissues (embryos, callus or leaves) is placed on the target shelf inside the chamber at a suitable distance from the stopping screen.
- Evacuation of bombardment chamber– The chamber door is closed and the vacuum pump is switched on. The chamber is evacuated to a pressure of about 25–29 inches of mercury.
- Firing of the gene gun– The fire button is pressed. Helium pressure increases until the rupture disk bursts producing a shockwave which accelerates the DNA-coated microcarriers toward the target cells.
- Recovery of bombarded tissues– The vacuum is released and the Petri dish is removed carefully. The bombarded tissues are transferred to recovery conditions (usually kept in dark for 12–24 hours) before applying selection or regeneration of transformed tissues.
Advantages of Bombardment (Biolistics) Method
- Universal host range – It is a genotype-independent method and can be used for transformation of almost all plant species. It is very useful for monocot plants such as cereals which are generally resistant to Agrobacterium infection.
- Broad tissue compatibility – It can transform many types of tissues such as leaves, embryos, callus and seedlings. In this method removal of cell wall to form protoplast is not required which makes the procedure easier.
- Direct transformation of organelles – It can deliver DNA directly into organelles such as chloroplast and mitochondria. This type of transformation is very difficult by other methods.
- Delivery of different biological molecules – The system can introduce many kinds of biological materials such as plasmid DNA, linear DNA fragments, RNA molecules and even proteins into the cell.
- No requirement of special vectors – In this method the gene of interest does not need to be inserted into special binary T-DNA vectors as required in Agrobacterium mediated transformation.
- Efficient co-transformation – Different plasmids or genes can be coated on the same microparticle and delivered simultaneously into a cell. Thus multiple genes can be transferred at the same time with high frequency.
- Simple and rapid procedure – The method is based on physical delivery of DNA coated particles and the protocol is relatively fast and simple. Only small amount of nucleic acid and fewer cells are required.
- Avoids biological or chemical toxicity – Since the method is purely physical it avoids the use of viral vectors or chemical reagents. It also eliminates problems such as bacterial overgrowth and antibiotic treatment.
Limitation of Bombardment (Biolistics) Method
- High cost – The bombardment method requires expensive instruments such as gene gun apparatus and vacuum systems. The consumable materials like gold particles and rupture discs are also costly.
- Multiple copy insertion – In this method many copies of the transgene is often integrated into the host genome. This may lead to gene silencing and unstable expression of the introduced gene.
- Cell and tissue damage – The high velocity of the microprojectiles can cause severe injury to the target cells. Large pores may be formed in the cell membrane which sometimes do not close and may result in cell death.
- Genomic rearrangements – The physical impact of particles can cause structural changes in the genome such as deletions or chromosomal rearrangements. This may lead to instability of the genome.
- Lower transformation efficiency – The efficiency of transformation in the biolistic method is generally lower when compared with biological methods such as Agrobacterium mediated transformation.
- Fragmentation of DNA – During the bombardment process the introduced DNA may become damaged or fragmented. Because of this the integrity of the transgene may be lost before proper integration.
- Requirement of extensive optimization – The success of this method depends on several physical and biological parameters such as helium pressure, particle size and target distance. These conditions must be optimized separately for different tissues and species.
Uses of Bombardment (Biolistics) Method
- Transformation of monocot crops – It is widely used for genetic transformation of monocot plants such as wheat, maize, rice and sorghum. These plants are often difficult to transform by Agrobacterium mediated method.
- Organelle transformation – The gene gun can introduce foreign DNA directly into organelles such as chloroplasts and mitochondria. This process is referred to as plastid transformation.
- Genome editing – The method is used for delivering genome editing components such as CRISPR-Cas9 into plant cells. In this process targeted modification of the genome can be achieved.
- Delivery of different biological molecules – It can deliver different types of molecules such as plasmid DNA, linear DNA fragments, RNA and proteins directly into the cells.
- Transient gene expression studies – The bombardment method is used for transient expression analysis. It helps in studying promoter activity and gene function in plant tissues.
- Production of stable transgenic organisms – The technique is used for generation of stable transgenic plants and animals where the foreign gene is integrated into the genome.
- Co-transformation of multiple genes – Several plasmids or genes can be coated on the same microprojectile particle. Thus multiple genes can be introduced into the same cell simultaneously.
- Transformation of animal cells – The gene gun can also be used for introduction of DNA into animal cells, embryos and tissues for research purposes.
- Transformation of microorganisms – It is used for genetic modification of microorganisms such as bacteria, yeast, fungi and algae.
- Viral inoculation in plants – The method is used for introducing viral DNA or RNA into plant tissues during plant pathology studies.
- Advanced technical analysis of biolistic particle bombardment in plant biotechnology: Principles, instrumentation, and procedural optimization. (n.d.).
- Bennur, P. L., O’Brien, M., Fernando, S. C., & Doblin, M. S. (2024). Improving transformation and regeneration efficiency in medicinal plants: insights from other recalcitrant species. Journal of Experimental Botany, 76(1), 52–75. https://doi.org/10.1093/jxb/erae189
- Bio-Rad Laboratories. (n.d.). Biolistic® PDS-1000/He particle delivery system (Instruction Manual).
- Bio-Rad Laboratories. (1997). Biolistic PDS-1000/He particle delivery system (Bulletin 1700).
- Bio-Rad Laboratories. (2006). Biolistic PDS-1000/He™ system (Bulletin 5447).
- Bio-Rad Laboratories. (2007). Biolistic delivery systems (Bulletin 5443).
- Bio-Rad Laboratories. (n.d.). PDS-1000 | He™ system.
- Creative BioMart. (n.d.). Biolistic bombardment nucleic acid delivery system.
Dormatey, R., Sun, C., Ali, K., Fiaz, S., Xu, D., Calderón-Urrea, A., Bi, Z., Zhang, J., & Bai, J. (2020). - ptxD/Phi as alternative selectable marker system for genetic transformation for bio-safety concerns: A review. PeerJ.
- Gallego, A. (n.d.). Deep dive into plant transformation: Protoplast-mediated, biolistic-mediated and Agrobacterium-mediated gene transfer. GoldBio.
- Gunadi, A., et al. (2017). What is the pros and cons of using gene gun versus using agrobacterium for transforming plants? ResearchGate.
- Helenius, E., et al. (2000). Biolistic protocol. Scribd.
- Keshavareddy, G., Kumar, A. R. V., & Ramu, V. S. (2018). Methods of plant transformation- A review. International Journal of Current Microbiology and Applied Sciences, 7(07), 2656–2668.
Li, et al. (n.d.). Overview of PDS-1000/He biolistic particle delivery system equipped with a flow guiding barrel [Figure]. ResearchGate. - OoCities.org. (n.d.). Protocols for biolistic gene transformation (bombardment).
- Ortiz, J. P. A., Reggiardo, M. I., Ravizzini, R. A., Altabe, S. G., Cervigni, G. D. L., Spitteler, M. A., Morata, M. M., Elias, F. E., & Vallejos, R. H. (1996). Hygromycin resistance as an efficient selectable marker for wheat stable transformation. Plant Cell Reports, 15(12), 877–881. https://doi.org/10.1007/BF00231579
- Plant Biotechnology Resource and Outreach Center. (n.d.). Biolistic-mediated gene transfer. Michigan State University.
- Rajkumari, N., Alex, S., Anith, K. N., Viji, M. M., & Kiran, A. G. (2021). Silver nanoparticles for biolistic transformation in Nicotiana tabacum L. 3 Biotech, 11(12), 497. https://doi.org/10.1007/s13205-021-03043-9
- Santin, A., Russo, M. T., Marotta, P., Manfellotto, F., Ruggiero, A., & Ferrante, M. (2022). Biolistic transformation of Pseudo-nitzschia multistriata. protocols.io. https://dx.doi.org/10.17504/protocols.io.b5jgq4jw
- Thorpe, C., Luo, W., Ji, Q., Eggenberger, A. L., Chicowski, A. S., Xu, W., Sandhu, R., Lee, K., Whitham, S. A., Qi, Y., Wang, K., & Jiang, S. (2025). Enhancing biolistic plant transformation and genome editing with a flow guiding barrel. Nature Communications, 16(1), 5624. https://doi.org/10.1038/s41467-025-60761-x
- Wang, Y., Zeng, J., Su, P., Zhao, H., Li, L., Xie, X., Zhang, Q., Wu, Y., Wang, R., Yang, G., He, G., & Li, Y. (2022). An established protocol for generating transgenic wheat for wheat functional genomics via particle bombardment. Frontiers in Plant Science, 13, 979540. https://doi.org/10.3389/fpls.2022.979540
- Zhao, Y., Gao, J., Huang, L., Zhang, Y., & Yang, Y. (2023). Rapid and efficient optimization method for a genetic transformation system of medicinal plants Erigeron breviscapus. International Journal of Molecular Sciences, 24(6), 5611. https://doi.org/10.3390/ijms24065611