What are Volvox?
The taxonomic genus Volvox, a member of the family Vovlocacae, constitutes a distinctive assemblage of green algae that manifests itself as a singular spherical colony composed of a remarkable aggregation of over 50,000 cells. The ubiquity of Volvox within freshwater ecosystems, encompassing diverse habitats such as ponds, puddles, and ditches, accentuates its ecological relevance. Intriguingly, despite its minuscule dimensions, Volvox attains a diameter of up to 500 micrometers at full maturation, facilitating its discernment by the unaided human eye.
Algae, particularly of the microscopic variety, pose an enigma to scientific inquiry, inviting contemplation upon the intricate influence wielded by diminutive organisms upon the macroscopic world. This expositional endeavor aspires to unravel the profundities embedded within the verdant algal realm, focusing upon the botanical entity recognized as Volvox.
The taxonomic characterization of Volvox encompasses approximately 20 distinct species, each engendering a unique ensemble of morphological attributes. Among these, several prominent species, alongside their defining features, warrant elucidation:
- Volvox aureus: This diminutive iteration of Volvox evinces a reduction in cellular count vis-à-vis conventional counterparts. Measuring a mere 0.5 millimeters in diameter, Volvox aureus assumes a threshold of visibility for the unassisted human eye. Notably, the cellular constituents of this species are endowed with an ovoid configuration.
- Volvox globator: Distinguished by its more expansive dimensions, Volvox globator achieves a diameter of up to 2 millimeters, rendering it observable without the aid of microscopic instrumentation. Despite its larger overall size, the individual cellular components of Volvox globator are characterized by a diminutive scale, measuring a mere four microns in diameter.
- Volvox carteri: Eponymously titled in honor of its discoverer, Volvox carteri has garnered scientific acclaim as a pivotal model organism, serving as an indispensable tool to fathom evolutionary dynamics and cellular differentiation. A salient attribute of this species lies in its reduced cellular diversity, comprising solely two distinct cell types. The somatic cells, equipped with biflagellated motility but devoid of reproductive capability, juxtapose against the immobile and seemingly immortal Gonidia, lauded for their prolific capacity to undergo division and reproduction.
- Volvox barberi: Of note, Volvox barberi stands apart as an exemplar of rapid motility, outstripping its conspecifics in terms of speed. This species boasts an impressive cellular count, with colony populations ranging from 10,000 to 50,000 cells. Intriguingly, Volvox barberi demonstrates a paradoxical negative buoyancy, compelling incessant locomotion to forestall submergence. Its swiftness attains a zenith of 3 mph, a feat remarkable within the algal realm.
Conversely, Volvox’s historical and contemporary utility as a model organism rests upon its unparalleled reproductive dynamics and exceptional locomotor agility. Pertinently, the colonial proclivity of Volvox cells assumes paramount significance, postulated to elucidate the evolutionary transition from unicellular precursors to multicellular entities. The encapsulated discourse, however, encapsulates merely a fraction of the taxonomic diversity inherent to the genus Volvox, with myriad species awaiting comprehensive scientific scrutiny.
In summation, Volvox emerges as an intricate botanical entity, instigating reflections upon the convergence of cellular complexity and ecological impact within the ambit of green algae. The illuminated species profiles, albeit select, serve as conduits for delving into the arcane intricacies underpinning Volvox’s enigmatic realm, as well as instilling a sense of anticipation for future explorations that shall further demystify the myriad forms and functions inherent to this captivating genus.
Structure of Volvox
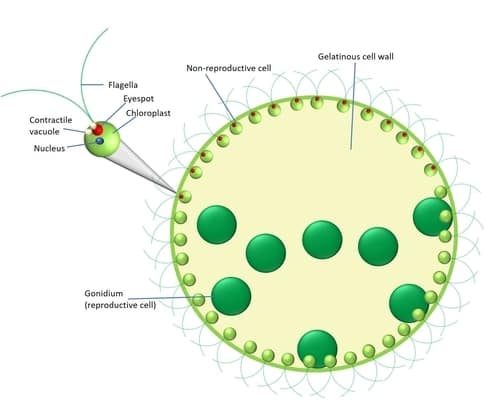
The elucidation of Volvox’s intricate structure is indispensable to unraveling the physiological underpinnings of its remarkable behavior. The visual representation provided above offers an illuminating conduit to apprehend the myriad constituents comprising Volvox’s architecture, each endowed with specialized functions that collectively orchestrate the organism’s locomotion, orientation, and response to environmental stimuli.
- Flagella: These slender, hair-like protrusions, emerging from the cellular body, serve as the hallmark of Volvox’s dynamic propulsion. Beyond mere locomotion, flagella exhibit a versatility that encompasses sensory perception and tactile interactions. In Volvox, a symmetrical assemblage of two flagella embellishes the anterior of the colonial structure. Notably, their inversion during colony inversion endows the colonial aggregate with a synchronized means of aquatic mobility, enabling harmonious phototactic movement toward solar sources. This choreographed maneuvering, an object of extensive contemporary inquiry, exemplifies an intricate manifestation of emergent collective behavior.
- Contractile Vacuole: Operating as a sentinel of osmotic equilibrium, the contractile vacuole assumes a pivotal role in the maintenance of intracellular fluid dynamics. While endowed with locomotory functions in certain biological contexts, in Volvox, this organelle assumes a vital role in osmoregulation, sustaining cellular rigidity to counteract the hydrostatic pressures imparted by aquatic submersion. The biomechanical resilience thus conferred contributes substantively to Volvox’s buoyancy and structural integrity.
- Eyespot: Situated strategically on the anterior pole, the enigmatic eyespot serves as Volvox’s rudimentary photoreceptor. While divergent in form from conventional ocular organs, this rudimentary light-sensing apparatus embodies the capacity to discern the directionality of photosynthetic light, facilitating precise navigation toward luminous sources. The absence of this sensorial apparatus would impede Volvox’s capacity to orchestrate its trajectory toward optimal light exposure.
- Nucleus: A central repository of genetic information, the eukaryotic nucleus imparts structural sophistication to Volvox’s cellular constitution. Encompassing hexagonal DNA strands that encode the organism’s genetic blueprint, the nucleus underscores the cellular potential for both sexual and asexual propagation, serving as an evolutionary crucible for genetic exchange and diversification.
- Chloroplast: The pivotal organelle of photosynthetic conversion, the chloroplast assumes eminence in Volvox’s energy acquisition and metabolic sustenance. Laden with chlorophyll pigments, the verdant chloroplast captures solar irradiance, culminating in ATP and NADPH synthesis while expelling oxygen into the surrounding milieu. The chloroplast’s historical significance, owing to its potential ancient origin as an independent cellular entity, underscores the intricate tapestry of cellular evolution.
- Gelatinous Cell Wall: A robust gelatinous barrier, formed by bi-flagellated somatic cells, envelops Volvox’s colony. This pliant yet structurally supportive layer confers both cohesion and flexibility, enabling coordinated organelle movement while safeguarding the organism’s integrity amidst fluidic environments.
- Gonidium: The asexual congregations of cells within Volvox’s colonial matrix, termed gonidia, present an intriguing tableau of reproductive prowess. Nurtured by ceaseless divisions, these immobile entities herald the genesis of nascent colonies, a testament to Volvox’s inherent adaptability and propensity for expansion.
- Cytoplasm: The intricate strands of cytoplasm, a structural conduit that unites individual cells, form the fabric of Volvox’s colonial entity. This elaborate network not only facilitates collective movement but also serves as a conduit for intercellular communication, fostering a seamless orchestration of the organism’s activities.
In summary, the structural tableau of Volvox stands as a testament to the intricate interplay of specialized cellular components, converging harmoniously to underpin its locomotion, light responsiveness, and reproductive proclivity. This holistic understanding of Volvox’s anatomy enriches our comprehension of the dynamic interplay between form and function within this singular botanical entity, transcending the conventional boundaries of conventional algal studies.
Requirements for observing Volvox under a microscope
The pursuit of microscopic inquiry into the intricate morphology and behavior of Volvox mandates a discerning selection of apparatus, each meticulously calibrated to fulfill the exigencies of observation at the cellular scale. The orchestrated amalgamation of requisite equipment lends itself to an expeditious and elucidatory exploration of Volvox, enabling the elucidation of its enigmatic characteristics through the prism of high-resolution microscopy.
- Microscope with Minimum 400x Magnification: The cornerstone of this investigatory endeavor resides within the choice of microscope. A microscope of no less than 400x magnification stands as a cardinal prerequisite, thereby enabling the resolution of intricate cellular structures and behavioral nuances inherent to the Volvox genus. This optical instrument serves as the conduit through which the otherwise diminutive and subvisible constituents of Volvox emerge into a realm of discernibility, facilitating profound insights into its anatomical intricacies and dynamic manifestations.
- Slide and Coverslip: Central to the microscopic examination of Volvox is the utilization of a slide and coverslip, constituting a foundational platform for immobilizing and safeguarding the biological specimen under scrutiny. These essentials collectively establish an environment conducive to meticulous focusing and sustained observation, ensuring the preservation of Volvox’s integrity while facilitating an unimpeded line of sight to the minutiae of its architecture.
- Water Sample Containing Volvox: The procurement of an appropriate aqueous medium encapsulating live Volvox specimens remains an imperative precondition. A judiciously acquired drop of pond water or lake water, harboring these algal entities, becomes a canvas upon which the symphony of Volvox’s microcosmic existence unfolds. This contextual milieu provides a receptive setting for the sustenance of Volvox and, by extension, facilitates its preservation and visual accessibility throughout the course of microscopic examination.
- Pipette or Dropper: The seamless transposition of the aquatic medium onto the slide necessitates the enlistment of a pipette or dropper. This utensil, effectuating a measured and judicious transfer of the water housing Volvox, not only preserves the integrity of the biological sample but also ensures a controlled dissemination of the organisms across the microscopic slide. The judicious application of this instrument fosters an unobtrusive deposition of Volvox, obviating disturbances that could impede subsequent microscopic analysis.
- Adequate Illumination: The pivotal role of illumination in microscopic exploration cannot be understated. A potent and luminous light source, tailored to the specific requirements of microscopic analysis, serves as the fulcrum around which the spectacle of Volvox unfolds. It engenders a heightened contrast and accentuated visual acuity, catalyzing the revelation of intricate cellular components and dynamic behaviors. This illuminative synergy, between specimen and light, orchestrates an immersive voyage into Volvox’s microcosmic landscape, unraveling its multifarious dimensions with unprecedented clarity.
In summation, the coherent assembly of these requisites—comprising an aptly potent microscope, strategic usage of slides and coverslips, a judiciously selected water sample with Volvox, a precision instrument for transferring the medium, and a proficient light source—composes a meticulously choreographed symphony of equipment, underpinning the scholarly pursuit of Volvox through the lens of microscopy. This orchestration not only attests to the meticulous preparations inherent to microscopic inquiry but also reaffirms the paramount significance of these pragmatic tools in fostering an enhanced comprehension of Volvox’s microscopic tapestry.
Procedure for Sample collection of Volvox
The acquisition of a representative and viable sample of Volvox for subsequent microscopic analysis mandates a systematic and meticulous approach, ensuring the preservation of the algal organism’s integrity and vitality. The ensuing procedure delineates a step-by-step guide to the collection of Volvox specimens, thereby facilitating an illuminating exploration of their intricate morphological and behavioral attributes.
- Equipment Preparation: Gather the necessary equipment, including a collection container (such as a clear glass jar or a plastic container), a fine mesh net or sieve, a small bucket or vessel for water collection, and a hand-held magnifying lens or binoculars for preliminary identification of potential Volvox habitats.
- Habitat Selection: Locate a suitable freshwater habitat, such as a pond, lake, or slow-moving stream, known to harbor Volvox populations. Environments rich in aquatic vegetation and exposed to sunlight are particularly conducive to Volvox growth.
- Visual Inspection: Use the magnifying lens or binoculars to scan the water surface and vegetation for signs of green spherical colonies resembling Volvox. Observe for any aggregations that may indicate the presence of Volvox.
- Water Collection: Submerge the fine mesh net or sieve into the water and gently sweep it through aquatic vegetation, being cautious not to disrupt the substrate. This action aids in entrapping Volvox colonies within the mesh.
- Transfer to Collection Container: Lift the mesh net or sieve out of the water and carefully examine its contents. If Volvox colonies are visible, transfer them into the small bucket or vessel filled with a small amount of the collected water. Gently agitate the water to ensure the release of Volvox from the mesh.
- Sample Preservation: Transfer the collected water containing Volvox into a clear glass jar or plastic container, ensuring that sufficient water volume is retained to sustain the organisms during transportation to the observation site. Seal the container securely to prevent spillage or contamination.
- Transport and Storage: Store the container in a cool, shaded place during transportation to the observation site. It is advisable to conduct the microscopic analysis as soon as possible to ensure the viability and vitality of the Volvox specimens.
- Microscopic Observation: Upon reaching the observation site, carefully transfer a small amount of the collected water containing Volvox onto a clean glass microscope slide. Add a coverslip to create a stable microscopic sample.
- Microscopy Setup: Place the prepared slide on the microscope stage and adjust the focus and illumination settings for optimal visualization. Begin with lower magnifications to locate and focus on Volvox colonies before transitioning to higher magnifications for detailed observation.
- Observation and Documentation: Systematically observe the Volvox colonies under the microscope, noting their structural characteristics, locomotion, and any other behaviors of interest. Utilize the microscope’s features to capture images or videos for documentation and further analysis.
Procedure for Sample Preparation
- Collection of Specimens: Retrieve a viable sample of Volvox colonies as outlined in the previous section on sample collection. Ensure that the sample is acquired from a suitable freshwater habitat, and that the organisms are healthy and representative of the population.
- Equipment Setup: Assemble the necessary equipment for sample preparation, including a clean glass microscope slide, a coverslip, a dropper or pipette, and a source of illumination, such as a microscope with appropriate magnification settings.
- Droplet Placement: Using a dropper or pipette, carefully place a small droplet of the collected water containing Volvox onto the center of the microscope slide. Ensure that the droplet is of a size that allows for easy placement of the coverslip without excess spillage.
- Applying the Coverslip: Gently lower one edge of the coverslip onto the water droplet at a slight angle. Gradually lower the coverslip until it makes contact with the droplet and the sample. This method reduces the likelihood of air bubbles being trapped beneath the coverslip.
- Minimizing Air Bubbles: While maintaining a slight angle, slowly lower the rest of the coverslip onto the slide, allowing the droplet to spread and displace excess water and air. This gradual approach helps minimize the formation of air bubbles, which can obstruct clear observation.
- Uniform Dispersion: Tap the coverslip gently with a blunt object (such as the handle of a pipette) to encourage the uniform dispersion of Volvox colonies within the water droplet. Take care to avoid excessive force that could damage the organisms or alter their arrangement.
- Excess Water Removal: If excess water accumulates around the edges of the coverslip, carefully blot it with a piece of absorbent paper or a lint-free tissue. The objective is to retain an adequate but not overwhelming amount of liquid for observation.
- Microscope Preparation: Place the prepared slide on the microscope stage and secure it in place. Adjust the focus, magnification, and illumination settings on the microscope to facilitate clear visualization of the Volvox specimens.
- Microscopic Observation: Systematically explore the slide using the microscope’s controls, starting with lower magnifications to locate and focus on Volvox colonies. Gradually increase the magnification to observe finer details of the individual cells, flagella, and any other relevant features.
- Documentation and Analysis: Capture images or videos of the observed Volvox specimens using the microscope’s built-in camera or an external imaging device. Record relevant information, such as colony size, cell arrangement, and any observed behaviors.
Observation of Volvox Under Microscope
- Microscope Setup: Initiate the observation process by ensuring the microscope is appropriately configured. Calibrate the magnification settings based on the expected size of Volvox colonies, typically starting with lower magnifications and gradually increasing as necessary. Fine-tune the illumination source to optimize contrast and clarity.
- Slide Placement: Position the prepared microscope slide, containing the Volvox sample immobilized beneath the coverslip, onto the microscope stage. Secure the slide in place using the stage clips or slide holder, ensuring stability for unhindered manipulation.
- Initial Scanning: Begin the observation by scanning the slide at low magnification to locate Volvox colonies. Focus the microscope on areas of interest, such as clusters of spherical structures, while systematically traversing the slide to ensure comprehensive coverage.
- Identifying Structural Features: Gradually increase the magnification to reveal finer details of Volvox morphology. Observe the spherical colonies, noting attributes such as colony size, arrangement of individual cells, and the presence of flagella at the anterior.
- Focus and Clarity: Employ the microscope’s focusing mechanisms to attain optimal clarity. Pay attention to the position of the specimen within the z-axis to ensure that the entire depth of the colony is in focus.
- Flagellar Dynamics: Direct attention to the flagella, the whip-like appendages responsible for Volvox locomotion. Observe their movement and coordination, which enable the colony’s phototactic response toward light sources.
- Chloroplasts and Cellular Detail: Zoom in further to investigate individual cells within the colony. Focus on cellular components such as chloroplasts, discernible by their green pigmentation, and nucleus, which houses genetic material.
- Phototactic Behavior: Examine the positioning of the eyespot, a rudimentary photoreceptor, and observe how Volvox orients itself toward light. Document the colony’s directional adjustments in response to changes in light source angles.
- Reproductive Structures: Look for specialized structures such as gonidia, which play a role in asexual reproduction. Document their arrangement and any observed reproductive activities, such as cell divisions.
- Documentation: Capture high-quality images or videos of the observed Volvox colonies using the microscope’s imaging capabilities. Ensure that the images accurately portray the structural features and behaviors under scrutiny.
- Analysis and Interpretation: Analyze the captured data, taking into account the observed characteristics, interactions, and dynamics of Volvox. Compare your observations with existing literature to contextualize your findings within the broader scientific understanding.
- Record Keeping: Maintain detailed records of your observations, including notes, images, and annotations. These records contribute to the scientific record and can serve as a foundation for further research or analysis.
Observation of Volvox Under Microscope Pictures
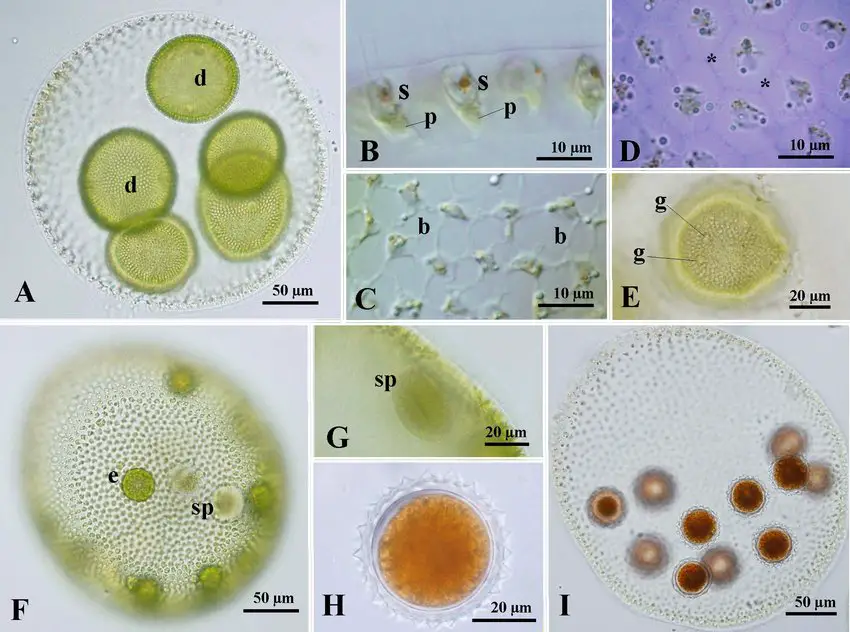

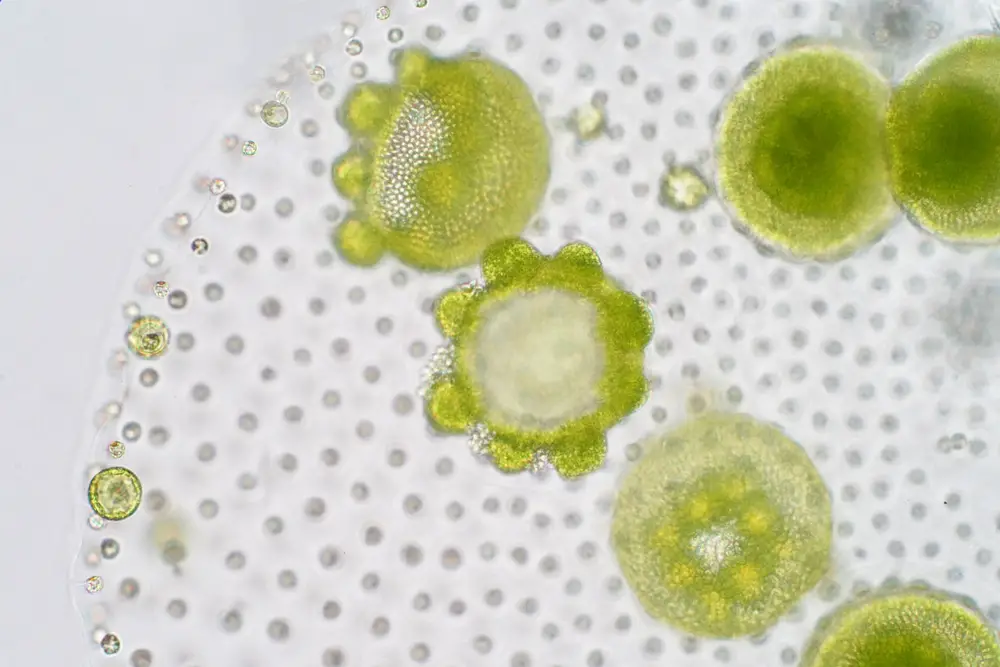
Observation of Volvox Under Microscope Video
FAQ
What is Volvox?
Volvox is a genus of green algae that forms spherical colonies composed of numerous individual cells. These colonies exhibit coordinated movement and are often found in freshwater habitats such as ponds and lakes.
Why is observing Volvox under a microscope important?
Microscopic observation allows us to study the intricate cellular structure, behavior, and interactions within Volvox colonies. This insight contributes to our understanding of evolutionary processes, cell differentiation, and collective behavior.
What microscope magnification is recommended for observing Volvox?
A microscope with at least 400x magnification is recommended to observe Volvox colonies in detail. This level of magnification enables clear visualization of individual cells, flagella, and other structural features.
How should I prepare a slide for Volvox observation?
Place a small droplet of collected water containing Volvox on a clean microscope slide. Gently lower a coverslip onto the droplet at a slight angle, gradually allowing it to settle and minimize air bubble formation.
What features should I look for when observing Volvox under the microscope?
Focus on the spherical colony structure, arrangement of individual cells, presence of flagella (whip-like appendages), chloroplasts (green pigmented structures), and any observable behaviors such as phototactic movement.
What is phototactic movement in Volvox?
Phototactic movement refers to the colony’s ability to move towards a light source, facilitated by the presence of an eyespot. Observe how the colony adjusts its orientation in response to changes in the direction of light.
What are gonidia in Volvox colonies?
Gonidia are specialized groups of cells within Volvox colonies that play a role in asexual reproduction. They eventually give rise to new colonies through cell division.
How can I document my observations of Volvox under the microscope?
Use the microscope’s imaging capabilities to capture images or videos of the observed features and behaviors. Take detailed notes, record magnification settings, and annotate your images to accurately document your findings.
What can studying Volvox reveal about evolutionary processes?
Volvox colonies provide insights into the transition from unicellular to multicellular life forms. The coordination among cells in the colony can offer clues about the early stages of multicellular organization.
Can I culture Volvox for long-term observations?
Yes, Volvox can be cultured in controlled laboratory settings for prolonged observations. By providing suitable growth conditions and nutrients, researchers can maintain and study Volvox colonies over extended periods.
How does Volvox reproduction occur, and can I observe it under the microscope?
Volvox reproduces both sexually and asexually. While asexual reproduction (through gonidia) is observable under the microscope, sexual reproduction involves specialized structures that may require more advanced techniques for observation.
What are the potential applications of studying Volvox under the microscope?
Studying Volvox can contribute to fields such as evolutionary biology, cell biology, ecology, and even robotics. Understanding collective behaviors and multicellularity in Volvox may have broader implications for understanding similar processes in other organisms.