Seliwanoff’s Test is a biochemical colour reaction that is used mainly to distinguish between ketose sugar and aldose sugar. It is performed by using Seliwanoff’s reagent (resorcinol + concentrated hydrochloric acid) and the sugar solution is heated with this reagent. In this test the acid act as dehydrating agent and the reaction is based on the fact that ketoses are dehydrated more rapidly than aldoses.
In this process ketose (example-fructose) is dehydrated quickly and furfural derivative is formed. This furfural derivative is then condensed with resorcinol and a deep cherry-red colour is produced within 1–2 minutes and this indicates positive test. In case of aldose (example-glucose) the reaction occurs slowly and within same short time it gives faint pink or light yellow colour or remains colourless and this indicates negative test.
Objectives of Seliwanoff’s test
- To detect the presence of ketohexoses (ketose sugars) in the given sample.
- To distinguish between ketoses and aldoses sugars. It is commonly used to differentiate a keto sugar like fructose from aldose sugars like glucose and galactose.
Principle of Seliwanoff’s test
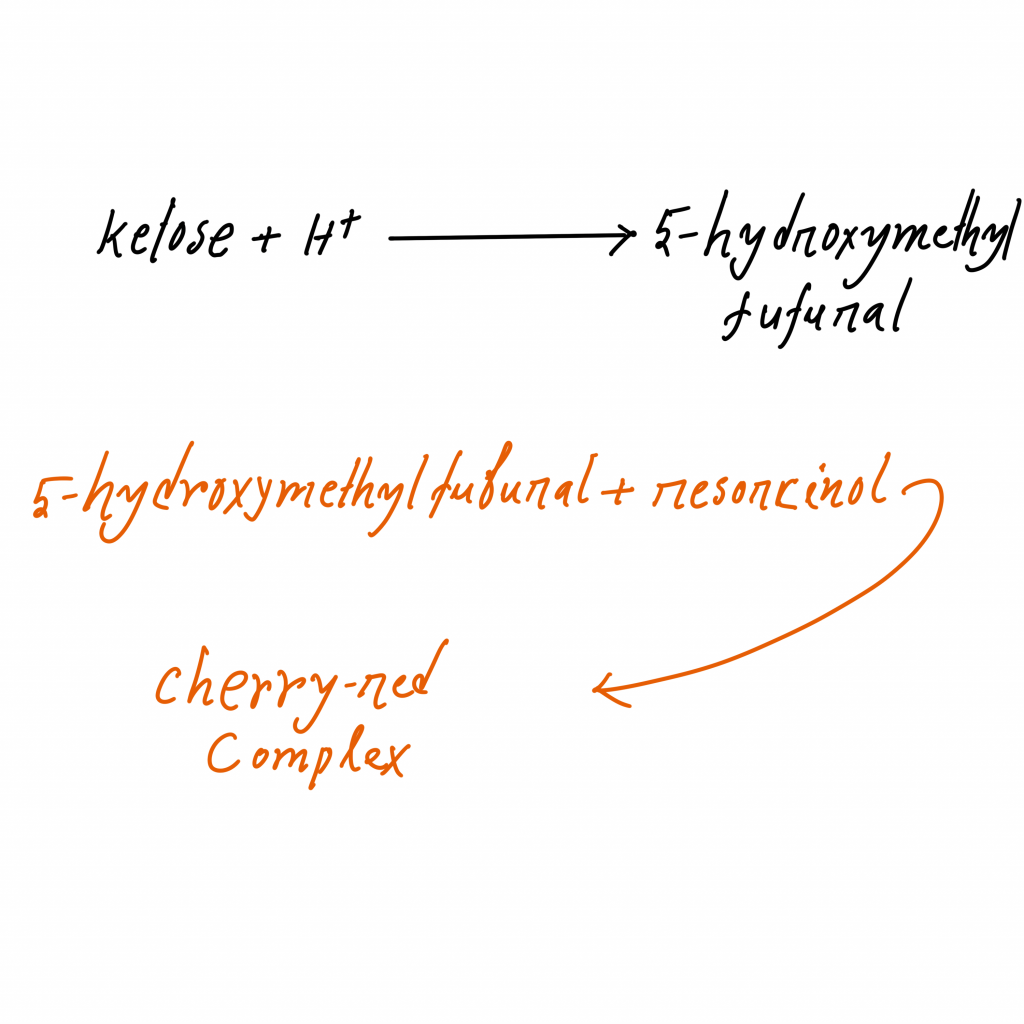
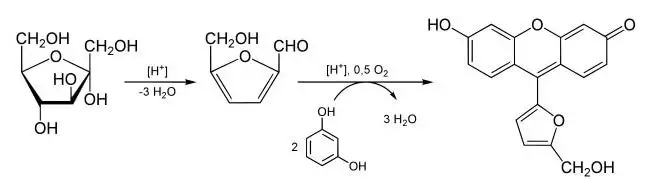
The principle of Seliwanoff’s test is based on the kinetic difference between ketose and aldose sugars when they are heated with acid. It is performed by using Seliwanoff’s reagent which contains concentrated hydrochloric acid and resorcinol (phenolic compound). In this test the hydrochloric acid rapidly dehydrates ketose sugars and an intermediate compound is formed which is known as 5-hydroxymethylfurfural.
This intermediate then undergoes condensation reaction with two equivalents of resorcinol and a deep cherry-red coloured complex (xanthenoid) is produced within 1–2 minutes and it indicates positive test for ketose. Aldoses are dehydrated very slowly in same condition so within same short time it gives faint pink or light yellow colour or remains colourless and it indicates negative test.
Requirements for Seliwanoff’s test
- Seliwanoff’s reagent (resorcinol + concentrated hydrochloric acid (HCl) + distilled water).
- Test samples (carbohydrate solutions such as fructose sucrose or glucose) usually prepared in 0.5% to 5% concentration.
- Distilled water (it is used as negative control or blank).
- Clean and dry test tubes.
- Test tube stand.
- Pipette or dropper for measuring and transferring solutions.
- Water bath (boiling at 100°C) for heating the test tubes and to initiate the dehydration reaction.
Procedure of Seliwanoff’s test
- Take clean and dry test tubes. Add the sample solution (about 1 ml or 10 drops) in one test tube. Add equal amount of distilled water in another test tube and it is used as negative control (blank).
- Add Seliwanoff’s reagent (about 2 ml to 3 ml) in each test tube. It is mixed properly.
- Keep the test tubes in boiling water bath (100°C) for exactly 1 to 2 minutes. This step is timed strictly and over boiling is avoided because prolonged heating can convert aldose into ketose and false positive result is obtained.
- Remove the test tubes from water bath and allow to cool at room temperature.
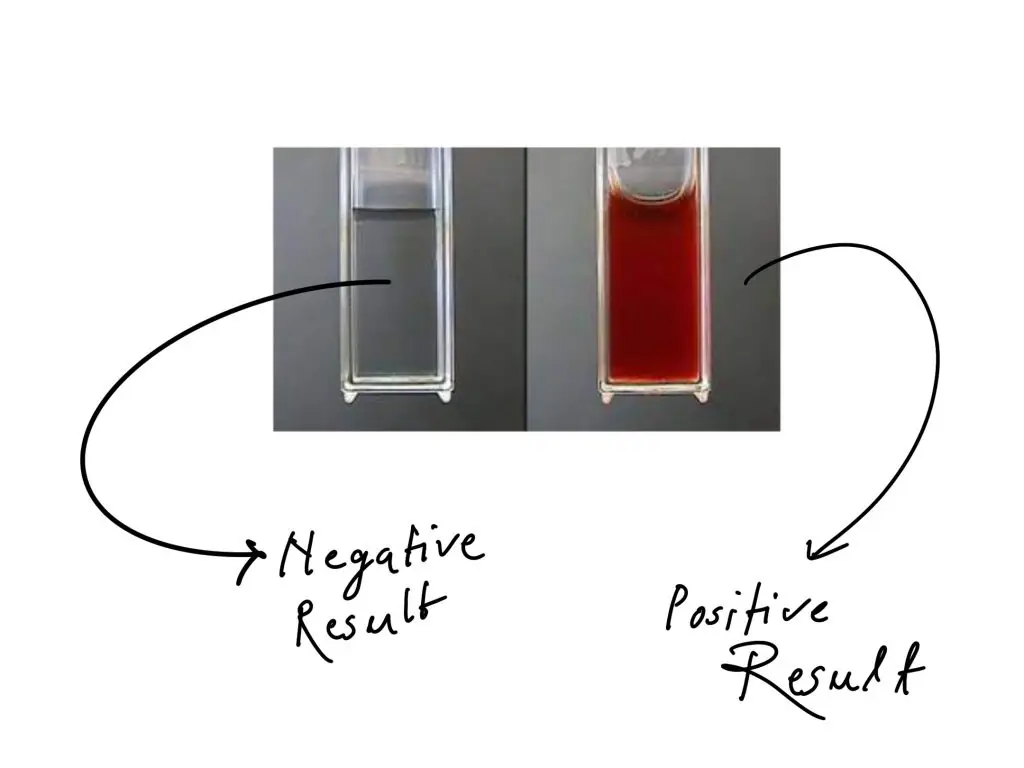
- Observe the colour formation. Deep cherry-red colour within 1–2 minutes indicates positive test for ketose. Faint pink or yellow colour or no colour change indicates negative test (aldose sugar).
Results of Seliwanoff’s test
Positive result (Ketohexoses)– It is indicated by rapid formation of deep cherry-red or burgundy colour within 1–2 minutes. It confirms the presence of ketose sugars such as fructose or sucrose.
Negative result (Aldoses)– The solution remains clear or it develops faint pink or light yellow colour slowly. It indicates the presence of aldose sugars such as glucose or galactose.
Positive result (Ketopentoses)– The solution develops bluish-green colour.
False positive result– If the solution is boiled for prolonged time the aldoses can isomerize into ketoses and it may produce cherry-red colour and inaccurate positive reading is obtained.
Uses of Seliwanoff’s test
- It is used for distinguishing between sugar types. It is mainly used to differentiate ketoses (ketone group sugar like fructose) from aldoses (aldehyde group sugar like glucose).
- It is used in medical diagnosis. It is used to detect presence of fructose in urine and it helps in diagnosing fructosuria and hereditary fructose intolerance.
- It is used in food quality control and adulteration. The test (and modified test) is used to quantify 5-Hydroxymethylfurfural (HMF) levels in honey syrups and balsamic vinegar to check quality excessive heating or adulteration.
- It is used for juice authenticity. It provides rapid method to verify nutritional value and authenticity of fruit juices by detecting natural fructose level.
- It is utilized in dairy industry analysis. It is used for rapid determination of lactulose in milk and it helps to distinguish between UHT (Ultra-High Temperature) and sterilized milk.
- It is used in industrial fermentation. It is used to determine fructose concentration colorimetrically and to monitor depletion of fructose in fermentation media.
- It is used in pharmaceutical applications. It is employed to detect ketoses used as stabilizing excipients or active ingredients in medications and product safety and effectiveness is ensured.
- It is used in high-throughput screening (HTS). It is adapted for screening high activity isomerase enzyme variants (example-L-rhamnose isomerase) by monitoring reduction of ketoses.
- It is used in prebiotic analysis. It can be used to measure encapsulation efficiency of prebiotics such as fructooligosaccharides (FOS) by analysing unentrapped components.
- It is used in herbal formulation testing. It is used to analyse derivatized inulin (fructose polymer) applied in herbal formulations.
Limitations of Seliwanoff’s test
- False positive result can be obtained due to prolonged boiling. If the solution is boiled for more than 2 minutes aldoses like glucose can be converted into ketoses in acid condition and cherry-red colour is produced.
- High sugar concentration can interfere in this test. High concentration of aldoses (more than 2%) can produce enough intermediate compounds and it may mimic positive red colour within standard time.
- It has lack of individual ketose specificity. It is a general qualitative test for ketoses and it cannot differentiate between specific ketose sugars (fructose vs sorbose) so secondary tests like Osazone test is required.
- Strict acid concentration is required. If hydrochloric acid concentration is too high (more than 12%) the selectivity is lost and aldoses can react rapidly and immediate false positive is obtained.
- At low detection limit the result becomes subjective. The test can detect ketose at very low concentration (example-0.015%) but the faint colour change is difficult to interpret reliably.

Precautions of Seliwanoff’s test
- Prolonged heating is avoided. The heating time is maintained strictly (1 to 2 minutes) because boiling for long time can convert aldose sugar (example-glucose) into ketose and false positive is obtained.
- High concentration of sugar sample is avoided. If aldose concentration is more than 2% it can interfere and it may mimic positive result so sample is diluted (about 0.5% to 1%).
- Concentration of hydrochloric acid (HCl) in reagent is maintained properly. It is kept below 12% (generally 3N to 6N) because high acid concentration can force aldoses to react rapidly and immediate false positive is obtained.
- After heating the test tubes are removed and gradual cooling is allowed at room temperature.
- Seliwanoff’s reagent is stored properly. It is kept in amber bottle to prevent photo-oxidation of resorcinol and dark coloured impurities is avoided. Fresh reagent is prepared after 2 to 3 months for maintaining sensitivity.
References
- A Level Biology. (n.d.). Tests for carbohydrates | Types, principles, apparatus.
- Al-Rasheed University College. (n.d.). Biochemistry laboratory Lab.2.
- Al-Sharifi, N. (n.d.). Seliwanoff’s test and Bial’s test [PowerPoint slides]. Al-Rasheed Pharmacy Department.
- Beşir Özgeçen, A., Yazici, F., Mortaş, M., & Gul, O. (2020). A novel spectrophotometric method based on Seliwanoff test to determine 5-(Hydroxymethyl) furfural (HMF) in honey: Development, in house validation and application. LWT – Food Science and Technology, 139, Article 110602. https://doi.org/10.1016/j.lwt.2020.110602
- Ghazi Mohammed, N. U. (2023). Biochemistry lab (For biology students/first class). University of Baghdad College of Science for Women, Department of Chemistry.
- Khan, S. (2025, August 11). Lab 9 – Tests for carbohydrates. Chemistry LibreTexts.
- King Saud University. (n.d.). Color tests for specific carbohydrates: Ketoses, pentoses, and polysaccharides (CLS 281 Basic Biochemistry and Biomolecules).
- Mwafi, N. (n.d.). Qualitative tests for carbohydrates [PowerPoint slides]. Faculty of Medicine, Mutah University.
- Roe, J. H., & Papadopoulos, N. M. (1954). The determination of fructose-6-phosphate and fructose-1,6-diphosphate. Journal of Biological Chemistry, 210(2), 703-707. https://doi.org/10.1016/S0021-9258(18)65397-9
- Sánchez-Viesca, F., & Gómez, R. (2018). Reactivities involved in the Seliwanoff reaction. Modern Chemistry, 6(1), 1-5. https://doi.org/10.11648/j.mc.20180601.11
- Sapkota, A. (2022, September 5). Seliwanoff’s test- Definition, principle, procedure, result, uses. Microbe Notes.
- Technical analysis of Seliwanoff’s colorimetric assay: Principles, molecular dynamics, and diagnostic utility in glycobiology. (n.d.).
- Thomas, J. A., Mawhinney, M., & Mason, W. (1968). Sex accessory fructose: An evaluation of biochemical techniques. Proceedings of the Society for Experimental Biology and Medicine, 127(3), 930-934.
- UKEssays. (2015, January 1). Qualitative tests used for carbohydrates biology essay.
- University of Life Sciences in Lublin. (n.d.). Sugars (identification of unknown saccharide). Department of Biochemistry.
- Vedantu. (n.d.). What is Seliwanoffs test class 11 chemistry CBSE.
- Wikipedia contributors. (2025, November 29). Seliwanoff’s test. In Wikipedia, The Free Encyclopedia.