Carbohydrate Fermentation Test is a biochemical test that is used to determine whether a specific microorganism (mainly bacteria) is capable of fermenting a particular carbohydrate such as glucose lactose or sucrose. It is used because different bacterial species possess different enzyme system and thus they vary in their ability to use specific sugar for energy. This variation in carbohydrate metabolism is important for differentiating and identifying bacterial groups especially Enterobacteriaceae.
This test is performed using a liquid nutrient broth that contains a single carbohydrate source and a pH indicator most commonly phenol red. When the bacterium ferments the available sugar it is anaerobically catabolized and organic acids are released as metabolic byproducts. The accumulation of acids lowers the pH of the medium and phenol red changes its colour from red-orange to yellow. This is referred to as a positive fermentation result.
If the organism cannot ferment the carbohydrate it will instead break down the peptones in broth producing alkaline byproducts such as ammonia. Due to this the broth remains red or it turns deeper pink or magenta colour. This indicates a negative result.
In this test gas production is also observed during fermentation. The gases such as carbon dioxide (CO2) and hydrogen (H2) is produced by some bacteria. To detect gas a small inverted glass vial called Durham tube is placed inside the medium. If gas is generated it displaces the liquid and a visible bubble is trapped at the top of the Durham tube.
Based on final observation the result can be categorized into three patterns. Fermentation with acid production only. Fermentation with both acid and gas production. No fermentation.
Objectives of Carbohydrate Fermentation Test
- To determine if a specific bacteria has the ability to ferment a particular carbohydrate.
- To evaluate the fermentation reactions of pure culture of microorganism by observing production of acid and or gas as end products.
- To distinguish between different bacterial groups or species based on their carbohydrate fermentation pattern.
- To help in presumptive identification and characterization of bacterial species based on types of sugar they are able to metabolize.
Principle of Carbohydrate Fermentation Test
Carbohydrate fermentation test works on the principle that different bacteria have different ability to use and ferment a specific carbohydrate for energy production. It is due to presence of different enzyme system in different bacterial species. When a microorganism ferments the sugar present in the liquid medium organic acids are produced as metabolic byproducts.
These acids gets accumulated in the broth and lowers the pH of the medium. This change in pH is detected by a colour change in a pH indicator that is added in the medium such as phenol red bromocresol purple or Andrade’s indicator. For example phenol red changes from red-orange at neutral pH to yellow colour in acidic condition. This indicates acid production and thus positive fermentation.
In some bacteria along with acid production gas is also produced during fermentation. The gases are mainly carbon dioxide (CO2) and hydrogen (H2). To detect gas production a small inverted glass tube known as Durham tube is kept submerged in the broth. If gas is produced it displaces the liquid and a visible air bubble is trapped inside the Durham tube.
Thus by observing acid production and gas production pattern with different sugars the bacteria can be differentiated and identified based on their metabolic capability.
Requirement for Carbohydrate Fermentation Test
- Culture media– A liquid nutrient broth (Phenol red broth base) containing peptones (pancreatic digest of casein or beef extract) and sodium chloride to maintain osmotic balance.
- Carbohydrates– A specific fermentable sugar is added in the broth such as glucose lactose sucrose maltose or mannitol.
- pH indicator– A chemical reagent is used to detect change in pH due to acid production. Phenol red is most commonly used. Other indicators are Andrade’s indicator bromocresol purple and bromothymol blue.
- Durham tube– A small inverted glass vial is placed inside the test tube and fully submerged to detect gas production (CO2 and H2) during fermentation.
- Laboratory equipment– Test tubes inoculating loop or needle autoclave for sterilization and incubator to maintain temperature (35–37°C) for bacterial growth.
- Test organisms– A pure fresh culture of bacteria or yeast is required usually from 18 to 24 hour old culture.
Culture Media composition for Carbohydrate Fermentation Test (per 1 Liter)
- Peptone (Trypticase or proteose peptone or casein peptone)– 10.0 g.
- Sodium chloride (NaCl)– 5.0 g.
- Beef extract (optional)– 1.0 g.
- pH indicator– Phenol red 0.018 g or Bromocresol purple 0.100 g.
- Specific carbohydrate (glucose lactose sucrose etc.)– 5.0 to 10.0 g.
- Final pH– adjusted to about 7.4 ± 0.2 at 25°C.
Preparation of culture media
- Peptone sodium chloride pH indicator and beef extract (if used) are mixed in distilled or deionized water. Gentle heating is done if required for complete dissolution.
- The specific single carbohydrate is added in the broth and it is mixed well until it gets dissolved.
- About 4 to 7 mL of the prepared broth is dispensed into each test tube.
- A small Durham tube is placed inside each tube in inverted position and it is kept fully submerged. It is ensured that no air bubble is trapped inside the Durham tube.
- The tubes are loosely capped or cotton plugged to allow gas exchange.
- Sterilization is done by autoclaving at 15 lbs pressure (116 to 121°C) for 15 minutes. Some carbohydrates (arabinose lactose maltose sucrose xylose) are heat labile and they should be autoclaved only for 3 minutes at 121°C.
- For heat labile carbohydrate the base broth without carbohydrate is autoclaved for 15 minutes at 121°C and then it is cooled. The carbohydrate is dissolved separately in water and sterilized by membrane filtration (0.45 µm) and then it is aseptically added to cooled broth.
Reagents composition for Carbohydrate Fermentation Test (per 1000 mL)
- Peptone (Proteose or Casein or Trypticase)– 10.0 g.
- Sodium chloride (NaCl)– 5.0 g.
- Beef extract (optional)– 1.0 g.
- Phenol red (pH indicator)– 0.018 g (18 mg). (Alternatively bromocresol purple 0.100 g can be used).
- Specific carbohydrate (glucose lactose sucrose maltose etc.)– 10.0 g (1% concentration).
- Distilled or demineralized water– 1000 mL.
Preparation of reagents
- The basal media components (peptone beef extract NaCl and phenol red) are measured and mixed in distilled water.
- The specific carbohydrate is added and it is dissolved completely by shaking or by gentle heating.
- The pH is checked and adjusted so that the final pH is about 7.4.
- About 4 to 7 mL of prepared broth is dispensed into individual test tubes.
- A Durham tube (inverted glass vial) is placed inside each tube and kept fully submerged. It is ensured that no air bubbles are trapped inside the Durham tube.
- The tubes are loosely capped or cotton plugged and sterilization is done by autoclaving at 121°C for 15 minutes.
- Heat sensitive carbohydrates (lactose sucrose maltose arabinose salicin trehalose xylose) should be autoclaved only for 3 minutes at 121°C. Alternatively the basal broth is autoclaved separately for 15 minutes and the carbohydrate solution is filter sterilized and aseptically added to cooled broth.
Carbohydrate Fermentation Test (Phenol Red Broth) – Procedure
- It is ensured that Durham tube kept inside the medium is completely filled with liquid and no air bubble is present inside it.
- Each test tube is labeled with the name of carbohydrate present and name of bacterial species to be tested.
- A well isolated colony is picked from a fresh (18–24 hour) pure culture using sterile inoculating loop or needle and it is inoculated into the broth. Alternatively 1–2 drops of broth culture can also be used for inoculation.
- The test tube is gently swirled to distribute the bacteria evenly in the broth. The Durham tube is not disturbed and the liquid is not allowed to touch the cap.
- The cap of the tube is kept slightly loose (about quarter turn) for gas exchange.
- The inoculated tubes along with an uninoculated control tube are incubated at 35°C to 37°C for 18 to 48 hours.
- The tubes are examined daily for colour change in medium for acid production and for gas bubble trapped in Durham tube for gas production.
- If no colour change and no gas bubble is seen the tubes are re-incubated for extra time (another 24 hours to up to 30 days depending on microorganism) to confirm negative result as some organism are slow fermenter.
Result and Interpretation of Carbohydrate Fermentation Test
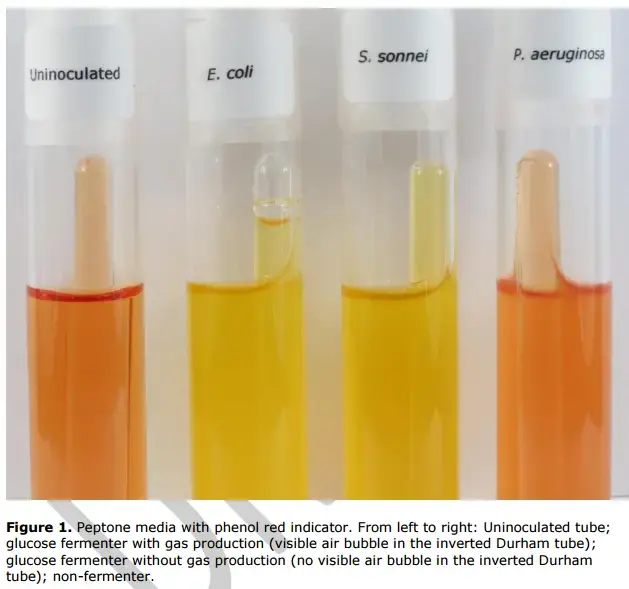
Positive for acid production (Fermentation)– The liquid medium changes colour from red-orange to yellow. This indicates that microorganism fermented the specific carbohydrate and organic acids are produced.
Positive for acid and gas production– The medium turns yellow and a visible gas bubble is trapped inside the Durham tube. This indicates that both acid and gas (CO2 or other gas) is produced during fermentation.
Negative for fermentation– The medium retains its original red or orange colour. It indicates no acid is produced and organism is unable to ferment the carbohydrate. No gas bubble is seen in Durham tube.
Alkaline reaction (Peptone degradation)– The medium turns deeper red bright pink or fuchsia colour. It indicates negative fermentation where bacteria breaks down peptones in broth and alkaline byproducts such as ammonia is produced.
Delayed fermentation– The medium may turn orange colour. This indicates delayed or slow fermentation and further incubation is required.
List of organisms giving positive and negative result in Carbohydrate Fermentation Test
| Organism | Positive result (Ferments) | Negative result (Does not ferment) |
|---|---|---|
| Escherichia coli | Glucose, Lactose, Maltose, Arabinose, Mannitol, Sorbitol, Xylose, Galactose | Cellobiose, Adonitol, Inositol, Sucrose (variable) |
| Klebsiella pneumoniae | Glucose, Lactose, Sucrose, Maltose, Arabinose, Cellobiose, Mannitol, Sorbitol, Xylose, Glucoside, Mannose, Trehalose | — |
| Proteus mirabilis | Glucose, Xylose | Lactose, Sucrose, Maltose, Arabinose, Cellobiose, Mannitol, Sorbitol, Mannose |
| Proteus vulgaris | Glucose, Sucrose, Maltose | Lactose |
| Salmonella enterica (Typhi, Typhimurium) | Glucose, Maltose, Mannitol, Xylose, Dulcitol | Lactose, Sucrose, Arabinose, Cellobiose, Sorbitol, Glycerol, Salicin |
| Pseudomonas aeruginosa | Mannitol | Glucose (non fermenter), Lactose, Sucrose, Maltose, Arabinose, Cellobiose, Sorbitol, Xylose |
| Shigella spp. | Glucose (all). Mannitol (S. flexneri, S. sonnei) | Lactose, Sucrose, Cellobiose. Mannitol (S. dysenteriae) |
| Klebsiella aerogenes | Adonitol, Arabinose, Inositol, Raffinose, Rhamnose, Salicin, Sucrose | — |
| Serratia marcescens | — | Arabinose, Dulcitol, Galactose, Raffinose, Rhamnose |
| Alcaligenes faecalis | — | No carbohydrate fermentation (non saccharolytic) |
| Staphylococcus aureus | Glucose, Lactose and or Sucrose | — |
| Citrobacter freundii | Lactose, Glycerol | — |
Precautions of Carbohydrate Fermentation Test
- Air bubbles in Durham tube– It is ensured that no preexisting air bubble is trapped inside the inverted Durham tube before inoculation. Otherwise false positive result for gas production can be obtained.
- Avoid excessive agitation– The tubes should not be over shaken during inoculation and it should not be kept in shaking incubator. Excess shaking can force air bubble into Durham tube.
- Proper reading time– The result should be read within proper time period usually 18 to 24 hours for most enterics. If it is read too late the carbohydrate may get exhausted and organism starts breaking down peptones producing alkaline byproducts and reversion can occur turning medium back to red or pink.
- Extended incubation when needed– Some slow growing organism may require prolonged incubation (up to 5 days or more) before reporting a negative result.
- Uninoculated control– An uninoculated control tube should always be run parallel for colour comparison. Some carbohydrate can cause slight acidic colour shift naturally in medium.
- Loosen caps– The caps of test tubes are kept slightly loosened (about quarter turn) during incubation to allow gas exchange.
- Aseptic technique– Strict aseptic technique is maintained during inoculation. Any contamination can give false positive result.
- Proper labeling– All tubes are clearly labeled (carbohydrate name and organism name). The broths look similar even with different carbohydrate so mislabeling can create error.
Uses of Carbohydrate Fermentation Test
- It is used for presumptive identification of microorganism based on their carbohydrate utilization pattern.
- It is used to differentiate Gram negative bacilli mainly the enteric bacteria of Enterobacteriaceae family.
- It is used for species level differentiation within same genus based on ability to ferment specific sugar (example Proteus vulgaris and Proteus mirabilis by maltose fermentation).
- It is used to differentiate yeast species such as Candida spp. and Saccharomyces spp.
- It is used to determine metabolic capability of organism to use a given carbohydrate for energy production and to observe end products such as acid and or gas.
Limitations of Carbohydrate Fermentation Test
- It is not a confirmatory test. It cannot be used alone for definitive identification and other biochemical immunological or molecular tests are required.
- False negative result can be obtained due to reversion. If the test is read too late the carbohydrate gets exhausted and peptones are metabolized producing alkaline ammonia and medium turns back to red or pink hiding the initial acid production.
- False positive colour change can occur. Reading too early can give inaccurate result and some carbohydrates can make basal medium slightly acidic giving a deceptive colour shift before bacterial growth.
- Deceptive gas production can be seen. If Durham tube is not completely filled during sterilization or tube is vigorously shaken after inoculation false positive gas bubble can appear.
- It is highly susceptible to contamination. Even minor contamination can give false positive fermentation reading.
- Poor bacterial growth can occur in standard fermentation medium. Some strains grow very poorly or fails to grow due to different nutritional requirement.
- Maltose accelerated death phenomenon may occur. Unrestricted uptake of some sugars such as maltose can crash intracellular pH and organism may die before completing metabolism giving weak or slow positive.
- In complex media reactions can be masked. In tests like Triple Sugar Iron (TSI) agar black precipitate of H2S can mask the yellow colour change of acid production.
- Preparation is cumbersome. Many different batches of media are required as different single carbohydrates are used for testing.
References
- -1-3 Enterobacteriaceae. (n.d.). Virtual Microbiology.
- A Dive Into Yeast’s Sugar Diet—Comparing the Metabolic Response of Glucose, Fructose, Sucrose, and Maltose Under Dynamic Feast/Famine Conditions. (n.d.). PubMed Central.
- American Society for Microbiology. (n.d.). Protocols: Carbohydrate fermentation by bacteria.
- American Society for Microbiology. (2012, November 1). Image gallery: Fermentation.
- BenchChem Technical Support Team. (2025, December). Application notes: Use of phenol red in microbiology for detecting bacterial growth. BenchChem.
- Chauhan, A. (2026, February 20). Oxidation fermentation test: How it determines bacterial metabolism. Flabs.
- Classification, identification, and clinical significance of Proteus, Providencia, and Morganella. (n.d.). PubMed Central.
- Comprehensive analysis of carbohydrate fermentation and differential diagnostic methodologies in clinical microbiology. (n.d.).
- Cooper, C. R., Jr. (2019). Carbohydrate fermentation test. Youngstown State University.
- Elder, J. K., & Simmons, G. C. (1963). Use of carbohydrate fermentation reactions for differentiating strains of group D streptococci. Queensland Journal of Agricultural Science, 20, 271-279.
- Fermentation test – Principle, procedure, uses and interpretation. (n.d.). Microbiology Info.
- Fermentative foods: Microbiology, biochemistry, potential human health benefits and public health issues. (n.d.). PubMed Central.
- Fricker, C. R., Warden, P. S., & Eldred, B. J. (2010). Understanding the cause of false negative beta-D-glucuronidase reactions in culture media containing fermentable carbohydrate. Letters in Applied Microbiology, 50(6), 547-551. https://doi.org/10.1111/j.1472-765X.2010.02834.x
- Gunsalus, R., & Schröder, I. (n.d.). Fermentation. Microbe Scholar – UCLA.
- Hackmann, T. J. (2024). The vast landscape of carbohydrate fermentation in prokaryotes. FEMS Microbiology Reviews, 48(4), fuae016. https://doi.org/10.1093/femsre/fuae016
- Hanson, A. (n.d.). Oxidative-fermentative test protocol. American Society for Microbiology.
- Hardy Diagnostics. (2020). Phenol red carbohydrate test media [Instructions for use].
- Hartline, R. (2023, February 18). 1.22: Fermentation. Biology LibreTexts.
- Ismail, A. A. (2017). When laboratory tests can mislead even when they appear plausible. Clinical Medicine, 17(4), 329-332. https://doi.org/10.7861/clinmedicine.17-4-329
- Jahreis, K., Bentler, L., Bockmann, J., Hans, S., Meyer, A., Siepelmeyer, J., & Lengeler, J. W. (2002). Adaptation of sucrose metabolism in the Escherichia coli wild-type strain EC3132. Journal of Bacteriology, 184(19), 5307-5316. https://doi.org/10.1128/JB.184.19.5307-5316.2002
- Kaiser, G. (2024, January 2). 8.4: Fermentation of carbohydrates w. procedures and results. Biology LibreTexts.
- Khan Academy. (n.d.). Fermentation and anaerobic respiration.
- LAB: 7 Practical pathogenic bacteria family: Enterobacteriaceae lactose none fermenting genus: Proteus. spp genus: Shigella. (n.d.).
- Lactose transport system: Topics by Science.gov. (n.d.). Science.gov.
- MacKenzie, E. (2025, October 14). 31.4: Phenol red fermentation test. Biology LibreTexts.
- Mörtberg, M., & Neujahr, H. Y. (1986). Transport and hydrolysis of disaccharides by Trichosporon cutaneum. Journal of Bacteriology, 168(2), 734-738. https://doi.org/10.1128/jb.168.2.734-738.1986
- Oregon State Ecampus. (n.d.). EX. 6: Metabolism and fermentation.
- Petersen, J., & McLaughlin, S. (2021, June 10). 7.1: Introduction to biochemical tests part I. Biology LibreTexts.
- Reiner, K. (2012, November 1). Carbohydrate fermentation protocol. American Society for Microbiology.
- Remel. (2011, March 29). Phenol red broth base [Instructions for use]. Thermo Fisher Scientific.
- Remel. (2011, March 29). Phenol red broth w/ and w/o carbohydrate w/ and w/o Durham tube [Instructions for use]. Thermo Fisher Scientific.
- Romer Labs. (2016, July 22). Preventing false-positive results in pathogen testing.
- Senior, B. W. (1981). A rapid and simple method for distinguishing colonies of proteus from those of Salmonella and Shigella. Journal of Medical Microbiology, 14(1), 151-152. https://doi.org/10.1099/00222615-14-1-151
- The Scr and Csc pathways for sucrose utilization co-exist in E. coli, but only the Scr pathway is widespread in other Enterobacteriaceae. (n.d.). PubMed Central.
- The Triple Sugar Iron (TSI) test – Procedure, uses and interpretation. (n.d.). Microbiology Info.
- Watson, R. (n.d.). Summary of biochemical tests. University of Wyoming.