What are Bt Crops?
Bt crops are the transgenic crops in which a gene from the soil bacterium Bacillus thuringiensis is introduced into the plant. It is the gene that produces special insecticidal proteins inside the plant body and these proteins are referred to as Cry or Vip proteins. When insect larvae feed on the plant the proteins is activated in their alkaline gut and this forms pores in the gut lining which finally kills the insect. It is the process by which the plant itself becomes resistant to major pests like bollworms, corn borers and rootworms without depending on chemical sprays. These Bt proteins are harmless to humans and animals because these organisms have acidic stomach and do not have the specific receptors for toxin activation. Some of the main Bt crops are Bt cotton, Bt maize, Bt soybean and Bt brinjal and these crops help in reducing pesticide use and increasing crop yield by preventing insect damage.
Definition of Bt Crops
Bt crops, or Bacillus thuringiensis crops, are genetically modified plants that incorporate genes from the bacterium Bacillus thuringiensis. These crops produce specific proteins (Cry proteins) that are toxic to certain insect pests, providing built-in protection against infestations. Common examples include cotton, corn, and brinjal. Bt crops reduce the need for chemical insecticides and are considered environmentally friendly due to the biodegradability of the proteins they produce.
The history of Bt crops
- Bt was first discovered in 1901 in diseased silkworms by Shigetane Ishiwatari.
- In 1915 the bacterium was again isolated from the Mediterranean flour moth by Berliner who named it Bacillus thuringiensis.
- It is the period of 1920s when Bt started to be used as a natural biopesticide spray by farmers.
- The first commercial Bt product was prepared in France in 1938 and it was known as Sporeine.
- In 1981 the first Bt insecticidal protein gene was cloned and sequenced.
- In 1985 Plant Genetic Systems developed the first genetically modified plants using Bt genes.
- It is in 1986–1987 that Bt genes were introduced successfully into maize, tobacco and tomato plants showing insect resistance.
- In 1995 the first Bt crops were registered and in 1996 the commercial cultivation of Bt corn and Bt cotton started in different countries.
- Bt crop adoption increased and by 2023 these crops were cultivated in 206.3 million hectares across many countries.
- Second-generation Bt crops with two pyramided genes were developed in early 2000s to slow insect resistance.
- Third-generation Bt crops were formed by adding a combination of Cry and Vip genes.
- Recent development involved combining Bt genes with RNA interference (RNAi) to build stronger insect-resistant crop varieties.
Bacillus thuringiensis
- It is a naturally occurring, aerobic, gram-positive, spore-forming bacterium.
- It is commonly known as Bt.
- It is found widely in nature such as soil, plant leaves, freshwater, stored grain dust, and dead insects.
- During its life cycle, especially in sporulation and vegetative growth phase, different proteins are produced.
- These proteins are parasporal crystal proteins and soluble proteins.
- The major toxins are Cry, Cyt and Vip toxins.
- These toxins are highly specific and lethal to certain insect groups.
- They mainly act on Lepidoptera (caterpillars), Coleoptera (beetles), and Diptera (mosquitoes and flies).
- When the insect ingests the toxin, the alkaline gut dissolves the crystal proteins.
- The activated toxin binds to specific receptors in the midgut.
- Pores are formed in the gut wall and the cells rupture.
- Finally the insect is killed.
- It is biodegradable and considered safe to humans, mammals, birds, plants and beneficial insects.
- These organisms do not have the required alkaline gut and specific receptors for toxin activation.
- It is used as a biological pesticide spray in organic farming.
- The Bt genes are extracted and inserted into crops such as corn, cotton and soybeans.
- These are called Bt crops which have in-built pest resistance.
Types of Bt crops
- Bt corn (maize)– It is engineered to resist European corn borer, southwestern corn borer and corn rootworm.
- Bt cotton– It is modified to resist cotton bollworm, pink bollworm and tobacco budworm.
- Bt soybean– It is developed to protect the crop from lepidopteran pests like velvetbean caterpillar and soybean looper.
- Bt eggplant (brinjal)– It is the crop designed to resist the fruit and shoot borer which is a destructive pest.
- Bt potato– It is engineered to resist Colorado potato beetle and potato tuber moth.
- Bt rice– It is genetically modified to resist rice stem borers and planthoppers.
- Bt sugarcane– It is engineered for resistance against sugarcane borer.
- Bt tomato– It is modified to express Bt proteins giving insect resistance.
- Other Bt crops– These are canola, broccoli, collards, chickpeas, spinach, tobacco and cauliflower where Bt toxins is expressed.
The Cry protein: mode of action
- Ingestion– It is taken inside when the susceptible insect feeds on Bt plant parts containing the inactive Cry protein crystals (protoxins).
- Solubilization and activation– It is the process where the alkaline midgut of insect dissolves the crystals and the protoxin is then cleaved by gut proteases to form the active toxin.
- Receptor binding– The activated toxin binds to specific receptors present on the apical brush border membrane of midgut epithelial cells.
- Membrane insertion and pore formation– After binding, the toxin undergoes structural change and inserts into the membrane forming pores.
- Cell breakdown (lysis)– These pores disturb the membrane potential and osmotic balance and the epithelial cells swell and rupture.
- Insect death– The damaged gut lining allows gut content to leak into the body cavity resulting in septicemia and death.
- Alternative signaling pathway– This is referred to as a model where receptor binding trigger cellular signals leading to programmed cell death.
How bt works?
- Ingestion- It is the first step where the insect feeds on Bt plant parts and takes in the inactive Bt crystal proteins (protoxins).
- Solubilization- It is the process where the alkaline midgut of the insect (pH 9.0–10.5) dissolves the crystal proteins.
- Enzymatic activation- In this step the dissolved proteins are cut by gut proteases which convert the protoxin into the active toxic fragment.
- Receptor binding- The activated toxin binds to specific receptors on the midgut epithelial cells. These receptors are not present in humans and other mammals and the toxin is digested like normal protein.
- Pore formation- After binding the toxin undergo a structural change, oligomerizes and inserts into the membrane forming pores.
- Cell lysis and death- These pores disturb osmotic balance and the gut cells swell and rupture. The gut bacteria then invade the body cavity and the insect stops feeding and dies due to septicemia and starvation.
Cry Protein Toxin and Specificity
It is the insecticidal crystal protein formed by Bacillus thuringiensis (Bt) during sporulation. The protein is produced as crystalline inclusions which is referred to as δ-endotoxins.
Origin– It is produced when the bacterium forms spores and the crystals appear along with the spore structure.
Mechanism– It is the process where the insect must ingest the protoxin. Inside the alkaline midgut the crystals dissolve and proteases convert the protoxin into active toxin. This active toxin binds to specific receptors on the epithelial cells and then inserts into the membrane forming pores which destroy the cells.
Safety– These proteins need alkaline midgut and specific receptors to become active. Humans and vertebrates lack such receptors, so the proteins are digested like ordinary proteins and do not show toxicity.
Specificity of Cry Proteins
- Cry1– It is active mainly against Lepidoptera (moths and butterflies).
- Cry2– It act against Lepidoptera and Diptera.
- Cry3– It targets Coleoptera (beetle pests).
- Cry4– It is specific against Diptera like mosquito larvae.
- Cry7 and Cry8– These are effective against Coleoptera.
- Cry9– It act against Lepidopteran pests.
- Cry10 and Cry11– It is effective mainly against Diptera insects.
- Cry18, Cry23, Cry34, Cry37 and Cry43– These proteins is active against different Coleopteran insects.
These are the Cry groups with defined insect specificity depending on the receptor type present in the insect midgut.
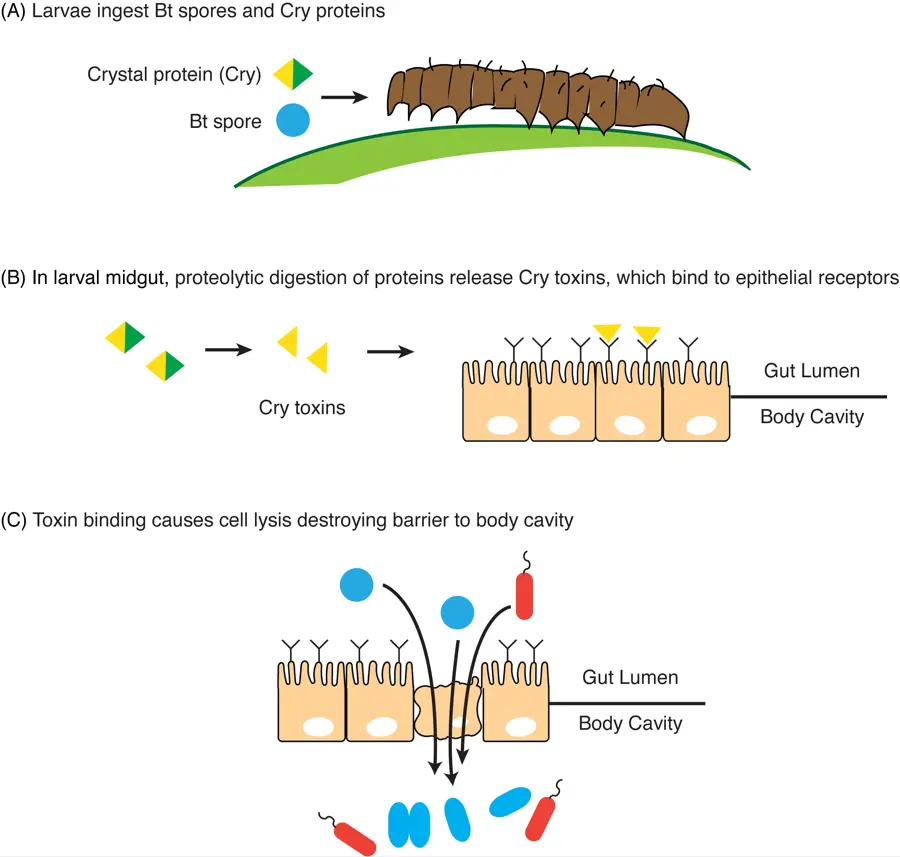
The mechanism of Bt toxicity
1. Production of Cry proteins– It is the step where Bacillus thuringiensis produces crystalline proteins during sporulation. These proteins remain inactive until they are taken inside by the insect larva.
2. Activation inside the insect gut– It is the process where the Cry protein, on reaching the alkaline midgut of the larva, is partially degraded. This degradation releases the active fragments of the protein.
3. Specificity of Cry toxins– The activated toxin binds only to specific receptors present on the midgut epithelial cells. This specificity is similar to a key fitting a lock. Only insects having these receptors, like Lepidoptera and Coleoptera, are affected. Mammals do not have such receptors, so no toxicity is shown.
4. Binding to receptors– After activation the Cry toxin binds to the gut cell receptor. This binding is important as it allows more Cry proteins to assemble at the same site.
5. Pore formation in the membrane– In this step the Cry proteins form pores in the membrane. The formation of these pores leads to lysis of epithelial cells.
6. Damage to the midgut– The rupture of epithelial cells damages the midgut lining. This creates openings through which Bt spores and natural gut microbes enter the insect body cavity.
7. Septicemia and death– Once inside, these microbes multiply quickly in the nutrient-rich body fluid. This leads to septicemia and the insect dies.
8. Safety evaluation– Research shows that Cry proteins are degraded like normal dietary proteins in the acidic mammalian stomach. Experiments from EPA showed no harmful effect even after giving very high doses to test animals. It is considered safe for humans, children and other vertebrates.
9. Agricultural importance– It is the use of Bt toxins not only to prevent pest damage but also to reduce insect-transmitted diseases in crop plants which may produce harmful compounds.
How to make Bt Crops?
- Identification and Isolation of Bt Gene
It is the step where the specific insect-toxic gene from Bacillus thuringiensis is identified. The Cry or Vip gene is isolated because these is responsible for producing insecticidal proteins. - Gene Cloning and Codon Optimization
The isolated Bt gene is cloned to make multiple copies. It is often changed synthetically because the bacterial codons do not express well in plant cells, and this optimization helps in forming higher amounts of the Bt protein. - Construction of Gene Cassette
In this step the Bt gene is inserted into a gene cassette. The cassette has promoter (to control expression of the protein) and a marker gene which is used for detecting the transformed cells. - Transformation of Plant Cells
The cassette is introduced into the plant genome. The main methods used are–
– Agrobacterium-mediated method, where Agrobacterium tumefaciens carries the gene into the plant cell.
– Direct methods, like gene gun method in which DNA coated particles is shot into the plant tissue. Electroporation and pollen tube pathway are also used sometimes. - Selection of Transformed Cells
These transformed cells are screened with the help of marker gene. Only the cells where Bt DNA is inserted successfully are selected for regeneration. - Regeneration of Whole Plant
Selected cells is grown in nutrient medium and gradually it develops into a complete plant. It is the step where the inserted gene becomes stable in the plant. - Evaluation and Field Testing
The regenerated Bt plants are tested under controlled and field conditions. The plants are observed for insect resistance, growth pattern and overall performance.
These are the main steps used for developing Bt crops.
Types of Crops Used for Bt Toxin Production
- Corn (Maize)
- Cotton
- Soybeans
- Potatoes
- Rice
- Eggplant (Brinjal)
- Tomatoes
- Sugarcane
- Canola (Oilseed Rape)
- Cowpeas
- Tobacco
- Other vegetables and legumes (broccoli, collards, chickpeas, spinach, cauliflower)
These are the major crops where Bt toxin is expressed for insect resistance.
Insects Controlled by Bt
Lepidoptera (Moths and Caterpillars)
- Cotton bollworm (Helicoverpa armigera)
- Corn earworm / Bollworm (Helicoverpa zea)
- Pink bollworm (Pectinophora gossypiella)
- Tobacco budworm (Heliothis virescens)
- European corn borer (Ostrinia nubilalis)
- Southwestern corn borer (Diatraea grandiosella)
- Asian corn borer (Ostrinia furnacalis)
- Fall armyworm (Spodoptera frugiperda)
- Beet armyworm (Spodoptera exigua)
- Sugarcane borer (Diatraea saccharalis)
- African stem borer (Busseola fusca)
- Asiatic rice borer (Chilo suppressalis)
- Rice leaf folder (Cnaphalocrocis medinalis)
- Brinjal fruit and shoot borer (Leucinodes orbonalis)
- Potato tuber moth (Phthorimaea operculella)
- Velvetbean caterpillar (Anticarsia gemmatalis)
- Soybean looper
Coleoptera (Beetles)
18. Western corn rootworm (Diabrotica virgifera virgifera)
19. Northern corn rootworm (Diabrotica barberi)
20. Colorado potato beetle (Leptinotarsa decemlineata)
Diptera (Mosquitoes and Flies)
21. Mosquitoes (e.g., Aedes aegypti, Culex quinquefasciatus, Anopheles spp.)
22. Black flies (Simulium damnosum)
Hemiptera (True Bugs)
23. Mirid bugs (Lygus hesperus, Apolygus lucorum)
24. Rice planthoppers (Laodelphax striatellus, Sogatella furcifera)
These are the major insects controlled by Bt toxins in different crop systems.
Advantages of Bt Crops
- Higher crop yield is obtained because the insect damage is very less and the plants can complete their growth without major losses.
- The use of chemical pesticide is reduced since the Bt toxin is already produced inside the crop and this control the targeted insects.
- Farmer profit is increased as the cost of buying pesticides, spraying and labor is reduced and at the same time the yield is increased.
- It is helpful for environment protection because less chemical is sprayed in the field, and it decreases soil and water contamination.
- Bt toxin is safe for beneficial organisms. It is specific to the pest group and does not harm humans, animals and useful insects.
- Human health is improved as less spraying reduces the exposure of workers to harmful chemicals.
- The crop quality is better because less insect feeding reduces the entry of fungal infection and it also lowers mycotoxin formation.
- It support food security by giving stable yield and reducing crop loss which help in providing more reliable food supply.
These are the main advantages observed when Bt crops is cultivated.
Limitations of Bt Crops
- Pest resistance can develop because long use of Bt crops put strong selection pressure on the insect population and some insects gradually become resistant to the toxin.
- The target range is narrow as Bt toxin only affect specific insect groups, and other secondary pests still damage the crop, so chemical pesticide is still required in many cases.
- Bt seeds are costly, and the high price of the seeds is difficult for small farmers, especially when the insect pressure is low and the benefit is not equal to the cost.
- Bt protein expression is affected by environment, and conditions like extreme temperature, soil salinity, water logging or nutrient stress can reduce the effectiveness of the Bt toxin.
- There is a risk of gene flow, where the Bt gene can move to nearby non-Bt crops or wild relatives by cross pollination which may create ecological concerns.
- Non-target organism concern is present because some closely related beneficial insects may also be affected even though Bt toxin is specific in action.
- Market control and public doubt is observed since the Bt technology is owned by a few corporations, and many consumers and groups express concern about long-term safety and ecological impact.
These are the main limitations that is observed in the use of Bt crops.
Uses of Bt Crops
- It is used for targeted pest control because the Bt crops produce insecticidal protein that kills specific insects like bollworm and corn borer.
- It reduces the use of chemical pesticides since the plant itself forms the Bt toxin and this decreases the spraying requirement.
- It helps in increasing crop yield as the feeding damage by insects is very less, and the plants grow with better productivity.
- It support farmer economy because less money is spent on pesticides, spraying and labour, and higher yield gives better income.
- It protects the environment by lowering chemical contamination of soil and water due to reduced pesticide application.
- It conserve beneficial insects because the Bt toxin is specific to target pests and it does not affect pollinators and natural predators.
- It improves farmer health as less handling of chemical pesticide reduces the risk of poisoning and related health issues.
- It improves food safety by reducing insect wounds on plants which also reduces fungal infection and lowers mycotoxin level.
- It helps in food security since stable yield and less crop loss support continuous food supply for increasing population.
These are the main uses observed when Bt crops is cultivated.
Examples of Bt Crops
- Bt cotton is engineered for protection against major lepidopteran insects like cotton bollworm and pink bollworm.
- Bt corn (maize) produce Bt toxin that control pests such as European corn borer, fall armyworm and also corn rootworm.
- Bt soybean is used for controlling soybean looper and velvetbean caterpillar and help in reducing chemical spray.
- Bt eggplant (brinjal) is developed mainly to resist the fruit and shoot borer which cause heavy loss in the crop.
- Bt potato is engineered to form Bt toxin against Colorado potato beetle and potato tuber moth.
- Bt rice provide resistance to rice stem borer and rice planthopper by expressing Bt proteins.
- Bt sugarcane is used for controlling the sugarcane borer using Bt genes like Cry1Ab.
- Bt tomato is modified to resist insect attack by producing Bt toxin in the plant.
- Other crops like canola, tobacco, broccoli, collards, chickpea, spinach and cauliflower are also engineered with Bt traits.
- Ahmad, A., Ghouri, M. Z., Jamil, A., Khan, S. H., Ahmad, N., & Mehboob-ur-Rahman. (2021). First-generation transgenic cotton crops. In Cotton precision breeding (pp. 229–255). Springer Nature. https://doi.org/10.1007/978-3-030-64504-5_10
- Ahmad, M. (2023). Plant breeding advancements with “CRISPR-Cas” genome editing technologies will assist future food security. Frontiers in Plant Science, 14, 1133036. https://doi.org/10.3389/fpls.2023.1133036
- Akinbo, O., Nkhabindze, B., Amedu, J., Ebegba, R., Asagbra, A., Ratemo, B. O., Dada, S. A., Muia, A., Mugiira, R., Chimphepo, L., Wristberg, L. O., Rouamba, M. W., Some, K., Nuaila, V. N. A., Atanásio, A., Shittu, O. B., Adetunji, C. O., Hamidi, L., Monga, E., Obukosia, S., Adegbaju, M. S., Acheampong, S., & Fotabong, E. (2025). Africa and zero hunger agenda: Genome editing policy landscape, challenges and opportunities. Frontiers in Bioengineering and Biotechnology, 13, 1526851. https://doi.org/10.3389/fbioe.2025.1526851
- Bravo, A., Gill, S. S., & Soberón, M. (2007). Mode of action of Bacillus thuringiensis Cry and Cyt toxins and their potential for insect control. Toxicon, 49(4), 423–435. https://doi.org/10.1016/j.toxicon.2006.11.022
- Clark, B. W., Phillips, T. A., & Coats, J. R. (2005). Environmental fate and effects of Bacillus thuringiensis (Bt) proteins from transgenic crops: A review. Journal of Agricultural and Food Chemistry, 53(12), 4643–4653.
- FAS China Staff. (2025). Biotechnology and other new production technologies annual (Report No. CH2025-0224). U.S. Department of Agriculture, Foreign Agricultural Service.
- FAS Staff. (2025). Ethiopia becomes the latest African country to approve biotech corn for commercial production (Report No. ET2025-0006). U.S. Department of Agriculture, Foreign Agricultural Service.
- Filatova, E. A. (2021). Intellectual property rights in the seed industry: Barriers to sustainable agriculture [Master’s thesis, University of Denver]. Digital Commons @ DU.
- Fuglie, K., & MacDonald, J. M. (2023, August 28). Expanded intellectual property protections for crop seeds increase innovation and market power for companies. Amber Waves, U.S. Department of Agriculture, Economic Research Service.
- Gervais, J., Cocks, M., & Mermer, S. (2025). Bacillus thuringiensis (Bt) in genetically modified crops fact sheet. National Pesticide Information Center, Oregon State University Extension Services.
- Gryspeirt, A., & Grégoire, J.-C. (2012). Effectiveness of the high dose/refuge strategy for managing pest resistance to Bacillus thuringiensis (Bt) plants expressing one or two toxins. Toxins, 4(10), 810–835. https://doi.org/10.3390/toxins4100810
- Gupta, M., Kumar, H., & Kaur, S. (2021). Vegetative insecticidal protein (Vip): A potential contender from Bacillus thuringiensis for efficient management of various detrimental agricultural pests. Frontiers in Microbiology, 12, 659736. https://doi.org/10.3389/fmicb.2021.659736
- Hu, D., Wang, D., Pan, H., & Liu, X. (2025). Molecular mechanisms underlying resistance to Bacillus thuringiensis Cry toxins in lepidopteran pests: An updated research perspective. Agronomy, 15(1), 155. https://doi.org/10.3390/agronomy15010155
- Huang, F., Andow, D. A., & Buschman, L. L. (2011). Success of the high-dose/refuge resistance management strategy after 15 years of Bt crop use in North America. Entomologia Experimentalis et Applicata, 140(1), 1–16. https://doi.org/10.1111/j.1570-7458.2011.01138.x
- International Service for the Acquisition of Agri-biotech Applications (ISAAA). (2020). Stacked traits in biotech crops (Pocket K No. 42).
- Jenney, P. (2023, June 30). A guide to seed intellectual property rights. Organic Seed Alliance.
- Kathage, J., & Qaim, M. (2012). Economic impacts and impact dynamics of Bt (Bacillus thuringiensis) cotton in India. Proceedings of the National Academy of Sciences, 109(29), 11652–11656. https://doi.org/10.1073/pnas.1203647109
- Kiresur, V. R., & Ichangi, M. (2011). Socio-economic impact of Bt cotton — A case study of Karnataka. Agricultural Economics Research Review, 24, 67–81.
- Li, K., Chen, M., Shi, J., & Mao, T. (2024). An overview of the production and use of Bacillus thuringiensis toxin. Open Life Sciences, 19(1), 20220902. https://doi.org/10.1515/biol-2022-0902
- Li, Y., Wang, C., Ge, L., Hu, C., Wu, G., Sun, Y., Song, L., Wu, X., Pan, A., Xu, Q., Shi, J., Liang, J., & Li, P. (2022). Environmental behaviors of Bacillus thuringiensis (Bt) insecticidal proteins and their effects on microbial ecology. Plants, 11(9), 1212. https://doi.org/10.3390/plants11091212
- Mahmud, T. (2025). Biotechnology and other new production technologies annual (Report No. BG2025-0011). U.S. Department of Agriculture, Foreign Agricultural Service.
- Maina, J. (2025, August 11). Africa’s quiet biotech revolution: Gene editing emerges from Europe’s anti-GMO shadow. Genetic Literacy Project.
- National Academies of Sciences, Engineering, and Medicine. (2016). Genetically engineered crops: Experiences and prospects. The National Academies Press.
- Ngongolo, K., & Mmbando, G. S. (2025). Adoption of genetically modified crops in Sub-Saharan Africa: Challenges, opportunities, and regulatory considerations for sustainable agricultural development. Discover Sustainability, 6(1), 641. https://doi.org/10.1007/s43621-025-01362-y
- Peterson, J. A., Obrycki, J. J., & Harwood, J. D. (2013). Bacillus thuringiensis: Transgenic crops. In S. E. Jorgensen (Ed.), Encyclopedia of environmental management (pp. 307–320). CRC Press. https://doi.org/10.1081/E-EEM-120046904
- Qin, Y., & Su, K. (2026). From lab to market: Industrialization barriers and regulation optimization for new breeding technologies in China. GM Crops & Food, 17(1), 2610592. https://doi.org/10.1080/21645698.2025.2610592
- Rasheed, A., Gill, R. A., Hassan, M. U., Mahmood, A., Qari, S., Zaman, Q. U., Ilyas, M., Aamer, M., Batool, M., Li, H., & Wu, Z. (2021). A critical review: Recent advancements in the use of CRISPR/Cas9 technology to enhance crops and alleviate global food crises. Current Issues in Molecular Biology, 43(3), 1950–1976. https://doi.org/10.3390/cimb43030135
- Reinders, J. D., Moar, W. J., Head, G. P., Hassan, S., & Meinke, L. J. (2023). Effects of SmartStax® and SmartStax® PRO maize on western corn rootworm (Diabrotica virgifera virgifera LeConte) larval feeding injury and adult life history parameters. PLoS ONE, 18(7), e0288372. https://doi.org/10.1371/journal.pone.0288372
- Soberón, M., & Bravo, A. (2025). The future of Bt proteins: From pore formation and insect resistance to the next generation of pest control. Toxins, 17(11), 522. https://doi.org/10.3390/toxins17110522
- Tokel, D., Genc, B. N., & Ozyigit, I. I. (2021). Economic impacts of Bt (Bacillus thuringiensis) cotton. Journal of Natural Fibers, 19(1). https://doi.org/10.1080/15440478.2020.1870613
- Xiao, Y., & Wu, K. (2019). Recent progress on the interaction between insects and Bacillus thuringiensis crops. Philosophical Transactions of the Royal Society B: Biological Sciences, 374(1767), 20180316. https://doi.org/10.1098/rstb.2018.0316
- Zhou, L., Alphey, N., Walker, A. S., Travers, L. M., Hasan, F., Morrison, N. I., Bonsall, M. B., & Raymond, B. (2018). Combining the high-dose/refuge strategy and self-limiting transgenic insects in resistance management—A test in experimental mesocosms. Evolutionary Applications, 11(5), 727–738. https://doi.org/10.1111/eva.12573