Transgenic disease-resistant crops are those plants in which a foreign resistance gene is inserted into the plant genome so that the plant can defend itself from different pathogens. It is the process where genes taken from other plants, microbes or sometimes from the pathogen itself are introduced to produce proteins that stop the infection. In this step, enzymes like chitinase and glucanase are formed which act on the cell wall of fungi or bacteria. For viral diseases, a small part of the viral coat protein is added into the plant and this is referred to as coat-protein mediated protection. It is the method that works like a defence signal inside the plant when the virus attacks. RNA interference (RNAi) is also used to silence those pathogen genes which is required for their infection cycle. These crops are important because the chemical pesticides used in the fields is reduced and the yield becomes more stable even when disease pressure is high. It is also helpful in maintaining food security since the plants remain protected throughout their growth.
History of Transgenic Crops
- It is in 1972 that recombinant DNA (rDNA) technology was developed and this is considered the basic foundation for producing genetically engineered plants.
- In 1982 the first genetically modified crop plant was prepared which was a tobacco plant that was engineered to resist antibiotics.
- In 1986 the first field trials of transgenic horticultural plants was done in the United States and France.
- In 1994 the first genetically engineered food was approved for commercial release and this was the Flavr-Savr tomato where the ripening gene is suppressed to increase shelf life.
- In the same year canola having altered oil content was released in the US and herbicide-resistant tobacco was approved in the European Union.
- In 1995 the first pesticide-producing crops was registered by EPA and Bt potato is introduced in this step.
- In 1996 commercial cultivation of GM crops started widely and herbicide-tolerant soybean and insect-resistant Bt cotton is grown.
- Bt cotton at this time was cultivated in the United States, Mexico and Australia.
- In the late 1990s transgenic technology is used to control viral diseases and GM papaya was commercialized in 1998 in Hawaii to protect the crop from Papaya Ringspot Virus.
- From the 2000s gene stacking is the process introduced where several traits like herbicide tolerance and insect resistance is combined in the same plant.
- It is in the present era that genome editing tools like CRISPR/Cas9 is used for targeted modifications in crops.
- As of 2021 the global commercial area of transgenic crops is more than 176 million hectares in different countries.
Step-by-Step Development Process of Transgenic Crops
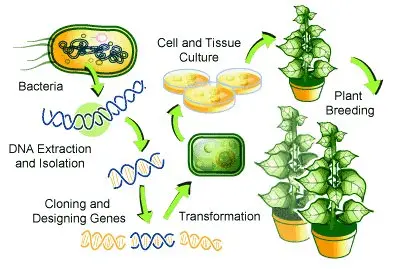
- Gene Identification and Isolation– It is the first step where the gene responsible for the desired character is identified and isolated from the donor organism. The gene may come from plant, microbe or any other source.
- Vector Construction – In this step the isolated gene is inserted into a plasmid vector. The vector normally carry promoter, selectable marker and terminator along with the target gene.
- Transformation (Gene Delivery)– The prepared vector is introduced into plant cells. These are done by Agrobacterium method or by particle bombardment where DNA-coated particles are shot into the cells.
- Selection of Transformed Cells– The treated cells are grown on selection medium containing antibiotic or herbicide. Only the cells that has received the foreign DNA will survive in the medium.
- Regeneration of Plants– The selected cells is regenerated into whole plants by tissue culture. Shoots and roots are induced using growth regulators and the plant is allowed to grow.
- Testing and Evaluation– The regenerated plants are tested to confirm stable integration of gene and proper expression of the new trait. It is usually examined in laboratory and greenhouse stages.
- Breeding and Introgression– The successful transgenic line is crossed with high-yielding varieties. This process help in transferring the new trait to commercial varieties while keeping other agronomic characters.
- Regulatory Approval and Release– Before release the plant undergo safety testing for allergenicity, toxicity and environmental effect. After approval the crop is allowed for cultivation and commercial use.
Resistance of transgenic crops to biotic stresses
- Insect and Pest Resistance
- It is achieved by inserting Bt genes so that Bt toxins are formed inside the plant which affects the insect midgut and the larvae cannot survive.
- Trypsin inhibitor and protease inhibitor genes are also used and these reduce the activity of digestive enzymes in insects, lowering their feeding ability.
- Fungal and Bacterial Resistance
- In this step chitinase and glucanase genes are expressed which degrade the fungal cell wall and the pathogens cannot grow properly.
- Antimicrobial peptides are introduced and these peptides create pores on microbial membranes.
- Resistance is also increased by stacking different R-genes that gives the plant multiple layers of protection.
- Viral Resistance
- The coat protein gene of the virus is added into the plant and this is referred to as coat-protein mediated resistance. It prevents viral multiplication inside the cells.
- RNA interference (RNAi) is used where the viral genes are silenced and the infection is reduced.
- Nematode Resistance
- Cystatin genes are used which inhibit nematode proteases and the nematode feeding on roots is decreased.
- It helps in reducing nematode population and protects the root system during plant growth.
Resistance of Transgenic Crops To Abiotic Stresses
- Osmotic and Water Deficit Tolerance
- It is achieved by accumulating osmoprotectants like proline, glycine betaine, and soluble carbohydrates which help in maintaining turgor pressure during water deficit.
- Genes for osmolyte synthesis such as P5CS and BADH are expressed and proline or glycine betaine increases inside the plant cells.
- In this step mannitol or fructan producing genes like mtlD are added which improves growth under saline or drought conditions.
- Drought Tolerance through Transcription Factors
- DREB genes are introduced and these act on stress-responsive promoter elements to activate drought and cold tolerance pathways.
- Overexpression of DREB in plants is associated with higher tolerance to freezing, water deficit and salinity.
- The major response is regulation of stress-related genes that support the plant during dehydration.
- Salinity Tolerance
- Vacuolar antiporter genes like AtNHX1 or OsNHX1 are inserted so that Na⁺ ions are pumped into vacuoles reducing cytosolic toxicity.
- SOS pathway genes help in exclusion of Na⁺ ions from the root cells and ion homeostasis is maintained.
- BADH and glycine betaine pathway also reduces salt injury by stabilizing membranes.
- Heat Stress Tolerance
- Heat shock proteins (HSPs) are expressed and these proteins stabilize the cellular proteins when high temperature denatures them.
- Antioxidant enzymes such as SOD, CAT and peroxidase are increased to remove ROS produced during heat stress.
- Polyamine producing genes like SAMDC increase spermidine and spermine which protect the cell from oxidative injury.
- Cold and Freezing Tolerance
- Fatty acid desaturase genes increase membrane fluidity and help the plant survive chilling conditions.
- DREB-related cold inducible transcription factors like Osmyb4 are introduced and these activate genes for cold adaptation.
- Overexpression of Mn-SOD and BADH protects the membrane from oxidative damage that occurs during chilling.
- Oxidative Stress Tolerance
- It is the process where enzymatic antioxidants such as SOD, catalase, and peroxidase maintain low ROS concentration.
- Transgenic plants expressing Mn-SOD or other SOD forms show better tolerance to paraquat and photooxidative stress.
- Glutathione reductase activity is also increased for detoxification of reactive molecules inside the chloroplast.
- Anoxia and Hypoxia Tolerance
- Genes encoding non-symbiotic hemoglobins from barley or bacteria are introduced which help in maintaining oxygen availability inside the cell.
- Faster germination and better growth under anaerobic condition is observed due to improved energy balance.
- Tolerance Through Gene Stacking
- Several stress-responsive genes are combined in one plant so that it resists multiple stresses at the same time.
- Co-expression of AVP1 with AtNHX1 improves both drought and salinity tolerance.
- AVP1 with OsSIZ1 is used to protect the plant from combined heat and drought stress.
Herbicide resistance of transgenic crops
- Meaning of Herbicide Resistance
- It is the process where crops are genetically modified so that they can tolerate broad-spectrum herbicides sprayed in the field.
- The major benefit is that the weeds are killed but the crop remains unaffected.
- Genes Used for Glyphosate Resistance
- The epsps gene from Agrobacterium is inserted and this allows survival in glyphosate-treated fields.
- In some plants the gat gene is also used which detoxifies glyphosate inside the cell.
- Genes Used for Glufosinate and Bialaphos Resistance
- pat or bar genes are introduced and these genes produce an enzyme that detoxifies glufosinate.
- In this step the herbicide is converted into a non-toxic compound inside the plant.
- Genes for 2,4-D Resistance
- Resistance is obtained by using aad-1 or aad-12 genes.
- These genes allow the plant to break down the herbicide and continue normal growth.
- Genes for Dicamba Resistance
- The dmo gene is inserted and this gene helps in degrading dicamba.
- It provides the plant protection when dicamba is applied to control broadleaf weeds.
- Resistance to Other Herbicides
- The csr1-2 gene gives resistance to sulfonylurea.
- The bxn gene gives tolerance to bromoxynil.
- Common Herbicide-Resistant Crops
- These include soybean, maize, cotton, canola, sugar beet, and alfalfa.
- It is widely used because the weed control becomes easier.
- Agricultural Benefits
- Weed management becomes simple and the labor needed is reduced.
- Less toxic herbicides can replace older chemicals.
- It supports no-till and low-till farming which helps in preventing soil erosion.
- Challenges and Concerns
- Continuous use of the same herbicide leads to superweeds which become resistant.
- Farmers may shift to multiple herbicide mixtures or mechanical tillage when resistance increases.
Uses of Transgenic Crops
- It is used for insect and pest resistance where Bt genes are introduced and the plant produce proteins toxic to particular insects.
- Herbicide tolerance is another major use where crops is made to survive broad-spectrum herbicides and this helps in easy weed control.
- These crops is also engineered for disease resistance and genes like chitinase or viral coat protein help the plant to resist fungal, bacterial and viral infections.
- It is used for abiotic stress tolerance so plants can survive drought, salinity, heat and low temperature conditions.
- Nutritional enhancement is an important use and crops is modified to improve vitamins, minerals or essential amino acids as seen in Golden Rice.
- In this step crop quality and shelf-life is improved by delaying ripening or preventing browning which reduce post-harvest losses.
- Transgenic plants is used in biopharming where the plant acts as a biological system to produce therapeutic proteins, antibodies or edible vaccines.
- These crops is also utilised for biofuel production and for making industrial products like special oils, biodegradable plastics or enzymes for biomass conversion.
- Plant architecture and ornamental characters is modified to change growth pattern or introduce new colours in flowers.
Advantages of Transgenic Crops
- It is used for increasing crop yield and stability because the plants can avoid major losses caused by pests and weeds.
- Pest and disease resistance is a major advantage where the crop is engineered to protect itself from insects, viral diseases or fungal pathogens.
- Herbicide tolerance is also achieved and the crop can survive broad-spectrum herbicides, helping in easy weed control with less toxic applications.
- These crops require reduced chemical use and this help in protecting water sources and beneficial insects in the field.
- Soil conservation is improved because herbicide-tolerant crops support no-till farming which reduce soil erosion and save fuel.
- It is economical for farmers as production cost is lowered and higher yield gives more profit.
- Improved human health and worker safety occur since fewer pesticide sprays is required and exposure to chemicals becomes lower.
- It is useful in abiotic stress tolerance where plants continue to grow under drought, heat, cold or saline conditions.
- Nutritional enhancement is also possible and crops is modified to increase vitamins, minerals or better fatty acids.
- Improved shelf-life and quality is another advantage where delayed ripening or reduced browning help in decreasing food waste.
- Transgenic crops is also used in industrial and pharmaceutical purposes where plants act as biofactories for producing proteins, vaccines or better biofuel feedstocks.
Limitations of Transgenic Crops
- It is observed that constant use of transgenic crops create strong selection pressure and pests or weeds may develop resistance which reduce long-term effectiveness.
- Gene flow is another limitation where the transgenes can move through pollen and contaminate wild relatives or non-transgenic crops.
- These crops may also cause unintended environmental effects and non-target insects, pollinators or soil microbes can be affected by the management practices.
- High development and regulatory cost is a major issue because creating and approving new traits is expensive and time consuming.
- Consumer resistance is present in many regions and some countries restrict or ban GM crops which create trade barriers.
- Seed cost is high since transgenic seeds are patented and farmers cannot save or replant the seeds legally.
- In this step complex management requirements appear as farmers must follow insect-resistance management like refuge planting which is not followed properly many times.
- Yield drag may occur when multiple transgenes is added and the plant metabolism is disturbed leading to reduced performance.
Examples of Transgenic Crops
- Cotton – It is engineered for insect resistance like Bt cotton and also for herbicide tolerance.
- Maize (Corn) – These are modified for Bt insect resistance, herbicide tolerance and also drought-tolerant types.
- Soybean – It is engineered to tolerate broad herbicides and some varieties have improved nutritional oil content.
- Rice – Golden Rice is an example which produce beta-carotene to reduce vitamin A deficiency.
- Papaya – It is engineered to resist Papaya Ringspot Virus and varieties like SunUp and Rainbow is common.
- Potato – These crops is modified for insect resistance, late blight resistance and reduced browning during cooking.
- Tomato – It is engineered for delayed ripening, improved shelf-life and better nutritional characters.
- Canola / Rapeseed – These plants is modified for herbicide tolerance and improved oil properties like DHA canola.
- Alfalfa – It is engineered for glyphosate tolerance and reduced lignin for better digestibility.
- Brinjal (Eggplant) – Bt brinjal is used for resistance to fruit and shoot borer insect.
- Squash and Watermelon – These are modified for resistance to viral diseases like CMV and ZYMV.
- Apple – Arctic Apple is engineered for non-browning flesh and also for disease resistance.
- Beans – Some beans is biofortified to increase iron content and also made resistant to viral infections.
- Sugar Beet – It is engineered mainly for herbicide tolerance to make weed control easier.
- Ornamentals (Carnations and Roses) – These plants is modified for new colour traits like blue or violet petals.
- Akhtar, N., Perween, S., & Ansari, A. M. (n.d.). Resistance to Papaya Ringspot Virus: A review. Agriculture Association of Textile Chemical and Critical Reviews Journal.
- Aleem, M., Aleem, S., Sharif, I., Aleem, M., Shahzad, R., Khan, M. I., Batool, A., Sarwar, G., Farooq, J., & Ahmad, P. (2022). Whole-genome identification of APX and CAT gene families in cultivated and wild soybeans and their regulatory function in plant development and stress response. Antioxidants, 11(8), 1626.
- Anugraha, A. C., Thomas, T., & Thomas, D. T. (2023). Transgenic technology in crop improvement. In N. Kumar (Ed.), Biotechnology and Crop Improvement: Tissue Culture and Transgenic Approaches (pp. 1-24). CRC Press.
- Arends, B., Reisig, D. D., Gundry, S., Huseth, A. S., Reay-Jones, F. P. F., Greene, J. K., & Kennedy, G. G. (2021). Effectiveness of the natural resistance management refuge for Bt-cotton is dominated by local abundance of soybean and maize. Scientific Reports, 11(1), 17601.
- Ceccon, C. C., Caverzan, A., Margis, R., Salvadori, J. R., & Grando, M. F. (2020). Gene stacking as a strategy to confer characteristics of agronomic importance in plants by genetic engineering. Ciência Rural, 50(6), e20190207.
- dos Santos, C., & Franco, O. L. (2023). Pathogenesis-related proteins (PRs) with enzyme activity activating plant defense responses. Plants, 12(11), 2226.
- Esmaeili, N., Shen, G., & Zhang, H. (2022). Genetic manipulation for abiotic stress resistance traits in crops. Frontiers in Plant Science, 13, 1011985.
- F. B. C., & A. P. (2025). Antioxidant defense systems in plants: Mechanisms, regulation, and biotechnological strategies for enhanced oxidative stress tolerance. Life, 15(8), 1293.
- Farvardin, A., González-Hernández, A. I., Llorens, E., Camañes, G., Scalschi, L., & Vicedo, B. (2024). The dual role of antimicrobial proteins and peptides: Exploring their direct impact and plant defense-enhancing abilities. Plants, 13(15), 2059.
- Food and Agriculture Organization of the United Nations. (2004). Economic impacts of transgenic crops. In The State of Food and Agriculture 2003-2004: Agricultural Biotechnology: Meeting the Needs of the Poor? FAO.
- Gervais, J., Cocks, M., & Mermer, S. (2025). Bacillus thuringiensis (Bt) in genetically modified crops fact sheet. National Pesticide Information Center, Oregon State University Extension Services.
- Hardee, D. D., Van Duyn, J. W., Layton, M. B., & Bagwell, R. D. (2001). Bt cotton & management of the tobacco budworm-bollworm complex (ARS–154). U.S. Department of Agriculture, Agricultural Research Service.
- International Service for the Acquisition of Agri-biotech Applications (ISAAA). (2020). Pocket K No. 42: Stacked traits in biotech crops.
- International Service for the Acquisition of Agri-biotech Applications (ISAAA). (2024). ISAAA webinar: Global status of commercialized biotech/GM crops in 2024.
- Jo, K.-R., Kim, C.-J., Kim, S.-J., Kim, T.-Y., Bergervoet, M., Jongsma, M. A., Visser, R. G. F., Jacobsen, E., & Vossen, J. H. (2014). Development of late blight resistant potatoes by cisgene stacking. BMC Biotechnology, 14, 50.
- Journal of Sustainable Technology in Agriculture. (2025). Genetic engineering for drought tolerance in crop plants: Advances and strategies. Journal of Sustainable Technology in Agriculture, 1(2).
- Kertbundit, S., Pongtanom, N., Ruanjan, P., & Chantasingh, D. (2007). Resistance of transgenic papaya plants to Papaya ringspot virus. Biologia Plantarum, 51(2), 333-339.
- Khademi, M., Varasteh-Shams, M., Nazarian-Firouzabadi, F., & Ismaili, A. (2020). New recombinant antimicrobial peptides confer resistance to fungal pathogens in tobacco plants. Frontiers in Plant Science, 11, 1236.
- Kheemji, F. Z., Shair, G., Israr, A., Farooq, N., Hammad, M., Shakeel, M., Shafiq, M., & Sherazi, S. (2026). Pathogenesis-related proteins in plants: Molecular features, functional diversity, and biotechnological applications. Journal of Physical, Biomedical and Biological Sciences, 5(1), 52.
- Kuhl, J. C., Zarka, K., Coombs, J., Kirk, W. W., & Douches, D. S. (2007). Late blight resistance of RB transgenic potato lines. Journal of the American Society for Horticultural Science, 132(6), 783-789.
- Legan, A. W., Allan, C. W., Jensen, Z. N., Degain, B. A., Yang, F., Kerns, D. L., Benowitz, K. M., Fabrick, J. A., Li, X., Carrière, Y., Matzkin, L. M., & Tabashnik, B. E. (2024). Mismatch between lab-generated and field-evolved resistance to transgenic Bt crops in Helicoverpa zea. Proceedings of the National Academy of Sciences, 121(47), e2416091121.
- Lin, J., Hazaisi, H., Guan, Y., & Bai, M. (2026). Multiplex gene editing drives revolution in crop breeding: Overlaid editing of multiple genes and customization of complex traits. Advanced Biotechnology, 4(1), 5.
- Naresh, M., & Patil, B. L. (2026). Distinct patterns of virus derived-siRNAs in response to Papaya ringspot virus infection in differentially responding genotypes of papaya (Carica papaya L.) and its wild relative. 3 Biotech, 16(1), 52.
- National Academies of Sciences, Engineering, and Medicine. (2016). Social and economic effects of genetically engineered crops. In Genetically Engineered Crops: Experiences and Prospects. National Academies Press.
- National Research Council. (2010). The impact of genetically engineered crops on farm sustainability in the United States. The National Academies Press.
- Paluchowska, P., Śliwka, J., & Yin, Z. (2022). Late blight resistance genes in potato breeding. Planta, 255(6), 127.
- Panek, B. (2024). Anti-herbivory for fall armyworm and corn earworm management [Master’s thesis, The Pennsylvania State University]. Electronic Theses and Dissertations for Graduate School.
- Parmar, N., Singh, K. H., Sharma, D., Singh, L., Kumar, P., Nanjundan, J., Khan, Y. J., Chauhan, D. K., & Thakur, A. K. (2017). Genetic engineering strategies for biotic and abiotic stress tolerance and quality enhancement in horticultural crops: A comprehensive review. 3 Biotech, 7(4), 239.
- Patil, L. N., Patil, A. A., Patil, S. A., Sancheti, S. D., & Agrawal, V. K. (2025). Advancements in crop yield improvement through genetic engineering. Current Agriculture Research Journal, 12(3).
- Peng, T., Sun, X., & Mumm, R. H. (2013). Optimized breeding strategies for multiple trait integration: I. Minimizing linkage drag in single event introgression. Molecular Breeding, 33(1), 89-104.
- Pilson, D., & Prendeville, H. R. (2004). Ecological effects of transgenic crops and the escape of transgenes into wild populations. Annual Review of Ecology, Evolution, and Systematics, 35, 149-174.
- Sanford, J. C., & Johnston, S. A. (1985). The concept of parasite-derived resistance: Deriving resistance genes from the parasite’s own genome. Journal of Theoretical Biology, 113(2), 395-405.
- Schünemann, R., Knaak, N., & Fiuza, L. M. (2014). Mode of action and specificity of Bacillus thuringiensis toxins in the control of caterpillars and stink bugs in soybean culture. ISRN Microbiology, 2014, 135675.
- Soostani, S. B., Ranjbar, M., Memarian, A., Mohammadi, M., & Yaghini, Z. (2025). Regulation of APX, SOD, and PAL genes by chitosan under salt stress in rapeseed (Brassica napus L.). BMC Plant Biology, 25, 824.
- Sustainability Directory. (2024). Bt toxins in agriculture. Energy & Climate.
- Van Deynze, A., Bradford, K. J., Daniell, H., DiTomaso, J. M., Kalaitzandonakes, N., Mallory-Smith, C., Stewart, C. N., Jr., Strauss, S. H., & Van Acker, R. (2011). The science of gene flow in agriculture and its role in co-existence [Meeting Report]. USDA NIFA / University of California, Davis.
- Velankar, R., Nerkar, G., Nagpurkar, M., & Jagtap, K. (2024). Genetically modified crops: A pivotal endeavor in biotechnology. In H. Tombuloglu & G. Tombuloglu (Eds.), Genetically Modified Organisms. IntechOpen.
- Verma, S. B. (2025). Advances in transgenic technologies and their role in crop breeding and improvement: From first-generation GM crops to next-gen precision breeding. Quest Journals Journal of Research in Agriculture and Animal Science, 12(4), 6-11.
- Wikipedia contributors. (2026). Papaya ringspot virus. In Wikipedia, The Free Encyclopedia.
- Yang, F., Santiago González, J. C., Williams, J., Cook, D. C., Gilreath, R. T., & Kerns, D. L. (2019). Occurrence and ear damage of Helicoverpa zea on transgenic Bacillus thuringiensis maize in the field in Texas, U.S. and its susceptibility to Vip3A protein. Toxins, 11(2), 102.
- Yu, H., Yang, Q., Fu, F., & Li, W. (2022). Three strategies of transgenic manipulation for crop improvement. Frontiers in Plant Science, 13, 948518.
- Zarka, K. A., Jagd, L. M., & Douches, D. S. (2024). T-DNA characterization of genetically modified 3-R-gene late blight-resistant potato events with a novel procedure utilizing the Samplix Xdrop® enrichment technology. Frontiers in Plant Science, 15, 1330429.