What is the Electrofusion of Plant Cells?
Electrofusion is a technique used for the fusion of two or more plant cells by the application of an electric field. It is commonly used for the fusion of plant protoplasts in plant biotechnology. In this method the plasma membranes of adjacent cells are temporarily disturbed and the cells combine together to form a single hybrid cell.
In this process the fusion generally occurs in two steps. First a low-voltage alternating current (AC) is applied to the suspension of protoplasts. This causes the cells to move and align with each other by a process known as dielectrophoresis. The cells come very close to each other and form chain-like structures which are referred to as pearl chains. In this stage the membranes remain intact but the cells are arranged in close contact.
In the second step a short high-voltage direct current (DC) pulse is applied. This electrical pulse causes a temporary breakdown of the plasma membranes at the point of contact. Small pores are formed in the membranes and the adjacent cells fuse together forming a single hybrid cell. After fusion the membranes reseal again and the fused cell continues its normal activity.
Electrofusion is considered an important method for protoplast fusion because the process is rapid and highly efficient. It is also less toxic to the cells and provides better control over the fusion process. For this reason it is widely used in plant genetic engineering and plant breeding experiments.
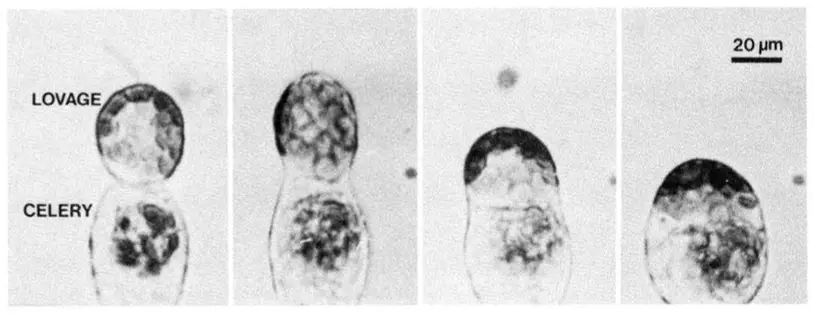
Principle of Electrofusion Technique
The principle of electrofusion is based on the application of controlled electrical fields to induce fusion between two or more plant cells. It is the process in which electrical energy is used to bring the cells into close contact and then cause the fusion of their plasma membranes. This technique mainly depends on the electrical properties of the cell membrane and the ability of electric pulses to temporarily disturb the membrane structure.
In this method the cells or protoplasts are first suspended in a suitable fusion medium inside a fusion chamber. A low-voltage alternating current (AC) is then applied to the cell suspension. This electrical field causes a phenomenon known as dielectrophoresis. In this process a dipole moment is induced in the cells and they move towards each other and align in a linear arrangement. These aligned cells form chain-like aggregates which are referred to as pearl chains.
After the cells are brought into very close membrane contact a short high-voltage direct current (DC) pulse is applied. This strong electric pulse causes a reversible electrical breakdown of the plasma membrane which is referred to as electroporation. Temporary pores are produced in the membrane at the contact region of the cells. Through these pores the membranes of adjacent cells merge together and a cytoplasmic connection is formed between them.
As the pores enlarge and the membrane edges reorganize the cells gradually fuse together forming a single multinucleated hybrid cell. In some cases a mild AC pulse is also applied after fusion to keep the fused cells together until the membranes stabilize. Thus the electrofusion technique is based on electrical alignment of cells followed by membrane breakdown and fusion.
Requirements for Electrofusion Technique
The electrofusion technique requires certain materials, equipment and suitable conditions for successful fusion of plant protoplasts. These requirements help in proper alignment of cells and fusion of their plasma membranes.
Electronic pulse generator – It is an instrument used to produce controlled electrical fields. It can generate alternating current (AC) as well as direct current (DC) pulses which are required for the electrofusion process.
Fusion chamber with electrodes – The protoplast suspension is placed in a fusion chamber containing parallel electrodes. These electrodes are generally made of gold, platinum, silver or stainless steel.
Inverted microscope – It is used to observe the alignment of protoplasts and the fusion process during the experiment.
Healthy parent plant cells – Viable plant cells are required as the source material for the isolation of protoplasts.
Cell wall degrading enzymes – Enzymes such as cellulase, macerozyme, driselase and pectinase are used for removing the cell wall and producing protoplasts.
Suitable protoplast density – The concentration of protoplasts is generally maintained around 1×105 to 6×105 cells per ml for proper fusion.
Low conductivity fusion medium – A fusion medium with low ionic strength is required for proper alignment of cells during electrofusion.
Osmotic stabilizers – Substances such as mannitol or sorbitol (0.4 M–0.7 M) are added to maintain osmotic balance and to prevent bursting of protoplasts.
Calcium ions (Ca²⁺) – Calcium ions are added in small amount to stabilize the plasma membrane and help in resealing of the membrane after fusion.
Electrical field conditions – A low-voltage alternating current (AC) field is first applied for alignment of cells forming pearl chains and then a short high-voltage direct current (DC) pulse is applied which causes membrane breakdown and fusion of the cells.
Protocol of Electrofusion Technique
The electrofusion technique is used to fuse two protoplasts by applying an electric field. It allows the membranes of adjacent cells to merge and form hybrid cells. The procedure is carried out through several sequential steps.
Step 1 – Plant Pre-treatment and Protoplast Isolation
- Healthy parent plants are first selected and grown under controlled conditions.
- In many cases the plant tissues are pre-treated to stabilize cells. The plants are kept in dark at 10°C for about 24 hours or 25°C for about 48 hours.
- Suitable tissues such as leaves or stems are excised and cut into small pieces using a sterile scalpel.
- The chopped tissues are placed in an enzyme solution containing 1% cellulase and 0.5% macerozyme.
- This enzyme mixture digests the cell wall and releases the protoplasts.
- The mixture is incubated 14–16 hours in dark at 25–30°C with gentle shaking so that the cell walls is completely degraded.
Step 2 – Purification and Preparation of Protoplast Suspension
- The digested tissue mixture is filtered through nylon mesh (55–80 µm pore size) to remove cell debris and undigested materials.
- The filtrate containing protoplasts is purified by centrifugation, often using a sucrose density gradient.
- The intact protoplasts float at the interface and are separated from heavy cellular debris.
- These purified protoplasts are washed and resuspended in a low conductivity fusion medium.
- The medium generally contains 0.4–0.7 M mannitol as osmotic stabilizer and sometimes 0.5 mM Ca²⁺ which stabilize the plasma membrane.
- The density of protoplast suspension is adjusted using a hemocytometer.
- Usually the optimal density is 1 × 10⁵ to 6 × 10⁵ protoplasts per mL.
- The viability of protoplasts may be checked using fluorescein diacetate (FDA) stain.
Step 3 – Chamber Loading and AC Alignment
- The two parental protoplast populations are mixed together generally in 1:1 ratio.
- A small volume of the mixture (20–80 µL) is transferred into a sterile electrofusion chamber fitted with parallel electrodes.
- The chamber is placed under an inverted microscope to observe the process.
- A low-voltage alternating current (AC) field is applied.
- Typical parameters are 40–100 V/cm at about 1 MHz for 10–20 seconds.
- The AC field induces dipole formation in cells causing them to move toward electrodes and align in chains.
- These aligned cells form close contacts which is referred to as “pearl chain formation.”
Step 4 – Fusion Pulse (DC Field)
- After proper alignment a high-voltage direct current (DC) pulse is applied.
- The electric pulse usually ranges from 0.5–3.0 kV/cm for 40–80 microseconds.
- This pulse causes temporary breakdown of the plasma membrane, a phenomenon called electroporation.
- Small aqueous pores are formed at the contact region between adjacent protoplasts.
- Through these pores the membranes of the cells fuse together.
- Sometimes a mild AC field is reapplied briefly to keep the fused cells together until the membrane reseals.
- Under the microscope the fused cells initially appear as figure-8 shaped structures which later become a single spherical hybrid cell (heterokaryon).
Step 5 – Post-fusion Recovery and Culture
- After fusion the cells are allowed to rest for about 30 minutes in the chamber or in a sterile Petri dish.
- This recovery period helps the membranes to reorganize and stabilize.
- The fused protoplasts are then transferred into a suitable liquid or semi-solid culture medium.
- Often the cells are embedded in low-gelling temperature agarose to provide mechanical support.
- The medium is supplemented with plant growth regulators such as auxins (NAA or 2,4-D) and cytokinins (BAP or zeatin).
- These regulators stimulate cell wall regeneration and cell division, which generally begins after 2–3 days.
- The dividing cells form microcalli, which are cultured in dark until they reach around 3 mm size.
- Later they are transferred to shoot regeneration medium under light conditions for the development of complete hybrid plants.
Advantages of Electrofusion method
- It gives high efficiency in cell fusion and the yield of hybrids is much higher compared to chemical fusion methods such as PEG.
- It does not require toxic chemical stimulants, therefore the cells maintain high viability and structural integrity.
- The fusion process can be precisely controlled by regulating electrical parameters like voltage, frequency and pulse duration.
- The method produces highly reproducible results because the physical conditions can be controlled easily.
- It is a simple and rapid technique and many fusion events can be completed within a short period of time.
- The fusion process can be observed directly under microscope which allows real-time monitoring of the cells.
- It can fuse a wide variety of plant and animal cells and there is no limitation of host range.
- The electrically fused cells generally show better growth during early stages and the somatic hybrids often shows higher fertility.
- Large numbers of protoplasts can be fused at the same time and it is also suitable for small or rare cell populations.
Limitations of Electrofusion method
- The technique requires specialized equipment such as electronic generators and fusion chambers, therefore the procedure becomes costly and technically demanding.
- The optimization of electrical parameters like voltage and pulse duration is difficult and time-consuming because different species and cell types require different conditions.
- The protoplasts sometimes adhere to the metal electrodes (gold, silver or platinum) present in the fusion chamber which may lead to damage or breakage of cells.
- The success of fusion largely depends on the physiological condition of the protoplasts and suitable conditions for one type of cells may not work for other cells.
- In some plants, especially many dicot species, the protoplasts show resistance to electrical fusion and the efficiency becomes lower.
- When the density of protoplasts is very high, fusion of multiple cells may occur leading to multinucleate cells which generally show poor survival in culture.
- The fusion results in hybrids with polyploid chromosome numbers and further analysis is required to stabilize chromosome number and remove unwanted genes.
- Immediately after fusion, hybrid cells are difficult to distinguish from parental cells, therefore identification and selection becomes complicated.
- The regeneration of a complete plant from fused protoplasts is difficult in many species and this becomes a major limitation of the technique.
Uses of Electrofusion Method
- It is used in plant breeding for the production of somatic hybrids by fusing cells of sexually incompatible plant species.
- It is used for transferring complex polygenic traits between plants which is difficult by normal genetic engineering methods.
- It is used in developing crop plants with resistance to diseases caused by viruses, bacteria and fungi.
- It is also used to develop plants that can tolerate environmental stresses such as drought, salinity and cold conditions.
- It is used for the production of cybrids (cytoplasmic hybrids) where cytoplasm and organelles of one cell combine with the nucleus of another cell.
- It is used in transferring cytoplasmic male sterility (CMS) which is important for hybrid seed production.
- It is used in plant breeding to produce triploid plants by fusion of diploid and haploid protoplasts which helps in development of seedless fruits.
- It is used for introducing foreign DNA, RNA, proteins, drugs or dyes directly into target cells.
- It is used in medicine for the production of hybridoma cells which produce monoclonal antibodies.
- It is used in cancer research for developing tumor vaccines by fusion of dendritic cells with tumor cells.
- It is used in nuclear transfer where the nucleus of one cell is transferred into the cytoplasm of another cell.
- It is also used for extraction of intracellular molecules and proteins from biological cells.
- BTX Harvard Apparatus. (n.d.). Tech-Trends volume 2, series 2: Plant protoplast fusion.
- BTX. (n.d.). Electrofusion vs PEG. BTX Online.
- Central Plastics Company. (2003). Electrofusion installation procedure manual.
- Cheng, J., & Saunders, J. A. (1995). Protoplast electrofusion and regeneration in potato. Methods in Molecular Biology, 55, 181–188. https://doi.org/10.1385/0-89603-328-7:181
- Chhatrapati Shahu Ji Maharaj University. (n.d.). Protoplast fusion. Gyan Sanchay.
- Daniell, H., Khan, M. S., & Allison, L. (2002). Milestones in chloroplast genetic engineering: An environmentally friendly era in biotechnology. Trends in Plant Science, 7(2), 84–91. https://doi.org/10.1016/s1360-1385(01)02193-8
- Daniell, H., Kumar, S., & Dufourmantel, N. (2005). Breakthrough in chloroplast genetic engineering of agronomically important crops. Trends in Biotechnology, 23(5), 238–245. https://doi.org/10.1016/j.tibtech.2005.03.008
- Donovan, A., Isaac, S., & Collin, H. A. (1990). Electrofusion of plant cells. Methods in Molecular Biology, 6, 373–379. https://doi.org/10.1385/0-89603-161-6:373
- Gieniec, M., Siwek, J., Oleszkiewicz, T., Maćkowska, K., Klimek-Chodacka, M., Grzebelus, E., & Baranski, R. (2020). Real-time detection of somatic hybrid cells during electrofusion of carrot protoplasts with stably labelled mitochondria. Scientific Reports, 10, Article 18811. https://doi.org/10.1038/s41598-020-75983-w
- Greplová, M., Polzerová, H., & Domkářová, J. (2007). Procedure of protoplast electrofusion (in Solanum genus). Potato Research Institute Havlíčkův Brod.
- Hayes Industrial Solutions. (2024). Electrofusion procedure. Thermoplastic Pipe Fusion Welding Equipment.
- Liu, S., Li, X., Zhu, J., Jin, Y., Xia, C., Zheng, B., Silvestri, C., & Cui, F. (2024). Modern technologies provide new opportunities for somatic hybridization in the breeding of woody plants. Plants, 13(18), Article 2539. https://doi.org/10.3390/plants13182539
- Marczakiewicz-Perera, P., Winkelmann, T., Köhler, M., & Cao, J. (2025). Droplet-based microfluidics platform for investigation of protoplast development of three exemplary plant species. Scientific Reports, 15(1), Article 40332. https://doi.org/10.1038/s41598-025-28956-w
- Narra, M. N., Nakazato, I., Polley, B., Arimura, S., Woronuk, G. N., & Bhowmik, P. K. (2025). Recent trends and advances in chloroplast engineering and transformation methods. Frontiers in Plant Science, 16, Article 1526578. https://doi.org/10.3389/fpls.2025.1526578
- Navrátilová, B. (2004). Protoplast cultures and protoplast fusion focused on Brassicaceae – a review. Horticultural Science, 31(4), 140–157.
- Nea, L. J., & Bates, G. W. (1987). Factors affecting protoplast electrofusion efficiency. Plant Cell Reports, 6(5), 337–340. https://doi.org/10.1007/BF00269554
- Ollitrault, P., Dambier, D., Cabasson, C., Teisson, C., & Luro, F. (1994). Protoplast fusion in citrus. Fruits, 49(5-6), 401–403.
- Pongchawee, K., Na-Nakorn, U., Lamseejan, S., Poompuang, S., & Phansiri, S. (2006). Factors affecting the protoplast isolation and culture of Anubias nana Engler. International Journal of Botany, 2(2), 193–200.
- Raj, A. (2018). Protoplast fusion and somatic hybridization | biotechnology. Biology Discussion.
Sonidel. (n.d.). Plant protoplast fusion (Protocol No. PL-001 / PL-002). - Tempelaar, M. J., & Jones, M. G. K. (1985). Fusion characteristics of plant protoplasts in electric fields. Planta, 165(2), 205–216. https://doi.org/10.1007/BF00395043
- The biophysics and methodological evolution of plant protoplast electrofusion: Requirements, protocols, and strategic applications in somatic hybridization. (n.d.).
- Trafton, A. (2019, February 25). An easier way to engineer plants. MIT News, Massachusetts Institute of Technology.
- Trick, H. N., & Bates, G. W. (1995). Electrofusion of plant protoplasts. Methods in Molecular Biology, 55, 165–173. https://doi.org/10.1385/0-89603-328-7:165
- Van Wert, S. L., & Saunders, J. A. (1992). Electrofusion and electroporation of plants. Plant Physiology, 99(2), 365–367. https://doi.org/10.1104/pp.99.2.365