Agrobacterium is a rod-shaped, Gram-negative, aerobic soil bacterium which acts as a plant pathogen. It infects plants mainly through wound sites present in plant tissues. When the bacterium enters the plant through these wounded regions it causes diseases such as crown gall disease and hairy root disease. These diseases are characterized by tumor-like swellings or abnormal root growth in the infected plants.
One important feature of this bacterium is its ability to transfer a part of its own DNA into the plant cell. This transferred DNA is called T-DNA (Transfer DNA) which is present on a tumor-inducing plasmid (Ti-plasmid). During infection this T-DNA is transferred into the genome of the host plant cell and becomes integrated with the plant DNA. Because of this integration the infected plant cells start producing large amount of growth hormones which results in uncontrolled cell division and tumor formation. The transformed plant cells also produce special compounds called opines which are utilized by the bacterium as a nutrient source.
Because of this natural ability of DNA transfer, Agrobacterium is widely used in plant biotechnology. In genetic engineering the tumor-causing genes present in the Ti-plasmid are removed and replaced with useful foreign genes. Thus the bacterium is used as a biological vector for transferring desirable genes into plants to produce genetically modified or transgenic plants.
What is Agrobacterium-Mediated Gene Transfer?
Agrobacterium–mediated gene transfer is a genetic engineering technique by which foreign genes are introduced into plant cells with the help of the bacterium Agrobacterium. This process is based on the natural ability of the bacterium to transfer a segment of its DNA into the genome of a plant cell. In nature Agrobacterium contains a tumor–inducing plasmid (Ti–plasmid) which carries a DNA region known as T–DNA (Transfer DNA). During infection this T–DNA is transferred into the plant cell and becomes integrated with the plant chromosome causing disease. In genetic engineering this natural process is modified by removing the tumor–causing genes from the Ti–plasmid and replacing them with a desired foreign gene.
In laboratory conditions a binary vector system is generally used for gene transfer. In this system the gene of interest is inserted between the border sequences of T–DNA in a small plasmid vector. The virulence genes (vir genes) which help in cutting and transferring the T–DNA are present on another helper plasmid inside the bacterium. When the genetically modified Agrobacterium is allowed to infect plant tissues such as leaf discs, embryos or roots, the bacterium attaches to the plant cells and transfers the engineered T–DNA into the nucleus of the plant cell. The transferred DNA becomes integrated with the plant chromosome and the plant cell starts expressing the new gene. These transformed cells are then cultured by tissue culture techniques to produce whole transgenic plants. This method is widely used in plant biotechnology because it is simple, efficient and produces stable gene transfer in plants.
Agrobacterium-mediated Gene Transfer Principle
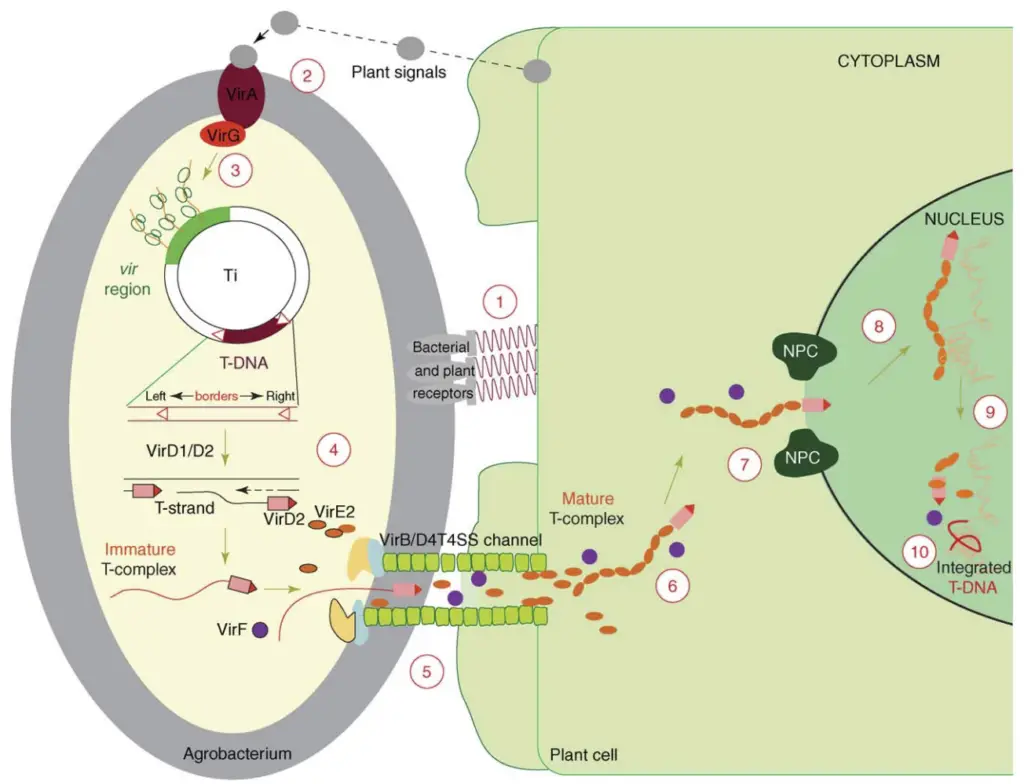
The principle of Agrobacterium-mediated gene transfer is based on the natural ability of the soil bacterium Agrobacterium tumefaciens to transfer a part of its DNA into plant cells. In this process a specific DNA segment called T–DNA (Transfer DNA) which is present on the Ti plasmid (Tumor inducing plasmid) of the bacterium is transferred into the plant genome.
This process occurs when the bacterium infects wounded plant tissues. The wounded plant cells release certain phenolic compounds such as acetosyringone. These chemical signals are detected by the bacterium and activate a group of virulence genes (vir genes) present in the Ti plasmid. The vir genes produce proteins which help in excision and transfer of the T–DNA from the bacterial cell.
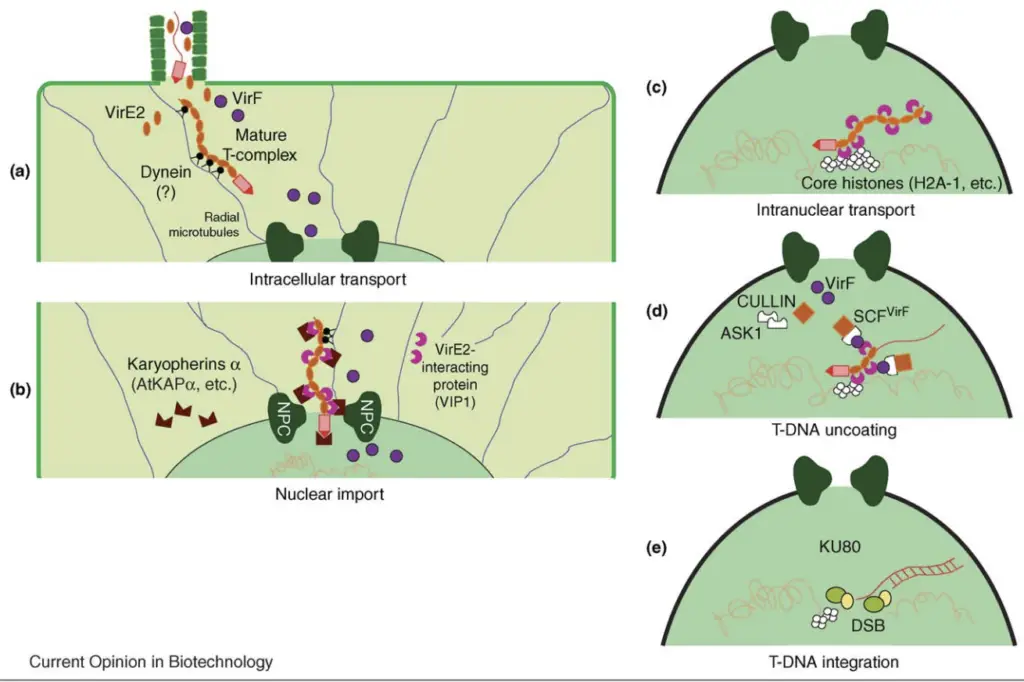
In this step a single stranded copy of T–DNA is formed and it is transported from the bacterial cell into the plant cell through a specialized transfer channel. The virulence proteins guide the T–DNA into the nucleus of the plant cell. Inside the nucleus the T–DNA becomes integrated into the plant chromosomes and it becomes a permanent part of the plant genome.
In genetic engineering the Ti plasmid is modified before the transfer process. The tumor causing genes are removed from the plasmid and replaced with a foreign gene of interest. The bacterium then transfers this modified T–DNA into the plant genome where it is expressed by the plant cells. This natural DNA transfer system makes Agrobacterium-mediated transformation one of the most efficient and widely used methods for producing transgenic plants.
Requirements for Agrobacterium-mediated Gene Transfer
Some of the important requirements for Agrobacterium-mediated gene transfer are as follows–
Materials and Equipment
- Laminar hood is required for maintaining sterile conditions during tissue culture work.
- Controlled tissue culture room is required (generally maintained at about 25°C with 16/8 hour light and dark cycle).
- Shaker incubator and autoclave is required for bacterial culture preparation and sterilization of media.
- Centrifuge and spectrophotometer are required for bacterial culture handling and measurement of cell density.
- Vacuum pump is sometimes used to facilitate infiltration of Agrobacterium into plant tissues.
- Glassware such as beakers, cylinders, Petri dishes, Duran bottles and flasks are required for preparation and handling of media.
- Tissue culture vessels and sterile 50 ml plastic tubes are required for culturing explants and bacterial suspension.
- Forceps, scalpels and surgical blades are required for cutting and handling plant explants.
- Pipettes are used for transferring bacterial suspension and culture media.
- Filter paper and parafilm are required for sealing and handling culture plates.
Reagents and Biological Materials
- Plant explant (such as stem, embryo or cotyledon tissue) which acts as the target tissue for gene transfer.
- Agrobacterium strain containing the gene of interest is used as the vector organism.
- Sterilization agents such as 13% Sodium hypochlorite and 75% Ethanol are used for surface sterilization of explants.
- Sterile distilled water and 35% Hydrochloric acid are used during washing and preparation steps.
- Culture media components such as B5 medium, sucrose, agar, gellan gum powder, tryptone, yeast extract and sodium chloride are required for plant and bacterial growth.
- Plant growth regulator such as Abscisic acid is sometimes used during tissue culture stages.
- Antibiotics such as Rifampicin, Kanamycin monosulfate and Carbenicillin disodium salt are used for bacterial control and selection of transformed plants.
- PCR primer star Mix is used for molecular confirmation of gene transfer through PCR analysis.
This type of molecular analysis is commonly used for confirmation of gene transfer because amplification of the transferred DNA fragment can be detected using the PCR technique.
Media Preparation for Agrobacterium-mediated Gene Transfer
Some of the important media used in Agrobacterium-mediated gene transfer are as follows–
LB Medium (for Agrobacterium culture)
- Dissolve 5 g yeast extract, 10 g tryptone and 5 g sodium chloride in 1 liter of distilled water.
- The medium is sterilized by autoclaving before use.
- For preparing 50 ml of medium, 50 µl rifampicin stock solution (100 mg/ml) is added.
- 50 µl kanamycin stock solution (50 mg/ml) is also added to maintain selection of the bacterial strain.
Seed Germination Medium (Murashige and Skoog medium)
- Dissolve 4.43 g Murashige and Skoog (MS) basal medium powder in 1 liter distilled water.
- Add 3 g sucrose to the medium as a carbon source.
- Add 2.5 g gellan gum powder for solidification of the medium.
- The mixture is autoclaved and poured into sterile Petri plates.
- About 25 ml medium is poured into each plate under laminar air flow.
Co–cultivation Medium
- MS basal medium is used as the base medium.
- Add 750 µl BAP (2 mg/ml) to the medium.
- Add 500 µl ABA (2 mg/ml) for regulating plant cell growth.
- The medium is sterilized and about 25 ml is poured into sterile Petri plates.
Shooting Medium
- The co–cultivation medium is used as the base for shooting medium.
- Add 50 µl kanamycin (50 mg/ml) for selection of transformed plant cells.
- Add 2.5 ml carbenicillin (200 mg/ml) to eliminate Agrobacterium after transformation.
Rooting Medium
- Rooting medium is prepared from MS medium.
- Add 50 µl kanamycin (50 mg/ml) for selection of transformed plants.
- Add 1 ml carbenicillin (200 mg/ml) to suppress bacterial growth.
Agrobacterium-mediated Gene Transfer Protocol
- Sterilization and germination of seeds – The seeds are first sterilized using sterilizing agents such as chlorine gas, ethanol and sodium hypochlorite. The sterilized seeds are then placed on seed germination medium and incubated in dark conditions for about 2 days to obtain young seedlings.
- Preparation of Agrobacterium inoculum – A single colony of Agrobacterium containing the recombinant plasmid is inoculated into LB medium containing suitable antibiotics such as rifampicin and kanamycin. The culture is incubated in a shaker incubator at about 28°C until sufficient bacterial growth occurs.
- Preparation of explants – The germinated seeds are removed from the medium and placed on sterile Petri plates. The radicle and some parts of the cotyledons are removed using sterile forceps and scalpel. The detached cotyledons or other tissues are collected in MS liquid medium.
- Infection of explants – The prepared explants are immersed in Agrobacterium suspension for a short period. Sometimes vacuum infiltration or gentle shaking is used to improve bacterial infection of the plant tissues.
- Co–cultivation – The infected explants are transferred to co–cultivation medium and incubated for about 2–3 days at around 25–28°C. The plates are usually kept in dark conditions to facilitate T–DNA transfer from bacteria to plant cells.
- Elimination of Agrobacterium – After co–cultivation the explants are transferred to medium containing antibiotics such as carbenicillin or cefotaxime. These antibiotics suppress the growth of Agrobacterium and prevent bacterial overgrowth.
- Selection of transformed cells – The explants are then placed on selection medium containing selective antibiotics such as kanamycin. Only the plant cells that have successfully received and integrated the foreign gene survive and grow.
- Shoot initiation – The surviving explants are transferred to shooting medium containing suitable plant growth regulators. During this stage shoot formation begins and small shoots develop from transformed tissues.
- Root formation – The developed shoots are excised and transferred to rooting medium. In this medium the shoots develop roots and form complete plantlets.
- Plant acclimatization – The regenerated plantlets with developed roots are removed from culture vessels and washed carefully. The plants are transferred to pots containing moist soil and kept in controlled conditions for acclimatization.
- Molecular confirmation – Leaf samples of regenerated plants are analyzed by molecular techniques such as PCR in order to confirm the successful transfer and expression of the foreign gene.
Factors Affecting Agrobacterium-mediated Gene Transfer
Some of the important factors affecting Agrobacterium-mediated gene transfer are as follows–
- Explants – The type of plant tissue used as explant such as immature embryos, cotyledons, leaves or embryonic cultures affects the transformation efficiency. The developmental stage of explant also determines regeneration ability.
- Explant wounding – Wounding of plant tissue is necessary for successful transformation. It helps in releasing phenolic compounds from plant cells which attract the Agrobacterium.
- Plant species and genotype – Transformation efficiency depends on the plant species and genotype. Some species or cultivars are more responsive while others show low transformation frequency.
- Agrobacterium strains and vectors – The bacterial strain used such as EHA101, EHA105 or LBA4404 influences the efficiency of gene transfer. The type of vector system also affects T–DNA delivery.
- Co–cultivation conditions – The concentration of bacterial suspension, infection time and duration of co–cultivation plays an important role in successful T–DNA transfer.
- Temperature – Temperature affects the activity of bacterial virulence genes. The optimal temperature for T–DNA transfer is generally between 19°C to 22°C.
- Light conditions – Light exposure can influence plant hormone levels and phenolic compound production. In many protocols co–cultivation is carried out under dark conditions.
- Chemical inducers and culture media – Phenolic compounds such as acetosyringone are added to culture media to activate vir genes of the bacterium. The composition of culture medium also affects transformation.
- Plant growth regulators (PGRs) – Plant hormones such as auxins and cytokinins are required in culture media for cell division, callus formation and regeneration of transformed plants.
- Antibiotics – Antibiotics such as carbenicillin, cefotaxime or timentin are used after co–cultivation to eliminate Agrobacterium from plant tissues.
- Host defense response – Plant defense mechanisms such as production of reactive oxygen species and other signaling molecules may inhibit bacterial infection and reduce transformation efficiency.
Applications of Agrobacterium-mediated Gene Transfer
The important applications of Agrobacterium-mediated gene transfer are as follows–
- Crop Improvement and Yield Enhancement – It is used for modifying plant biosynthetic pathways and shelf-life of crops. This modification helps in increasing the overall crop yield and improving agricultural productivity.
- Biotic and Abiotic Stress Tolerance –It is used to develop plants that can tolerate environmental stress conditions like drought, salinity and temperature changes. Insect-resistant plants containing Bt toxin genes are also developed which reduce the use of harmful agrochemicals.
- Genome Editing and Targeted Mutagenesis – It is used for delivering genome editing tools such as CRISPR/Cas9, TALENs and Zinc Finger Nucleases (ZFNs). These tools allow knock-in, knock-down and knock-out of specific genes in plant genome.
- Functional Genomics and Gene Discovery – It is used as an important tool in functional genomics studies. In this method T-DNA tagging acts as a mutagen which helps in isolating novel plant genes and studying promoter activity and protein functions.
- Production of Biopharmaceuticals – Plants can be genetically modified to produce pharmaceutical proteins. These plants act as living factories for production of products such as interferons, vaccines, monoclonal antibodies and human growth factors.
- Biosynthesis of Complex Metabolites – It is used to reconstruct metabolic pathways in plants for production of valuable secondary metabolites. These include compounds such as alkaloids, terpenoids and antimalarial compound artemisinin.
- Gene Silencing Studies – Agrobacterium transformation is used for transient gene expression methods like agroinfiltration. It helps in expression of small interfering RNAs (siRNAs) and microRNAs (miRNAs) for gene silencing and functional analysis.
- Environmental Biomonitoring and Bioremediation – Transgenic plants can be produced which act as biomonitors to detect toxic environmental compounds. These plants are also used to detoxify contaminated soil and water.
- Transformation of Non-Plant Organisms – This method is not limited only to plants. It is also used for genetic transformation of organisms like fungi, yeast, algae and some animal cells for advanced biological research.
Limitations of Agrobacterium-mediated Gene Transfer
The major limitations of Agrobacterium-mediated gene transfer are as follows–
- Genotype and Species Dependence –This method shows limited host range. Many crop species, monocots, legumes and woody plants are difficult to transform and remain recalcitrant.
- Dependence on Tissue Culture –The process largely depends on plant tissue culture and regeneration techniques. These procedures are time-consuming, labour-intensive and may produce in vitro variations in regenerated plants.
- Explant Availability Problem – In many monocot plants immature embryos are used as explants for transformation. These embryos are available only for a short period during the year.
- Random Integration of T-DNA – The T-DNA integration in plant genome occurs randomly. Because of this, essential plant genes may be disrupted and transgene expression may become unpredictable.
- Bacterial Overgrowth – Sometimes Agrobacterium cells grow excessively after co-cultivation. This uncontrolled bacterial growth can damage or kill the plant explants.
- Antibiotic Cost and Toxicity – Specific antibiotics such as carbenicillin and cefotaxime are required to eliminate the bacteria. These antibiotics are expensive and sometimes toxic to plant tissues.
- False Positive Results – Some Agrobacterium cells may survive antibiotic selection and remain associated with plant tissues. This may produce false positive results during molecular screening.
- Limited Delivery of Molecules – This method cannot directly transfer linear DNA fragments, RNA or proteins. The gene of interest must be cloned into a T-DNA region of a binary vector before transformation.
- Difficulty in Site-Specific Integration – T-DNA is transferred into plant cell as a single-stranded molecule. Because of this, site-specific integration systems such as Cre-lox are difficult to perform.
- Requirement of Specialized Facilities – The method requires sterile culture rooms and biosafety laboratories for handling genetically modified organisms and bacterial pathogens.
- Unknown Plant Cellular Factors – Although the bacterial mechanism of T-DNA transfer is well studied, many plant cellular factors that influence transformation efficiency are still not completely understood.
- Agrawal, S., & Rami, E. A. (2022). A review: Agrobacterium-mediated gene transformation to increase plant productivity. The Journal of Phytopharmacology, 11(2), 111–117. https://doi.org/10.31254/phyto.2022.11211
- Aliu, E., Ji, Q., Wlazlo, A., Grosic, S., Azanu, M. K., Wang, K., & Lee, K. (2024). Enhancing Agrobacterium-mediated plant transformation efficiency through improved ternary vector systems and auxotrophic strains. Frontiers in Plant Science, 15, 1429353. https://doi.org/10.3389/fpls.2024.1429353
- Alvarez, J. M., & Ordás, R. J. (2013). Stable Agrobacterium-mediated transformation of maritime pine based on kanamycin selection. The Scientific World Journal, 2013, 681792. https://doi.org/10.1155/2013/681792
- Anand, A., Bass, S. H., Wu, E., Wang, N., McBride, K. E., Annaluru, N., Miller, M., Hua, M., & Jones, T. J. (2018). An improved ternary vector system for Agrobacterium-mediated rapid maize transformation. Plant Molecular Biology, 97(1), 187–200. https://doi.org/10.1007/s11103-018-0732-y
- Azizi-Dargahlou, S., & Pouresmaeil, M. (2023). Agrobacterium tumefaciens-mediated plant transformation: A review. Molecular Biotechnology, 66(7), 1563–1580. https://doi.org/10.1007/s12033-023-00788-x
- Chakraborty, M., Reddy, P. S., Narasu, M. L., Krishna, G., & Rana, D. (2016). Agrobacterium-mediated genetic transformation of commercially elite rice restorer line using nptII gene as a plant selection marker. Physiology and Molecular Biology of Plants, 22(1), 51–60. https://doi.org/10.1007/s12298-015-0334-y
- Christie, P. J., & Gordon, J. E. (2014). The Agrobacterium Ti plasmids. Microbiology Spectrum, 2(6). https://doi.org/10.1128/microbiolspec.PLAS-0010-2013
- de Vetten, N., Wolters, A.-M., Raemakers, K., van der Meer, I., ter Stege, R., Heeres, E., Heeres, P., & Visser, R. (2003). A transformation method for obtaining marker-free plants of a cross-pollinating and vegetatively propagated crop. Nature Biotechnology, 21(4), 439–442. https://doi.org/10.1038/nbt801
- Fister, A. S., Landherr, L., Maximova, S. N., & Guiltinan, M. J. (2018). Transient expression of CRISPR/Cas9 machinery targeting TcNPR3 enhances defense response in Theobroma cacao. Frontiers in Plant Science, 9, 268. https://doi.org/10.3389/fpls.2018.00268
- Gallego, A. (n.d.). Deep dive into plant transformation: Protoplast-mediated, biolistic-mediated and Agrobacterium-mediated gene transfer. GoldBio.
- Gayathri, M. K. (2026). Agrobacterium mediated genetic transformation efficiency in model plants. Journal for Research in Applied Sciences and Biotechnology, 5(1), 92–99. https://doi.org/10.55544/jrasb.5.1.9
- Gelvin, S. B. (2003). Agrobacterium-mediated plant transformation: The biology behind the “gene-jockeying” tool. Microbiology and Molecular Biology Reviews, 67(1), 16–37. https://doi.org/10.1128/MMBR.67.1.16-37.2003
- Goralogia, G. S., Willig, C., & Strauss, S. H. (2025). Engineering Agrobacterium for improved plant transformation. The Plant Journal, 121(5), e70015. https://doi.org/10.1111/tpj.70015
- Hiei, Y., Ishida, Y., & Komari, T. (2014). Progress of cereal transformation technology mediated by Agrobacterium tumefaciens. Frontiers in Plant Science, 5, 628. https://doi.org/10.3389/fpls.2014.00628
- Hwang, H.-H., Yu, M., & Lai, E.-M. (2017). Agrobacterium-mediated plant transformation: Biology and applications. The Arabidopsis Book, 15, e0186. https://doi.org/10.1199/tab.0186
- International Service for the Acquisition of Agri-biotech Applications (ISAAA). (n.d.). Pocket K No. 36: Marker-free GM plants.
- Jeong, J.-H., Jeon, E.-Y., Hwang, M. K., Song, Y. J., & Kim, J.-Y. (2024). Development of super-infective ternary vector systems for enhancing the Agrobacterium-mediated plant transformation and genome editing efficiency. Horticulture Research, 11(9), uhae187. https://doi.org/10.1093/hr/uhae187
- Jones, H. D., Doherty, A., & Wu, H. (2005). Review of methodologies and a protocol for the Agrobacterium-mediated transformation of wheat. Plant Methods, 1, 5. https://doi.org/10.1186/1746-4811-1-5
- Kambampati, S., Verma, P. K., & Janga, M. R. (2025). Plant transformation and genome editing for precise synthetic biology applications. SynBio, 3(3), 9. https://doi.org/10.3390/synbio3030009
- Kaur, M., Manchanda, P., Kalia, A., Ahmed, F. K., Nepovimova, E., Kuca, K., & Abd-Elsalam, K. A. (2021). Agroinfiltration mediated scalable transient gene expression in genome edited crop plants. International Journal of Molecular Sciences, 22(19), 10882. https://doi.org/10.3390/ijms221910882
- Kowalik, S., Samoń, M., & Przyborowski, M. (2026). Molecular regulators of in vitro regeneration in wheat: Roles of morphogenic factors in transformation, genome editing, and breeding. International Journal of Molecular Sciences, 27(3), 1271. https://doi.org/10.3390/ijms27031271
- Lacroix, B., & Citovsky, V. (2020). Biolistic approach for transient gene expression studies in plants. Methods in Molecular Biology, 2124, 125–139. https://doi.org/10.1007/978-1-0716-0356-7_6
- Liu, S., Wang, K., Geng, S., Hossain, M., Ye, X., Li, A., Mao, L., & Kogel, K.-H. (2024). Enemies at peace: Recent progress in Agrobacterium-mediated cereal transformation. The Crop Journal. https://doi.org/10.1016/j.cj.2023.12.009
- Magori, S., & Citovsky, V. (2011). Agrobacterium counteracts host-induced degradation of its effector F-box protein. Science Signaling, 4(195), ra69. https://doi.org/10.1126/scisignal.2002124
- Magori, S., & Citovsky, V. (2011). Epigenetic control of Agrobacterium T-DNA integration. Biochimica et Biophysica Acta (BBA), 1809(8), 388–394. https://doi.org/10.1016/j.bbagrm.2011.01.007
- Mohana, S., Mohammed, R. A., & Narasimha, G. (2025). Agrobacterium-mediated genetic transformation in crop plants. Methods in Molecular Biology, 2973, 275–281. https://doi.org/10.1007/978-1-0716-4803-2_25
- Mookkan, M., Nelson-Vasilchik, K., Hague, J., Zhang, Z. J., & Kausch, A. P. (2017). Selectable marker independent transformation of recalcitrant maize inbred B73 and sorghum P898012 mediated by morphogenic regulators BABY BOOM and WUSCHEL2. Plant Cell Reports, 36(9), 1477–1491. https://doi.org/10.1007/s00299-017-2169-1
- Nelson, A., Ranney, T., Liu, W., Kelliher, T., Duan, H., & Da, K. (2025). Overcoming recalcitrance: A review of regeneration methods and challenges in roses. Plants, 14(24), 3797. https://doi.org/10.3390/plants14243797
- Nester, E. W. (2015). Agrobacterium: Nature’s genetic engineer. Frontiers in Plant Science, 5, 730. https://doi.org/10.3389/fpls.2014.00730
- Ouyang, C., Liu, W., Chen, S., Zhao, H., Chen, X., Jin, X., Li, X., Wu, Y., Zeng, X., Huang, P., et al. (2024). Highly efficient Agrobacterium tumefaciens mediated transformation of oil palm using an EPSPS-glyphosate selection system. Plants, 13(23), 3343. https://doi.org/10.3390/plants13233343
- Pitzschke, A. (2013). Agrobacterium infection and plant defense—transformation success hangs by a thread. Frontiers in Plant Science, 4, 519. https://doi.org/10.3389/fpls.2013.00519
- Polowick, P. L., & Yan, W. (2023). A protocol for Agrobacterium-mediated genetic transformation of Lens culinaris Medik (lentil). Plant Cell, Tissue and Organ Culture, 152, 605–618. https://doi.org/10.1007/s11240-022-02434-x
- Pratiwi, R. A., & Surya, M. I. (2020). Agrobacterium-mediated transformation. In K.-Y. To (Ed.), Genetic Transformation in Crops. IntechOpen. https://doi.org/10.5772/intechopen.91132
- Schmitz, D. J., Ali, Z., Wang, C., Aljedaani, F., Hooykaas, P. J. J., Mahfouz, M. M., & de Pater, S. (2020). CRISPR/Cas9 mutagenesis by translocation of Cas9 protein into plant cells via the Agrobacterium Type IV secretion system. Frontiers in Genome Editing, 2, 6. https://doi.org/10.3389/fgeed.2020.00006
- Thole, V., Worland, B., Snape, J. W., & Vain, P. (2007). The pCLEAN dual binary vector system for Agrobacterium-mediated plant transformation. Plant Physiology, 145(4), 1211–1219. https://doi.org/10.1104/pp.107.108563
- Verweire, D., Verleyen, K., De Buck, S., Claeys, M., & Angenon, G. (2007). Marker-free transgenic plants through genetically programmed auto-excision. Plant Physiology, 145(4), 1220–1231. https://doi.org/10.1104/pp.107.106526
- Wang, G.-P., Yu, X.-D., Sun, Y.-W., Jones, H. D., & Xia, L.-Q. (2016). Generation of marker- and/or backbone-free transgenic wheat plants via Agrobacterium-mediated transformation. Frontiers in Plant Science, 7, 1324. https://doi.org/10.3389/fpls.2016.01324
- Wu, E., Lenderts, B., Glassman, K., Berezowska-Kaniewska, M., Christensen, H., Asmus, T., Zhen, S., Chu, U., Cho, M.-J., & Zhao, Z.-Y. (2014). Optimized Agrobacterium-mediated sorghum transformation protocol and molecular data of transgenic sorghum plants. In Vitro Cellular & Developmental Biology – Plant, 50(1), 9–18. https://doi.org/10.1007/s11627-013-9583-z
- Zhang, L., Meng, S., Liu, Y., Han, F., Xu, T., Zhao, Z., & Li, Z. (2024). Advances in and perspectives on transgenic technology and CRISPR-Cas9 gene editing in broccoli. Genes, 15(6), 668. https://doi.org/10.3390/genes15060668
- Zhang, Q., Zhang, Y., Lu, M.-H., Chai, Y.-P., Jiang, Y.-Y., Zhou, Y., Wang, X.-C., & Chen, Q.-J. (2019). A novel ternary vector system united with morphogenic genes enhances CRISPR/Cas delivery in maize. Plant Physiology, 181(4), 1441–1448. https://doi.org/10.1104/pp.19.00767
- Zhang, Z., Li, X., Ma, S., Shan, N., Zhang, X., & Sui, X. (2017). A protocol for Agrobacterium-mediated transformation of cucumber (Cucumis sativus L.) from cotyledon explants V.1. Protocol Exchange. https://doi.org/10.1038/protex.2017.107