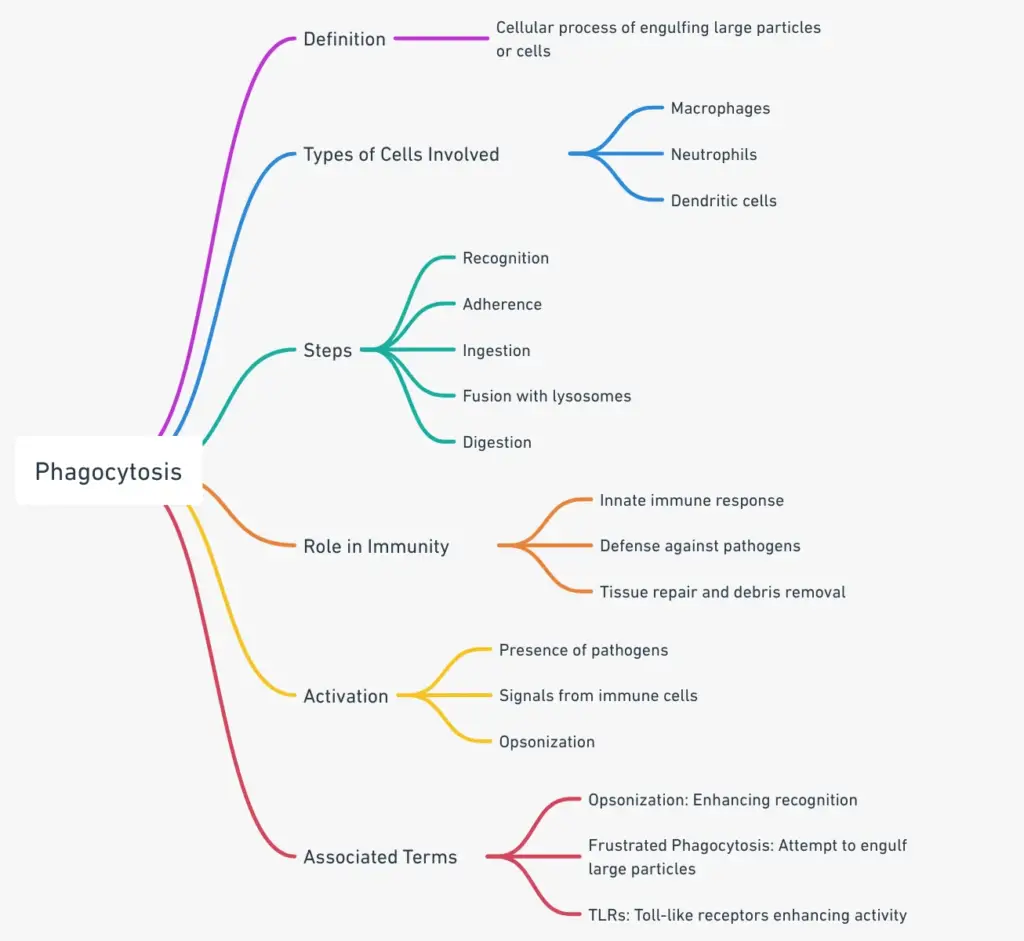
What is phagocytosis?
- Phagocytosis, derived from the Greek words “phago-” (devouring) and “-cyte” (cell), is a fundamental cellular process where cells engulf large particles, typically greater than 0.5 micrometers, into specialized compartments known as phagosomes. This process is pivotal for various cellular functions, ranging from the removal of cellular debris to the defense against pathogens.
- In the immune system, phagocytosis plays a crucial role in the body’s defense mechanism. Immune cells, such as macrophages and neutrophils, utilize this process to eliminate foreign invaders like bacteria, viruses, and other harmful particles. The engulfed particles are then directed to lysosomes, where they undergo enzymatic degradation, ensuring the intruder’s neutralization.
- The efficiency of phagocytosis is significantly augmented by a process called opsonization. In opsonization, the target particle, such as a bacterium, is coated with specific molecules like antibodies (IgG) or complement proteins (e.g., C3). This coating enhances the recognition and binding of the particle to phagocytic cells. Complement receptors, primarily CR1 and CR3, are the dominant receptors facilitating opsonization by the complement system. Furthermore, Polymorphonuclear Leukocytes (PMNs) express receptors for the Fc fragment of IgG, known as FcγRs. The predominant Fcγ receptors present on circulating PMNs are FcγRII (CD32) and FcγIII (CD16). The engagement of these receptors initiates an oxidative burst within PMNs, a rapid release of reactive oxygen species that aids in the destruction of the engulfed particle.
- The intricate interplay between antibodies, complement proteins, and their respective receptors on phagocytic cells ensures the efficient elimination of harmful entities. Once these receptors are engaged on the PMN surface, they activate a series of intracellular signaling pathways, culminating in the engulfment and eventual degradation of the target particle.
- Recent research has further elucidated the molecular mechanisms underlying phagocytosis. For instance, studies have highlighted the role of actin polymerization in the formation of pseudopods, cellular extensions that envelop the target particle. Additionally, the importance of specific signaling molecules, such as phosphoinositides and Rho-family GTPases, in orchestrating the phagocytic process has been emphasized.
- In conclusion, phagocytosis is a vital cellular process with implications in health and disease. Its efficient functioning ensures the body’s defense against foreign invaders and maintains cellular homeostasis by removing debris. Understanding the intricacies of this process can pave the way for therapeutic interventions in conditions where phagocytosis is compromised or overly activated.
Phagocytosis Definition
Phagocytosis is the cellular process where cells engulf and digest large particles or microorganisms, often for defense against pathogens or to remove cellular debris.
How does phagocytosis occurs?
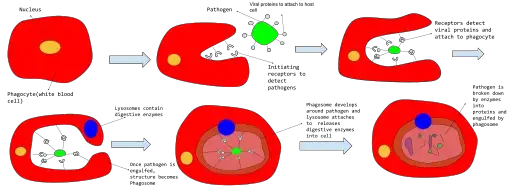
To successfully phagocytize a substance, cells must complete certain steps. Let’s take an example of a macrophage, a type immune cell, phagocyting a virus. To make this easier to understand, let’s imagine that we are following it. However, there are many types of cells that perform phagocytosis.
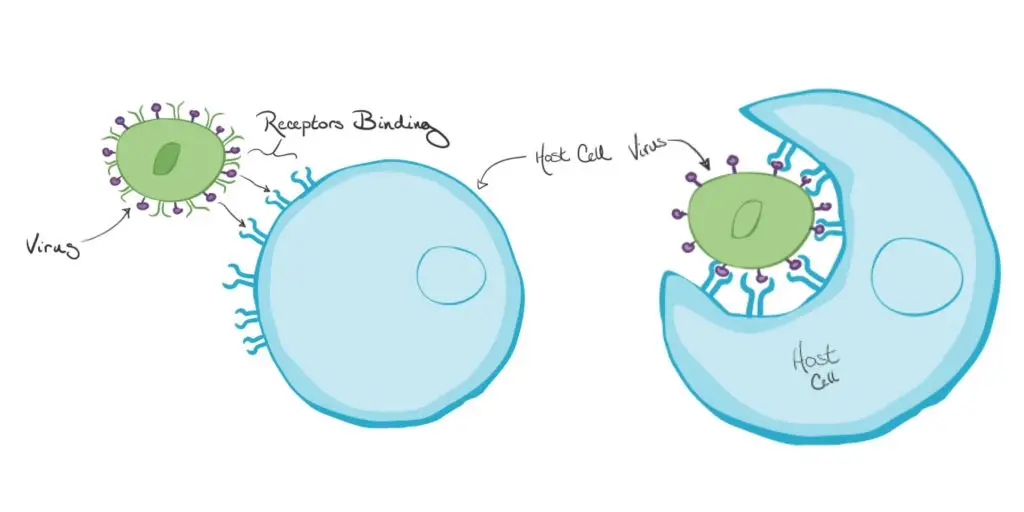
- Both the virus and the cell must come in contact. Sometimes, an immune cell accidentally encounters a virus in bloodstream. Sometimes cells can move through a process called “chemotaxis”. Chemotaxis is the movement of an organism/cell in response to chemical stimuli. Cytokines are small proteins that are used for cell signaling. Many immune system cells respond to them. Cytokines tell cells to move to the area where the virus (in this case, the particle) is located. This is common for infections that are specific to one area, such as a wound infected by bacteria.
- The virus binds with the macrophage’s cell surface receptors. Different cell types have different receptors. Some receptors can be generalized, meaning they can identify self-produced molecules versus potential threats (and that’s it). Others are more specific, such as toll-like receptors and antibodies. Without successful binding to the cell surface receptors, the macrophage cannot initiate phagocytosis. Some viruses can have specific surface receptors that are only available to macrophages. In order to infect a host cell, viruses must access its cytoplasm and nucleus. To do this they use their surface receptors which interact with immune cells to enter the cell. Sometimes, a virus can be successfully destroyed by a host cell and the infection is stopped. Sometimes, the virus can trick the host cell into engulfing the virus and gaining access to the necessary resources to reproduce. The immune system recognizes the infected cells and destroys them. This stops viral replication.
- The macrophage surrounds the virus and then engulfs it in the cell. Instead of moving large items across the plasma membrane which could permanently damage it, phagocytosis uses extensions from the cytoplasm (pseudopods), to surround the particle and seal it in a membrane. The virus and macrophage are bound to the cell’s surface. Once both pseudopods are visible, the virus will be enclosed. Remember that cells are flexible and fluid. The virus and macrophage are bound to the cell’s surface in our viral example. The virus is pulled inward by the cell, creating a pocket-like depression without causing damage to the plasma membrane. Remember that cells are flexible and fluid.
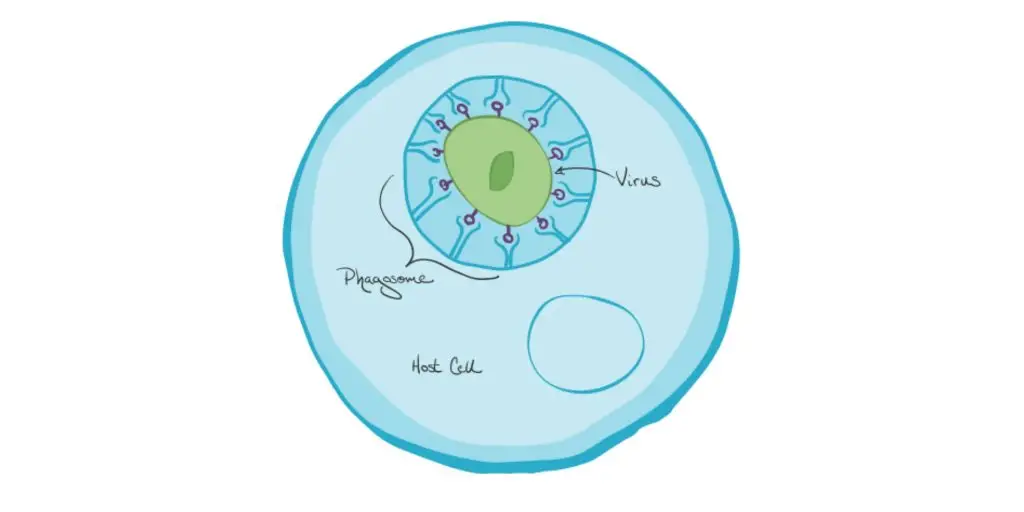
- The virus is enclosed within a bubble-like structure called a “phagosome” within the cytoplasm. The pseudopodsIn’s extensions create a pocket. They extend their lips towards one another to close the gap. This creates a pocket, where the plasma membrane moves around the particle to encase it inside the cell.
- The phagosome merges with a Lysosome to become a “phagolysosome”. Lysosomes can also be bubble-like structures similar to phagosomes and are responsible for removing waste from the cell. Lysis is a Greek word that means “to dissolve”, which makes it simple to recall the purpose of a lysosome. The phagosome would not be able to do any work with the contents of the container if it didn’t fuse with a Lysosome.
- Phagolysosome reduces pH to degrade its contents. The pH of the phagolysosome’s internal environment is drastically decreased by a lysosome, or phagolysosome. The phagolysosome’s pH is reduced, making it more acidic. This is a very effective method of neutralizing or killing any organisms within the phagolysosome to prevent them from infecting their cells. Some viruses use the lower pH to escape thephagolysosome, and then start reproducing inside the cells. Influenza (flu virus) activates a conformational change that allows it to escape to the cytoplasm.
- After the contents are neutralized, the residual body of the phagolysosome contains the waste products. The cell eventually expels the residual body.
Phagocytosis and the immune system
Phagocytosis plays a vital role in the immune system. Phagocytosis is performed by several types of immune system cells: macrophages and neutrophils as well as B lymphocytes and dendritic cell. By phagocyting foreign or pathogenic particles, cells of the immune systems can identify what they are fighting. The immune system can target specific particles that are circulating in the body by knowing the enemy.
The immune system also uses phagocytosis to infect and destroy pathogens, such as bacteria and viruses. The immune system reduces the speed at which the infection can spread by destroying infected cells. As we mentioned, the phagolysosome creates a acidic environment that can destroy or neutralize the contents. Other mechanisms can be used by the immune system cells to destroy pathogens within the phagolysosome.
- Oxygen Radicals: Oxygen radicals can be highly reactive molecules that react to proteins, lipids, and other biological molecules. The amount of oxygen radicals within a cell can rise dramatically during physiological stress. This can lead to oxidative stress which can cause cell structure destruction.
- Nitric Oxide: This reactive substance is similar to oxygen radicals. It reacts with superoxide and creates additional molecules that cause damage to various biological molecules.
- Antimicrobial Proteins: Antimicrobial Proteins are proteins that specifically kill or damage bacteria. Antimicrobial proteins include proteases that kill bacteria by destroying essential protein and lysozyme which targets the cell walls of Gram positive bacteria.
- Antimicrobial Peptides: Antimicrobial peptides work in the same way as antimicrobial proteins to kill and attack bacteria. Defensins and other antimicrobial peptides attack the membranes of bacterial cells.
- Binding Proteins: Binding proteins play an important role in the innate immune response system. They competely bind to proteins and ions that could otherwise be beneficial to bacteria or viral replication. Lactoferrin is a binding protein that can be found in mucosal membranes. It binds ironions which are essential for the growth of bacteria.
The types of cells in the immune system that engulf microorganisms via phagocytosis
- Neutrophils: Neutrophils, which are plentiful in the blood, enter quickly into tissues and phagocytize pathogens during acute inflammation.
- Macrophages: Macrophages and monocytes are closely related. Chronic inflammation is more common with these longer-lived cells. They also release important inflammatory paracrine.
- Dendritic Cells: Phagocytosis is essential for the creation of an immune response, rather than directly attacking pathogens.
- B Lymphocytes: Sometimes, a small amount of phagocytosis is necessary for these cells to become cells that produce antibodies.
There are two types of killing mechanisms for PMNs. Both are oxygen-dependent, while the other is not. The production of superoxide radicals, and other reactive oxygen species is stimulated by phagocytosis. These powerful microbicidal agents include hydrogen peroxide, chloramines, and others. NADPH oxidase, which is located in the cell membranes of the PMNs, generates superoxide (known as the respiratory burst). Superoxide is unstable, and it quickly dissolves to hydrogen peroxide or other substances.
These are microbicidal. These reactions occur inside the phagolysosome, also known as the phagosome.
How Does Phagocytosis Work?
Phagocytosis is a sophisticated cellular process that encompasses the engulfment and subsequent degradation of microorganisms. This mechanism is vital for the immune system’s defense against pathogens. Herein, we delve into the intricate steps and factors involved in the destruction of microbes within the phagolysosome.
- Generation of Reactive Oxygen Species (ROS): Within the phagolysosome membrane, a protein complex known as phagocyte oxidase plays a pivotal role in producing oxygen radicals. This is achieved by transferring a single electron from NADPH to oxygen, resulting in its partial reduction. The ensuing reactive molecules, due to their high reactivity, interact with various biological entities, including proteins and lipids.
- Role of Nitric Oxide: Nitric oxide, synthesized by nitric oxide synthase, is another reactive agent. It combines with superoxide, leading to the formation of molecules that inflict damage on a range of biological compounds.
- Anti-Microbial Arsenal: The phagolysosome is equipped with an array of antimicrobial proteins. Notably, lysosomes house proteases like elastase, which is pivotal for neutralizing several bacteria. Lysozyme, another antimicrobial protein, targets the cell walls of specific gram-positive bacteria.
- Peptide Defense: Defensins and other peptides are adept at targeting bacterial cell membranes. Analogous molecules are ubiquitous across the animal kingdom.
- Binding Proteins in Action: Lactoferrin, by binding iron ions, deprives bacteria of essential growth factors. Concurrently, another protein sequesters vitamin B12.
- Acidification of Phagolysosome: The phagolysosome environment is acidified by hydrogen ion transporters, a process crucial for microorganism destruction and the optimal function of the aforementioned proteases.
Beyond the direct annihilation of pathogens, phagocytes excrete molecules that influence neighboring cells, orchestrating a collective response to infections. These regulatory entities, termed cytokines, predominantly emanate from white blood cells and their kin, such as macrophages.
Several immune system cells employ phagocytosis to engulf pathogens:
- Neutrophils: Prevalent in blood, they swiftly migrate to tissues, playing a central role in acute inflammation by phagocytizing pathogens.
- Macrophages: Evolutionarily related to monocytes, these enduring cells are predominant in chronic inflammation and secrete vital inflammatory paracrines.
- Dendritic Cells: Their phagocytic activity is pivotal for orchestrating a specific immune response rather than direct pathogen destruction.
- B Lymphocytes: Limited phagocytosis in these cells often precedes their transformation into antibody-releasing cells.
Polymorphonuclear Leukocytes (PMNs) employ two primary killing strategies: oxygen-dependent and oxygen-independent. Microbe phagocytosis triggers the synthesis of superoxide radicals and other ROS. Among these, hydrogen peroxide and chloramines stand out as potent microbicidal agents. The enzyme NADPH oxidase, located in the PMN cell membrane, is responsible for superoxide generation, known as the respiratory burst. This superoxide, due to its instability, rapidly transforms into hydrogen peroxide and other microbicidal substances, all of which operate within the phagolysosome.
Steps of Phagocytosis
Phagocytosis is a fundamental cellular mechanism that facilitates the engulfment and degradation of particles, including pathogens. This process is vital for maintaining cellular homeostasis and defending against foreign invaders. Here, we elucidate the sequential steps involved in phagocytosis.
- Cell Activation: The initiation of phagocytosis begins with the activation of the phagocytic cell. This cell could be a phagocyte, an integral component of the immune system, or an organism like an amoeba that exhibits phagocytic behavior. For immune cells, activation is triggered upon proximity to bacterial entities or their fragments. Specific receptors on the phagocyte’s surface recognize and bind to these bacterial components, prompting a cellular response.
- Chemotaxis: Within the immune framework, a phenomenon known as chemotaxis may ensue. Chemotaxis refers to the directed movement of phagocytes towards a gradient of specific molecules. These immune cells detect chemical cues and navigate towards the source, which could be invading bacteria or damaged cellular components.
- Particle Attachment: The phagocyte then establishes a connection with the target particle, a prerequisite for the subsequent engulfment. Certain bacteria have evolved mechanisms to evade this attachment, thereby hindering their internalization and subsequent degradation.
- Particle Ingestion: Upon successful attachment, the phagocyte engulfs the particle. This engulfed entity is encased within a vesicle, termed a phagosome. This vesicular structure facilitates the transport of the ingested particle deeper into the cell.
- Digestion via Lysosomal Fusion: The phagosome undergoes fusion with a lysosome, a vesicle laden with hydrolytic enzymes. These enzymes are adept at molecular breakdown. The resultant structure post-fusion is termed a phagolysosome, within which the particle undergoes enzymatic degradation.
- Exocytosis and Waste Expulsion: Post-digestion, the cell is left with residual waste, comprising degraded molecules that lack utility for the cell. These waste products are packaged into vesicles and transported to the cell’s periphery. Through a process known as exocytosis, these vesicles merge with the cell membrane, releasing their contents outside the cell, thereby completing the phagocytic cycle.
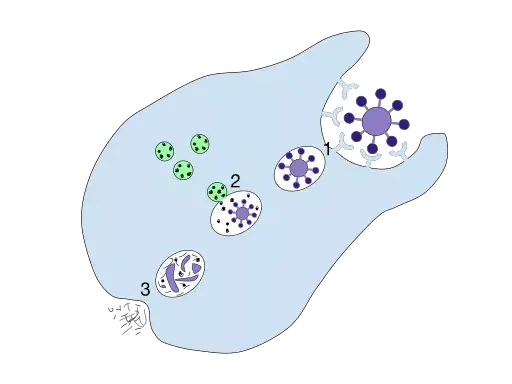
Example: Macrophage phagocytizing a virus.
- Both the virus and the cell must come in contact.
- The virus binds to macrophage cell surface receptors.
- The virus is infected by the macrophage, which then engulfs it in a pocket.
- The virus invaginates and is enclosed within a bubble-like structure called a “phagosome” in the cytoplasm.
- The phagosome merges with a Lysosome to become a “phagolysosome”.
- Phagolysosome reduces pH to degrade its contents
- After the contents are neutralized, the residual body of the phagolysosome contains the waste products.
- The cell eventually expels the remaining body.
Recognition of Foreign particles for Phagocytosis
Professional phagocytes (neutrophils and macrophages), as well as other leukocytes, can detect microbial macromolecules through the presence of repetitive structural patterns. These unique microbial signatures can be called microbe-associated molecular pattern (MAMPs), but they can also be called pathogen-associated molecular pattern (PAMPs).
Because the immune system can respond to microorganisms other than pathogens, we use MAMP. MAMPs refer to specific areas within common microbial macromolecules such as lipopolysaccharide, peptidoglycan and fungal cell wall components. They also contain viral nucleic acid and other microbial structures. MAMPs don’t identify any microorganism, but they alert the host to the presence of a microbe and sound the alarm for potential infection.
Extracellular microbial invaders, such as most fungi, many bacteria and many bacteria, can be detected by cells, while intracellular pathogens, such as viruses, bacteria and a few fungal species, can be detected. The innate immune system’s cells recognize extracellular infectious agents by recognizing receptors on the surface of these agents, while intracellular pathogens can be detected in the cytosol by host receptors. Pattern recognition receptors (PRRs) are all receptors that recognize MAMPs regardless of their cellular location.
We have already introduced soluble PRRs. They include the acute-phase protein, the mannose-binding proteins (of the lectin supplement pathway), and the C-reactive protein. The membrane-bound PRRs can be found in phagocytic cell membranes, which allow them to degrade and ingest the MAMP source. These receptors are crucial in distinguishing self from non-self. Let’s take a closer look at them.
1. C-Type Lectin Receptors
C-type lectin regulators (CLRs), a large class of calciumdependent membrane-bound protein, have one or more domains that bind to one type of carbohydrate (lectin), found on MAMPs. Mannose, fucose and glucan carbohydrates are some ligands found on bacteria (Mycobacterium tuberculosis, Helicobacter Pylori), fungi, helminths, and some viruses. CLRs can also bind N-acetylglucosamine and capsular polysaccharides from Streptococcus pneumoniae, Klebsiella pneumoniae. CLRs are concentrated in macrophages, dendritic and dendritic cell populations. A signaling cascade that triggers cytokine gene transcription is initiated when a CLR located on the surface of a Phagocyte binds to a MAMP. The CLR cytoplasmic domain, also known as the immunoreceptor-tyrosine based activation motif (ITAM), is responsible for this. Binding can also lead to receptor-MAMP degradation and internalization.
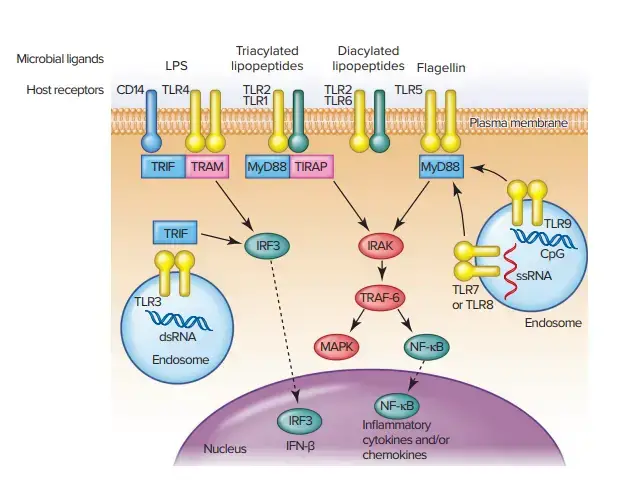
2. Toll-Like Receptors
TLRs, a class of transmembrane receivers, are important for innate immunity. They are expressed by many cells, including macrophages and dermis cells. They can be found attached to plasma membranes as well as membranes of endosomes and lysosomes. These receptors detect MAMPs that have gotten into the host cell’s cytosol. TLRs contain an extracellular (or extraorganelle) domain that binds MAMPs and a cytoplasmic Toll/interleukin-1 (TIR) domain which activates downstream transcription. The binding of a MAMP and a TLR triggers the TIR to recruit a particular adapter protein, MyD88 or TRIF. This activates a signal transmission cascade that leads to activation of transcription factors necessary for the expression of genes encoding cytokines. For example, activation via the MyD88-dependent adaptationer activates the transcription factor NFkB which activates transcription pro-inflammatory cytokine gene genes. There are at most 10 distinct human TLRs, which recognize different but often overlapping MAMPS to initiate an immune response.
3. NOD-Like Receptors
Contrary to transmembranous TLRs nucleotide binding and oligomerizationdomain (NOD),-like receptors can only be found in the cytosol of the host cell. NOD-like receptors can sense intracellular MAMPs, such as viral RNA, bacterial toxins and bacteria, along with host molecules known as “damage-associated molecular pattern” (DAMPs). These include uric acid, heat-shock protein, and uric acid. DAMPs refer to a variety of molecules that are caused by cell injury or stress. Once one of these signals is detected, the oligomerization occurs of several NLRs. Next, cofactors and enzyme caspase-1 can be recruited to the oligomerized units. The NLR-caspase complex, also known as the inflammasome, triggers pro-inflammatory cytokines. This happens when caspase-1 cleaves, activating the cytokines IL-1, and IL-18. These cytokines are also made in response to signals from other PRRs such as TLRs. In other words, NLRs (inside host cell cells) and TLRs on the surface of host cells and some intracellular membranes work together to produce a strong inflammatory response.
4. RIG-I-Like Receptors
Retinoic-acid-inducible gene I (RIG-I)-like receptors (RLRs) are yet another family of cytoplasmic receptors. These receptors were designed to sense viral DNA. The cytosol is home to RNA genomes that are replicated by viruses. This leads to the creation of dsRNA as well as triphosphate-capped, ssRNA. These forms of virus-specificRNA are recognized by RLRs and stimulate an antiviral response.
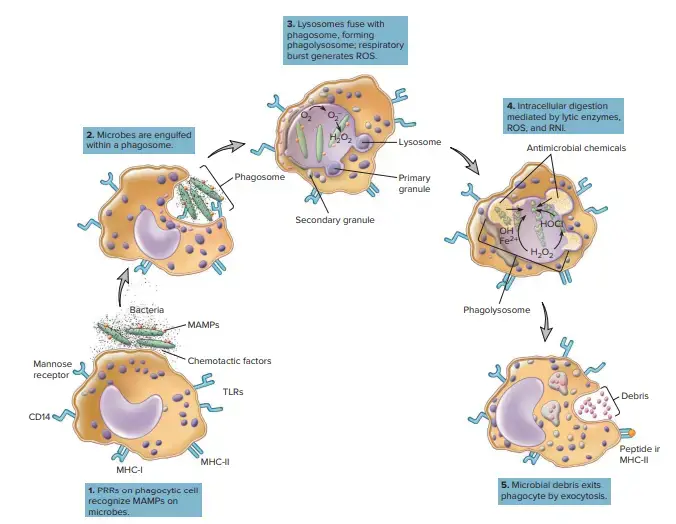
Intracellular Digestion of particles
Ingestion and digestion of foreign substances can be accomplished by two parallel processes: phagocytosis (Latin pseudo, false) and autophagy (Latin pseudo, foot). Extracellular particles are engulfed in pseudopodia (Latin pseudo; false; podium, foot), which form by the extension and contraction of the cell membrane. These pseudopodia surround the particle to create an internal phagosome. This can be receptor- or opsonin-mediated. When intracellular microorganisms or their products initiate autophagy
Intracellular membranes capture them through PRRs or they are coated with Ubiquitin, which is the molecule that tags proteins for recycling by proteasomes. Instead of targeting proteins, ubiquitin “labels” microorganisms to be captured by a Phaphophore (free-floating membrane in the cytosol). The phagophore surrounds the microorganism and creates the double-membrane self-phagosome. The autophagosome, on the other hand, is created from intracellular membranes.
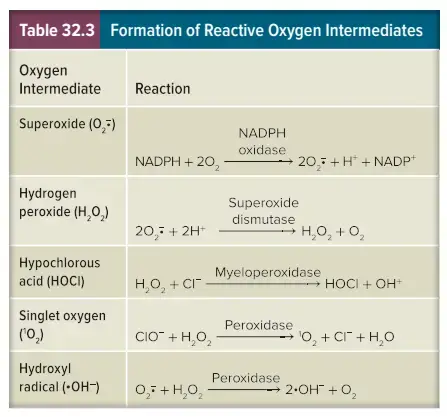
After microorganisms are enclosed in a autophagosome/phagosome, the lysosomes fuse with them, creating an autophagosome and phagolysosome. Lysosomes contain a range of hydrolases, including lysozyme and phospholipase. Acidic vacuolar pH increases the activity of these enzymes. These enzymes work together to destroy the microorganisms entrapped in them. Additionally, toxic reactive oxygen substances (ROS), such as superoxide radical, hydrogen peroxide, H2O2, singlet oxygen (1O2) and hydroxyl radicals (*OH), are also produced. Myeloperoxidase is a heme-protein enzyme that catalyzes production of hypochlorous acids (bleach) in neutrophils. The recruitment of an NADPH-oxidase to phagosome membrane or autosome membrane is the first step in the generation of ROS. This enzyme uses molecular oxygen (molecular oxygen) to oxidize NADPH into NADP+ and create superoxide radical. The respiratory burst, which is the simultaneous release of ROS and O2 consumption, is also known as O2 or ROS.
Reactive nitrogen species (RNS) have been also formed by neutrophils, mast cells, and macrophages. These molecules include nitric dioxide (NO) as well as its oxidized forms, such as nitrite and nitrate. RNS can be very powerful cytotoxic agents. Nitric oxide is the most potent RNS. By complexing with iron in electron transportation proteins, it can block cell respiration. RNS is used by Macrophages to destroy a variety infectious agents and also kill tumor cells.
Neutrophil granules contain a variety of other microbicidal substances such as cationic peptides, the bactericidal permeabilityincreasing protein (BPI), and broad-spectrum antimicrobial peptides, including defensins. These substances are stored in compartments for extracellular secretion and delivery to phagocytic vessels. Some viruses, Gram-positive and Gram–negative bacteria, yeasts, molds, and others are all possible microbial targets.
What is Exocytosis?
The phagocyte can do one of two things once the microbial invaders are killed and broken down into small antigenic pieces. Exocytosis, step 5 in the fiure, is when the cell might expel the microbial particles. This is basically a reverse process of phagocytic. The phagolysosome combines with the cell membrane and releases extracellular microbial fragments. Neutrophils are devoted to phagocytosis, exocytosing their cargo until they exhaust themselves and die. Macrophages and dendritic cell become antigen-presenting.
The phagolysosome passes microbial fragments to the endoplasmic retina. The peptides of the microbial fragments get combined with glycoproteins (major histocompatibility proteins (MHC)), destined for the cell membrane. This is done to insert a microbe-specific protein in the MHC. The peptide faces the outside when the MHC-peptide combination is attached to the macrophage plasma membrane or DC. This initiates the process of antigen presentation. It is crucial because it allows DCs and macrophages show or “presents” microbial antibodies to lymphocytes. This triggers the activation of adaptive immunity.
Significance of Phagocytosis
Phagocytosis, a cellular process of engulfing and digesting particles, plays a pivotal role in maintaining the health and functionality of multicellular organisms. Its significance extends beyond mere defense against pathogens, encompassing various facets of cellular and tissue dynamics. Here, we explore the profound implications of phagocytosis in the context of tissue development, repair, and overall organismal health.
- Cellular Turnover and Tissue Repair: Multicellular organisms are in a perpetual state of cellular turnover. As cells reach the end of their functional lifespan, known as senescence, they must be replaced to ensure the continuity of tissue function. This is especially crucial during developmental phases and periods of tissue repair. Phagocytosis facilitates the removal of these senescent cells, paving the way for the generation of new, functional cells.
- Role in Tissue Development and Remodeling: Empirical studies have underscored the indispensable role of phagocytosis in tissue development and remodeling. As tissues evolve and adapt, there’s a need for the orchestrated removal of specific cells to shape and refine the tissue structure. Phagocytosis aids in this selective removal, ensuring that tissues develop and remodel in a manner conducive to their intended function.
- Clearance of Cellular Debris: Over time, tissues accumulate cellular debris, remnants of old and damaged cells. If left unchecked, this debris can instigate tissue damage through a process known as secondary necrosis. Phagocytosis acts as a biological sanitation system, clearing away this debris and preventing potential harm. By doing so, it safeguards tissues from the deleterious effects of accumulated waste.
- Defense Against Pathogens: One of the primary roles of phagocytosis is in the immune response. Phagocytes, such as macrophages and neutrophils, actively seek out and engulf foreign invaders like bacteria, viruses, and fungi. This not only neutralizes immediate threats but also aids in antigen presentation, a process that activates other immune cells to recognize and combat specific pathogens.
- Maintenance of Homeostasis: By clearing out dead cells and cellular debris, phagocytosis contributes to the maintenance of tissue and systemic homeostasis. This ensures that the internal environment remains stable and conducive for cellular functions.
- Regulation of Inflammation: Phagocytosis plays a role in modulating inflammation. By removing dead cells and pathogens, it prevents the prolonged release of inflammatory mediators, thereby curbing excessive inflammatory responses which could be detrimental to the host.
- Facilitation of Tissue Regeneration: In the aftermath of tissue injury, phagocytosis clears away damaged cells and debris, creating a conducive environment for tissue regeneration and healing. This is vital for restoring the function of injured tissues.
- Role in Apoptosis: Apoptosis, or programmed cell death, is a controlled cellular suicide mechanism. Phagocytes recognize apoptotic cells and swiftly engulf them, preventing the release of potentially harmful cellular contents into the surrounding tissue.
- Nutrient Recycling: In single-celled organisms like amoebas, phagocytosis is a primary mode of nutrient acquisition. By engulfing nutrient-rich particles, these organisms can break them down and reuse the components for their metabolic processes.
- Interplay with Other Cellular Processes: The act of phagocytosis often triggers other cellular pathways, such as autophagy, where cells break down their own components. This interplay ensures that cells can adapt and respond to varying environmental conditions.
- Influence on Cellular Communication: Post-phagocytosis, cells often release cytokines and other signaling molecules that influence the behavior of neighboring cells. This aids in coordinating multicellular responses to various stimuli.
In summation, phagocytosis is not merely a defensive mechanism but a multifaceted process integral to the health, development, and maintenance of multicellular organisms. Its role in cellular turnover, tissue shaping, and debris clearance underscores its paramount importance in ensuring the optimal functionality and longevity of tissues.
Phagocytosis vs Pinocytosis
Both phagocytosis and pinocytosis are forms of endocytosis, where cells internalize extracellular material. However, they differ in their mechanisms, functions, and the materials they target. Here’s a comparative analysis of the two processes:
- Definition:
- Phagocytosis: Often termed “cell eating,” it involves the engulfment of large particles, such as bacteria, dead cells, or other sizable foreign materials.
- Pinocytosis: Known as “cell drinking,” this process involves the uptake of small solute particles and liquids from the extracellular environment.
- Vesicle Size:
- Phagocytosis: Forms larger vesicles known as phagosomes.
- Pinocytosis: Results in smaller vesicles, often termed pinosomes.
- Function:
- Phagocytosis: Primarily functions in defense mechanisms, where immune cells engulf and destroy pathogens. It also plays a role in tissue remodeling by removing dead or damaged cells.
- Pinocytosis: Mainly involved in nutrient uptake, allowing cells to sample and internalize extracellular fluids and their dissolved solutes.
- Mechanism:
- Phagocytosis: The cell extends its plasma membrane, forming pseudopods that surround and engulf the target particle.
- Pinocytosis: The cell membrane invaginates, forming a cup-like structure that pinches off to internalize fluid and solute particles.
- Specificity:
- Phagocytosis: Often a targeted process, where specific cells (like macrophages or neutrophils) recognize and engulf specific particles based on receptor-ligand interactions.
- Pinocytosis: Generally a non-specific process, where cells continuously internalize extracellular fluid and its contents.
- Energy Requirement:
- Phagocytosis: Requires considerable energy, given the larger size of the particles being engulfed.
- Pinocytosis: Also energy-dependent, but typically less energy-intensive than phagocytosis.
- Cell Types:
- Phagocytosis: Primarily performed by specialized cells like macrophages, neutrophils, and dendritic cells.
- Pinocytosis: Common in many cell types, especially those involved in nutrient absorption, such as cells in the kidney and intestine.
In summary, while both phagocytosis and pinocytosis are essential for cellular function and survival, they cater to different cellular needs. Phagocytosis focuses on the removal or destruction of large particles, often for defense, while pinocytosis ensures the continuous sampling and uptake of extracellular fluids and nutrients.
Endocytosis vs Phagocytosis
Endocytosis and phagocytosis are both processes by which cells internalize substances from their external environment. However, they differ in their mechanisms, functions, and the types of materials they transport. Here’s a comparison of the two:
Endocytosis
- Definition: A cellular process in which substances are brought into the cell by engulfing them in an energy-using process.
- Types:
- Pinocytosis: “Cell drinking”; the cell engulfs small particles or liquids.
- Receptor-mediated endocytosis: Specific molecules are taken in after they bind to a receptor.
- Vesicle Formation: Forms small vesicles.
- Function: Used to take in specific molecules or fluids.
- Materials Transported: Can transport proteins, lipids, and other molecules.
- Mechanism: Involves the inward folding of the cell membrane to form a vesicle.
Phagocytosis
- Definition: A type of endocytosis in which a cell engulfs large particles or whole cells, either as a defense mechanism or as a means to obtain food.
- Types: Not further categorized like endocytosis.
- Vesicle Formation: Forms large vesicles known as phagosomes.
- Function: Used by immune cells to engulf and destroy pathogens or by other cells to engulf other large particles.
- Materials Transported: Can transport larger particles, such as bacteria, dead cell debris, or other large particles.
- Mechanism: The cell membrane reaches out with pseudopodia (arm-like extensions) to surround and engulf the particle.
In summary, while both endocytosis and phagocytosis are mechanisms for internalizing substances, endocytosis is more general and can be receptor-mediated or not, while phagocytosis is specific to the engulfment of large particles or cells.
Phagocytosis Example
Phagocytosis is a crucial cellular process where cells engulf large particles, such as bacteria, dead cells, or other foreign debris. This process is primarily carried out by specialized cells in the immune system. Here are some examples of phagocytosis:
- White Blood Cells Engulfing Bacteria: One of the primary roles of certain white blood cells, like neutrophils and macrophages, is to identify, engulf, and destroy pathogenic bacteria that enter the body. Once inside the phagocyte, the bacteria are enclosed in a vesicle called a phagosome, which then fuses with a lysosome. The lysosome contains enzymes that digest and destroy the bacteria.
- Macrophages Clearing Dead Cells: Macrophages play a vital role in tissue repair and maintenance. They engulf and break down dead or dying cells, ensuring that tissues remain free of debris.
- Removal of Aged Red Blood Cells: The spleen contains macrophages that identify and engulf old, damaged, or misshapen red blood cells, breaking them down and recycling their components.
- Engulfment of Foreign Particles: If dust or other foreign particles are inhaled into the lungs, alveolar macrophages (found in the air sacs of the lungs) can engulf and break down these particles, helping to keep the lungs clean.
- Dictyostelium Discoideum Feeding: This is a type of soil-living amoeba. It feeds by engulfing bacteria through phagocytosis.
- Amoeba Engulfing Food: Amoebas, which are single-celled organisms, feed by extending pseudopods (arm-like extensions) around food particles, engulfing them, and then digesting them internally.
- Defense Mechanism in Simple Organisms: Many simple aquatic organisms, like Paramecium, use phagocytosis as a defense mechanism to engulf and digest potential pathogens.
- Formation of Osteoclasts: In the bone, large cells called osteoclasts form by the fusion of smaller cells. These osteoclasts then use a form of phagocytosis to resorb bone tissue, playing a crucial role in bone remodeling.
- Engulfment of Apoptotic Bodies: During the process of apoptosis (programmed cell death), cells that are dying are packaged into apoptotic bodies. These bodies are then recognized and engulfed by phagocytic cells, ensuring that the components of the dead cell are safely removed and recycled.
- Clearance of Immune Complexes: Macrophages and neutrophils play a role in clearing immune complexes (combinations of antibodies and antigens) from the bloodstream, preventing these complexes from depositing in tissues and causing damage.
These examples highlight the diverse roles of phagocytosis in both maintaining health and responding to infections or injuries.
Some Important Questions
1. What is phagocytosis?
Phagocytosis is a cellular process where a cell engulfs large particles or cells, either to clean up debris or to fight infections. It is a primary mechanism used by the immune system to remove pathogens and cell debris.
2. Is phagocytosis active or passive?
Phagocytosis is an active process that requires energy, typically derived from ATP, to engulf particles or cells.
3. What is the difference between phagocytosis and pinocytosis?
While both are forms of endocytosis, phagocytosis involves the engulfing of large particles or cells, whereas pinocytosis involves the ingestion of liquid into a cell by the budding of small vesicles from the cell membrane.
4. Does phagocytosis require energy?
Yes, phagocytosis is an energy-dependent process and requires ATP to proceed.
5. How to pronounce phagocytosis?
Phagocytosis is pronounced as “fag-oh-sy-TOH-sis”.
6. Which activated T cells increase phagocytosis and antibody formation?
Helper T cells, when activated, can stimulate B cells to produce antibodies and can also enhance the phagocytic activity of macrophages.
7. How does phagocytosis work?
Phagocytosis begins when the cell recognizes a foreign particle. The cell membrane then extends around the particle, forming a vesicle known as a phagosome. This phagosome then fuses with a lysosome, where enzymes digest the engulfed material.
8. Which external structure protects bacteria from phagocytosis?
The capsule, a gelatinous outer layer found in some bacteria, provides resistance against phagocytosis.
9. Is phagocytosis endocytosis?
Yes, phagocytosis is a type of endocytosis. Endocytosis is a broader term that refers to the process by which cells take in materials from their environment by engulfing them in a vesicle.
10. What is an example of phagocytosis?
An example of phagocytosis is when a macrophage, a type of white blood cell, engulfs and digests a bacterial cell that has entered the body.
11. Does phagocytosis require ATP?
Yes, phagocytosis requires energy in the form of ATP to engulf particles or cells.
12. What human cells carry out phagocytosis?
Macrophages, neutrophils, and dendritic cells are primary human cells that perform phagocytosis.
13. What is the process of phagocytosis?
The process of phagocytosis involves the recognition of the target particle, engulfment of the particle by wrapping it in a segment of the cell’s plasma membrane, and the formation of a vesicle. This vesicle then fuses with a lysosome, leading to the digestion of the engulfed material.
14. How does phagocytosis contribute to wound healing?
Phagocytosis plays a crucial role in wound healing by removing dead cells, bacteria, and other debris from the wound site, which aids in preventing infection and promoting tissue repair.
15. What cells perform phagocytosis?
Cells that perform phagocytosis include macrophages, neutrophils, and dendritic cells.
16. What is the function of phagocytosis?
The primary function of phagocytosis is to eliminate pathogens and cell debris. It plays a vital role in the immune response, helping to clear infections and also in tissue repair and remodeling.
17. Where does phagocytosis occur?
Phagocytosis occurs in various tissues throughout the body, especially where immune cells are present, such as the spleen, lymph nodes, and in tissues during an infection or inflammation.
18. Which cells specialize in phagocytosis?
Macrophages and neutrophils are cells that specialize in phagocytosis.
19. How do pinocytosis and phagocytosis differ?
Pinocytosis is the cellular process of ingesting liquids, while phagocytosis involves the ingestion of large particles or cells. Pinocytosis results in smaller vesicles compared to the larger phagosomes formed during phagocytosis.
20. Is phagocytosis a type of endocytosis?
Yes, phagocytosis is a specific form of endocytosis where large particles or cells are engulfed by the cell.
21. Is phagocytosis an innate immune response?
Yes, phagocytosis is a component of the innate immune response, providing a first line of defense against pathogens.
22. Is phagocytosis innate or adaptive immunity?
Phagocytosis is a part of the innate immune system, providing immediate but non-specific defense against pathogens.
23. What activates phagocytosis?
Phagocytosis is activated by the presence of pathogens or foreign particles, and also by certain signaling molecules released during an immune response.
24. What are the steps of phagocytosis?
The steps of phagocytosis include recognition, adherence, ingestion, fusion with lysosomes, and digestion of the engulfed material.
25. What causes phagocytosis?
The presence of foreign particles, pathogens, or signals from other immune cells can trigger phagocytosis.
26. What cells are involved in phagocytosis?
Macrophages, neutrophils, and dendritic cells are the primary cells involved in phagocytosis.
27. What enhances phagocytosis?
Opsonization, the process where pathogens are coated with antibodies or complement proteins, enhances phagocytosis by making the pathogens more recognizable to phagocytic cells.
28. What happens after phagocytosis?
After phagocytosis, the engulfed material is contained within a vesicle called a phagosome. This phagosome fuses with a lysosome, leading to the digestion and destruction of the engulfed material.
29. What happens during phagocytosis?
During phagocytosis, the cell recognizes and adheres to a target particle, engulfs it to form a phagosome, and then digests the contents after the phagosome fuses with a lysosome.
30. What is frustrated phagocytosis?
Frustrated phagocytosis occurs when a phagocytic cell attempts to engulf a particle or organism that is too large to be ingested, leading to the release of reactive oxygen species and inflammatory mediators.
31. What is phagocytosis in the immune system?
In the immune system, phagocytosis is a defense mechanism where cells like macrophages and neutrophils engulf and destroy pathogens or foreign particles.
32. What is the purpose of phagocytosis?
The primary purpose of phagocytosis is to defend the body against infections by engulfing and destroying pathogens. It also plays a role in tissue repair and the removal of cell debris.
33. What is the role of lysosomes in phagocytosis?
Lysosomes contain digestive enzymes that break down the engulfed material. When a phagosome fuses with a lysosome, these enzymes are released into the phagosome, leading to the digestion of the engulfed material.
34. What is the role of TLRs in phagocytosis?
Toll-like receptors (TLRs) are a type of pattern recognition receptor that can recognize specific molecular patterns on pathogens. When TLRs are activated, they can enhance the phagocytic activity of immune cells and promote the production of pro-inflammatory cytokines.
FAQ
What is phagocytosis?
Phagocytosis is a process by which cells engulf and internalize particles, such as bacteria, cellular debris, or foreign substances, for degradation and elimination.
Which cells are involved in phagocytosis?
Phagocytosis is primarily carried out by specialized cells called phagocytes, including macrophages, neutrophils, and dendritic cells. However, other cell types, such as amoebas, can also perform phagocytosis.
What triggers phagocytosis?
Phagocytosis can be triggered by the presence of foreign particles, pathogens, cellular debris, or chemical signals released by damaged cells. These signals activate phagocytes and initiate the phagocytic process.
What are the steps involved in phagocytosis?
The steps of phagocytosis include activation, chemotaxis, attachment, ingestion, fusion with lysosomes, digestion, and exocytosis. These steps collectively facilitate the engulfment, degradation, and elimination of the ingested particles.
What is the role of receptors in phagocytosis?
Receptors on the surface of phagocytes recognize and bind to specific molecules on the target particle, enabling the phagocyte to attach to and engulf the particle efficiently. These receptors play a crucial role in the recognition and initiation of phagocytosis.
How do phagocytes destroy engulfed particles?
Upon fusion with lysosomes, phagosomes form phagolysosomes. Within the phagolysosome, the particle is exposed to a variety of hydrolytic enzymes, including proteases and nucleases, which degrade the particle into smaller molecules.
Can phagocytes kill bacteria?
Yes, phagocytes can eliminate bacteria through a combination of mechanisms. Besides phagocytosis, phagocytes release toxic molecules such as reactive oxygen species (ROS) and antimicrobial peptides, which can directly kill bacteria.
What happens to the waste material after digestion?
After digestion, the indigestible components or waste materials are packaged into vesicles and expelled from the cell through a process called exocytosis. This ensures the removal of cellular waste products from the phagocytic cell.
Are there any diseases related to phagocytosis dysfunction?
Yes, phagocytosis dysfunction can lead to various disorders. For example, deficiencies in phagocytic cells can result in immunodeficiency disorders, making individuals more susceptible to infections. Additionally, impaired phagocytosis can contribute to inflammatory conditions and autoimmune diseases.
Is phagocytosis only involved in the immune system?
Although phagocytosis is a crucial mechanism in the immune system for eliminating pathogens, it is not limited to immune cells. Phagocytic activity can also be observed in other cell types, such as amoebas, which use phagocytosis for nutrient intake and locomotion.
Which of the following is not an event of phagocytosis?A. ChemotaxisB. Phagolysosome formationC. DestructionD. IngestionE. They are all events that occur during phagocytosis.
They are all events that occur during phagocytosis
Which External Structure Protects bacteria from phagocytosis?
The external structure that protects bacteria from phagocytosis is the “capsule” or “polysaccharide capsule.” The capsule is a gelatinous layer that surrounds some bacterial cells and provides resistance to phagocytosis by host immune cells.
References
- Biology Dictionary. (n.d.). Phagocytosis. https://biologydictionary.net/phagocytosis
- Frontiers in Immunology. (2020). [Title of the article]. https://www.frontiersin.org/articles/10.3389/fimmu.2020.01066
- Encyclopædia Britannica. (n.d.). Phagocytosis. https://www.britannica.com/science/phagocytosis
- Wikipedia. (n.d.). Phagocytosis. https://en.wikipedia.org/wiki/Phagocytosis
- NCBI. (n.d.). [Title of the article]. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5485277/
- NCBI. (n.d.). [Title of the book or section]. https://www.ncbi.nlm.nih.gov/books/NBK556043/
- Khan Academy. (n.d.). Phagocytosis. https://www.khanacademy.org/test-prep/mcat/cells/transport-across-a-cell-membrane/a/phagocytosis
- ScienceDirect. (n.d.). Phagocytosis. https://www.sciencedirect.com/topics/medicine-and-dentistry/phagocytosis
- British Society for Immunology. (n.d.). Phagocytosis. https://www.immunology.org/public-information/bitesized-immunology/systems-processes/phagocytosis
- TeachMePhysiology. (n.d.). Phagocytosis. https://teachmephysiology.com/immune-system/innate-immune-system/phagocytosis/
- Royal Society Publishing. (2020). [Title of the article]. https://royalsocietypublishing.org/doi/10.1098/rsfs.2020.0019
- Nature Reviews Immunology. (2023). [Title of the article]. https://www.nature.com/articles/s41577-023-00921-6
- JoVE. (n.d.). Phagocytosis. https://www.jove.com/science-education/10710/phagocytosis
- JoVE. (n.d.). Phagocytosis. https://www.jove.com/science-education/10710/phagocytosis (Note: This link is repeated, so it’s listed twice)
- Technology Networks. (n.d.). Phagocytosis vs. Pinocytosis: Definition and Function. https://www.technologynetworks.com/immunology/articles/phagocytosis-vs-pinocytosis-definition-and-function-343544
Thank You. This really helped me as a reference for my college project.
Mundhe, R. (2023) Phagocytosis – how immune system fights off infections in 6 steps, Biology Ideas. Biology Ideas. Available at: Biology Ideas (Accessed: March 20, 2023).